Abstract
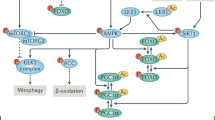
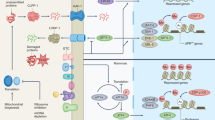
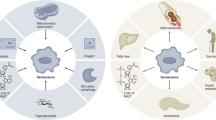
Here we review the environmental and genetic manipulations that link cellular and organismal metabolism to aging. In particular, we explore how nutrients are sensed and how various intracellular energy nodes seem to coordinate distinct metabolic alterations linked to extended longevity. In addition, the role of mitochondria as both a metabolic and signaling organelle is discussed. Finally, we review a host of new targeted pharmacological approaches that attempt to exploit the connection between aging and metabolism to treat a wide range of age-related diseases. Together, these insights are beginning to reveal answers to century-old mysteries and are providing a future road map for the rational extension of lifespan.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Debbie Maizels/Nature Publishing Group



Debbie Maizels/Nature Publishing Group
Similar content being viewed by others
References
Schäfer, D. Aging, longevity, and diet: historical remarks on calorie intake reduction. Gerontology 51, 126–130 (2005).
Fontana, L. & Partridge, L. Promoting health and longevity through diet: from model organisms to humans. Cell 161, 106–118 (2015).
Dillin, A. et al. Rates of behavior and aging specified by mitochondrial function during development. Science 298, 2398–2401 (2002).
Lee, S.S. et al. A systematic RNAi screen identifies a critical role for mitochondria in C. elegans longevity. Nat. Genet. 33, 40–48 (2003).
Trifunovic, A. et al. Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 429, 417–423 (2004).
Kujoth, G.C. et al. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science 309, 481–484 (2005).
Riera, C.E. & Dillin, A. Tipping the metabolic scales towards increased longevity in mammals. Nat. Cell Biol. 17, 196–203 (2015).
Speakman, J.R. & Mitchell, S.E. Caloric restriction. Mol. Aspects Med. 32, 159–221 (2011).
Brown-Borg, H.M., Borg, K.E., Meliska, C.J. & Bartke, A. Dwarf mice and the ageing process. Nature 384, 33 (1996).
Mattison, J.A. et al. Studies of aging in Ames dwarf mice: Effects of caloric restriction. J. Am. Aging Assoc. 23, 9–16 (2000).
Bartke, A. et al. Extending the lifespan of long-lived mice. Nature 414, 412 (2001).
Heilbronn, L.K. et al. Effect of 6-month calorie restriction on biomarkers of longevity, metabolic adaptation, and oxidative stress in overweight individuals: a randomized controlled trial. J. Am. Med. Assoc. 295, 1539–1548 (2006).
Ravussin, E. et al. A 2-year randomized controlled trial of human caloric restriction: feasibility and effects on predictors of health span and longevity. J. Gerontol. A Biol. Sci. Med. Sci. 70, 1097–1104 (2015).
Bertrand, H.A., Lynd, F.T., Masoro, E.J. & Yu, B.P. Changes in adipose mass and cellularity through the adult life of rats fed ad libitum or a life-prolonging restricted diet. J. Gerontol. 35, 827–835 (1980).
Barzilai, N., Banerjee, S., Hawkins, M., Chen, W. & Rossetti, L. Caloric restriction reverses hepatic insulin resistance in aging rats by decreasing visceral fat. J. Clin. Invest. 101, 1353–1361 (1998).
Liao, C.Y., Rikke, B.A., Johnson, T.E., Diaz, V. & Nelson, J.F. Genetic variation in the murine lifespan response to dietary restriction: from life extension to life shortening. Aging Cell 9, 92–95 (2010).
Liao, C.Y. et al. Fat maintenance is a predictor of the murine lifespan response to dietary restriction. Aging Cell 10, 629–639 (2011).
Greenberg, J.A. & Boozer, C.N. Metabolic mass, metabolic rate, caloric restriction, and aging in male Fischer 344 rats. Mech. Ageing Dev. 113, 37–48 (2000).
Selman, C. et al. Energy expenditure of calorically restricted rats is higher than predicted from their altered body composition. Mech. Ageing Dev. 126, 783–793 (2005).
Bartke, A. & Westbrook, R. Metabolic characteristics of long-lived mice. Front. Genet. 3, 288 (2012).
Petersen, K.F. et al. Effect of aging on muscle mitochondrial substrate utilization in humans. Proc. Natl. Acad. Sci. USA 112, 11330–11334 (2015).
Huffman, K.M. et al. Caloric restriction alters the metabolic response to a mixed-meal: results from a randomized, controlled trial. PLoS ONE 7, e28190 (2012).
Willette, A.A. et al. Interleukin-8 and interleukin-10, brain volume and microstructure, and the influence of calorie restriction in old rhesus macaques. Age (Dordr.) 35, 2215–2227 (2013).
Youm, Y.H. et al. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome–mediated inflammatory disease. Nat. Med. 21, 263–269 (2015).
Yang, H., Youm, Y.H. & Dixit, V.D. Inhibition of thymic adipogenesis by caloric restriction is coupled with reduction in age-related thymic involution. J. Immunol. 183, 3040–3052 (2009).
Masoro, E.J. Overview of caloric restriction and ageing. Mech. Ageing Dev. 126, 913–922 (2005).
Colman, R.J. et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 325, 201–204 (2009).
Mattison, J.A. et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 489, 318–321 (2012).
Albert, V. & Hall, M.N. mTOR signaling in cellular and organismal energetics. Curr. Opin. Cell Biol. 33, 55–66 (2015).
Vellai, T. et al. Genetics: influence of TOR kinase on lifespan in C. elegans. Nature 426, 620 (2003).
Kapahi, P. et al. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr. Biol. 14, 885–890 (2004).
Lamming, D.W. et al. Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science 335, 1638–1643 (2012).
Wu, J.J. et al. Increased mammalian lifespan and a segmental and tissue-specific slowing of aging after genetic reduction of mTOR expression. Cell Rep. 4, 913–920 (2013).
Kaeberlein, M. et al. Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science 310, 1193–1196 (2005).
Hansen, M. et al. Lifespan extension by conditions that inhibit translation in Caenorhabditis elegans. Aging Cell 6, 95–110 (2007).
Johnson, S.C., Rabinovitch, P.S. & Kaeberlein, M. mTOR is a key modulator of ageing and age-related disease. Nature 493, 338–345 (2013).
Desai, B.N., Myers, B.R. & Schreiber, S.L. FKBP12-rapamycin–associated protein associates with mitochondria and senses osmotic stress via mitochondrial dysfunction. Proc. Natl. Acad. Sci. USA 99, 4319–4324 (2002).
Schieke, S.M. et al. The mammalian target of rapamycin (mTOR) pathway regulates mitochondrial oxygen consumption and oxidative capacity. J. Biol. Chem. 281, 27643–27652 (2006).
Ramanathan, A. & Schreiber, S.L. Direct control of mitochondrial function by mTOR. Proc. Natl. Acad. Sci. USA 106, 22229–22232 (2009).
Bonawitz, N.D., Chatenay-Lapointe, M., Pan, Y. & Shadel, G.S. Reduced TOR signaling extends chronological life span via increased respiration and upregulation of mitochondrial gene expression. Cell Metab. 5, 265–277 (2007).
Bentzinger, C.F. et al. Skeletal muscle-specific ablation of raptor, but not of rictor, causes metabolic changes and results in muscle dystrophy. Cell Metab. 8, 411–424 (2008).
Polak, P. et al. Adipose-specific knockout of raptor results in lean mice with enhanced mitochondrial respiration. Cell Metab. 8, 399–410 (2008).
Yilmaz, Ö.H. et al. mTORC1 in the Paneth cell niche couples intestinal stem-cell function to calorie intake. Nature 486, 490–495 (2012).
Hirschey, M.D. & Zhao, Y. Metabolic regulation by lysine malonylation, succinylation and glutarylation. Mol. Cell. Proteomics 14, 2308–2315 (2015).
Lin, S.J., Defossez, P.A. & Guarente, L. Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science 289, 2126–2128 (2000).
Rogina, B. & Helfand, S.L. Sir2 mediates longevity in the fly through a pathway related to calorie restriction. Proc. Natl. Acad. Sci. USA 101, 15998–16003 (2004).
Kaeberlein, M., Kirkland, K.T., Fields, S. & Kennedy, B.K. Sir2-independent life span extension by calorie restriction in yeast. PLoS Biol. 2, e296 (2004).
Nemoto, S., Fergusson, M.M. & Finkel, T. Nutrient availability regulates SIRT1 through a Forkhead-dependent pathway. Science 306, 2105–2108 (2004).
Cohen, H.Y. et al. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science 305, 390–392 (2004).
Chen, D. et al. Tissue-specific regulation of SIRT1 by calorie restriction. Genes Dev. 22, 1753–1757 (2008).
Mercken, E.M. et al. SIRT1 but not its increased expression is essential for lifespan extension in caloric-restricted mice. Aging Cell 13, 193–196 (2014).
Satoh, A. et al. Sirt1 extends life span and delays aging in mice through the regulation of Nk2 homeobox 1 in the DMH and LH. Cell Metab. 18, 416–430 (2013).
Kanfi, Y. et al. The sirtuin SIRT6 regulates lifespan in male mice. Nature 483, 218–221 (2012).
Ahn, B.H. et al. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc. Natl. Acad. Sci. USA 105, 14447–14452 (2008).
Hirschey, M.D. et al. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 464, 121–125 (2010).
Qiu, X., Brown, K., Hirschey, M.D., Verdin, E. & Chen, D. Calorie restriction reduces oxidative stress by SIRT3-mediated SOD2 activation. Cell Metab. 12, 662–667 (2010).
Tao, R. et al. Sirt3-mediated deacetylation of evolutionarily conserved lysine 122 regulates MnSOD activity in response to stress. Mol. Cell 40, 893–904 (2010).
Hebert, A.S. et al. Calorie restriction and SIRT3 trigger global reprogramming of the mitochondrial protein acetylome. Mol. Cell 49, 186–199 (2013).
Someya, S. et al. Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell 143, 802–812 (2010).
Barger, J.L. et al. A conserved transcriptional signature of delayed aging and reduced disease vulnerability is partially mediated by SIRT3. PLoS ONE 10, e0120738 (2015).
Hardie, D.G. AMPK: positive and negative regulation, and its role in whole-body energy homeostasis. Curr. Opin. Cell Biol. 33, 1–7 (2015).
Greer, E.L. et al. An AMPK-FOXO pathway mediates longevity induced by a novel method of dietary restriction in C. elegans. Curr. Biol. 17, 1646–1656 (2007).
Schulz, T.J. et al. Glucose restriction extends Caenorhabditis elegans life span by inducing mitochondrial respiration and increasing oxidative stress. Cell Metab. 6, 280–293 (2007).
Stenesen, D. et al. Adenosine nucleotide biosynthesis and AMPK regulate adult life span and mediate the longevity benefit of caloric restriction in flies. Cell Metab. 17, 101–112 (2013).
Burkewitz, K., Zhang, Y. & Mair, W.B. AMPK at the nexus of energetics and aging. Cell Metab. 20, 10–25 (2014).
Cantó, C. & Auwerx, J. Calorie restriction: is AMPK a key sensor and effector? Physiology (Bethesda) 26, 214–224 (2011).
Ulgherait, M., Rana, A., Rera, M., Graniel, J. & Walker, D.W. AMPK modulates tissue and organismal aging in a non-cell-autonomous manner. Cell Rep. 8, 1767–1780 (2014).
Meléndez, A. et al. Autophagy genes are essential for dauer development and life-span extension in C. elegans. Science 301, 1387–1391 (2003).
Morselli, E. et al. Caloric restriction and resveratrol promote longevity through the Sirtuin-1–dependent induction of autophagy. Cell Death Dis. 1, e10 (2010).
Ruckenstuhl, C. et al. Lifespan extension by methionine restriction requires autophagy-dependent vacuolar acidification. PLoS Genet. 10, e1004347 (2014).
Pyo, J.O. et al. Overexpression of Atg5 in mice activates autophagy and extends lifespan. Nat. Commun. 4, 2300 (2013).
Lapierre, L.R. et al. The TFEB orthologue HLH-30 regulates autophagy and modulates longevity in Caenorhabditis elegans. Nat. Commun. 4, 2267 (2013).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R.A. C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Kimura, K.D., Tissenbaum, H.A., Liu, Y. & Ruvkun, G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science 277, 942–946 (1997).
Murphy, C.T. et al. Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424, 277–283 (2003).
Tatar, M. et al. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 292, 107–110 (2001).
Holzenberger, M. et al. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature 421, 182–187 (2003).
Xu, J. et al. Longevity effect of IGF-1R+/− mutation depends on genetic background-specific receptor activation. Aging Cell 13, 19–28 (2014).
Taguchi, A., Wartschow, L.M. & White, M.F. Brain IRS2 signaling coordinates life span and nutrient homeostasis. Science 317, 369–372 (2007).
Conover, C.A. & Bale, L.K. Loss of pregnancy-associated plasma protein A extends lifespan in mice. Aging Cell 6, 727–729 (2007).
Suh, Y. et al. Functionally significant insulin-like growth factor I receptor mutations in centenarians. Proc. Natl. Acad. Sci. USA 105, 3438–3442 (2008).
Wheeler, H.E. & Kim, S.K. Genetics and genomics of human ageing. Phil. Trans. R. Soc. Lond. B 366, 43–50 (2011).
Brown-Borg, H.M. The somatotropic axis and longevity in mice. Am. J. Physiol. Endocrinol. Metab. 309, E503–E510 (2015).
Zhang, G. et al. Hypothalamic programming of systemic ageing involving IKK-β, NF-κB and GnRH. Nature 497, 211–216 (2013).
Madeo, F., Zimmermann, A., Maiuri, M.C. & Kroemer, G. Essential role for autophagy in lifespan extension. J. Clin. Invest. 125, 85–93 (2015).
Lee, I.H. et al. A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proc. Natl. Acad. Sci. USA 105, 3374–3379 (2008).
Short, K.R. et al. Decline in skeletal muscle mitochondrial function with aging in humans. Proc. Natl. Acad. Sci. USA 102, 5618–5623 (2005).
Rea, S.L., Ventura, N. & Johnson, T.E. Relationship between mitochondrial electron transport chain dysfunction, development, and life extension in Caenorhabditis elegans. PLoS Biol. 5, e259 (2007).
Liu, X. et al. Evolutionary conservation of the clk-1–dependent mechanism of longevity: loss of mclk1 increases cellular fitness and lifespan in mice. Genes Dev. 19, 2424–2434 (2005).
Dell′Agnello, C. et al. Increased longevity and refractoriness to Ca2+-dependent neurodegeneration in Surf1 knockout mice. Hum. Mol. Genet. 16, 431–444 (2007).
Chang, H.W., Shtessel, L. & Lee, S.S. Collaboration between mitochondria and the nucleus is key to long life in Caenorhabditis elegans. Free Radic. Biol. Med. 78, 168–178 (2015).
Lee, S.J., Hwang, A.B. & Kenyon, C. Inhibition of respiration extends C. elegans life span via reactive oxygen species that increase HIF-1 activity. Curr. Biol. 20, 2131–2136 (2010).
Hwang, A.B. et al. Feedback regulation via AMPK and HIF-1 mediates ROS-dependent longevity in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 111, E4458–E4467 (2014).
Ventura, N. et al. p53/CEP-1 increases or decreases lifespan, depending on level of mitochondrial bioenergetic stress. Aging Cell 8, 380–393 (2009).
Walter, L., Baruah, A., Chang, H.W., Pace, H.M. & Lee, S.S. The homeobox protein CEH-23 mediates prolonged longevity in response to impaired mitochondrial electron transport chain in C. elegans. PLoS Biol. 9, e1001084 (2011).
Yee, C., Yang, W. & Hekimi, S. The intrinsic apoptosis pathway mediates the pro-longevity response to mitochondrial ROS in C. elegans. Cell 157, 897–909 (2014).
Schmeisser, S. et al. Neuronal ROS signaling rather than AMPK/sirtuin–mediated energy sensing links dietary restriction to lifespan extension. Mol. Metab. 2, 92–102 (2013).
Zhang, H., Davies, K.J. & Forman, H.J. Oxidative stress response and Nrf2 signaling in aging. Free Radic. Biol. Med. 88, 314–336 (2015).
Paek, J. et al. Mitochondrial SKN-1/Nrf mediates a conserved starvation response. Cell Metab. 16, 526–537 (2012).
Schmeisser, S. et al. Mitochondrial hormesis links low-dose arsenite exposure to lifespan extension. Aging Cell 12, 508–517 (2013).
Pearson, K.J. et al. Nrf2 mediates cancer protection but not prolongevity induced by caloric restriction. Proc. Natl. Acad. Sci. USA 105, 2325–2330 (2008).
Harman, D. Aging: a theory based on free radical and radiation chemistry. J. Gerontol. 11, 298–300 (1956).
Barja, G. The mitochondrial free radical theory of aging. Prog. Mol. Biol. Transl. Sci. 127, 1–27 (2014).
Yun, J. & Finkel, T. Mitohormesis. Cell Metab. 19, 757–766 (2014).
Ristow, M. Unraveling the truth about antioxidants: mitohormesis explains ROS-induced health benefits. Nat. Med. 20, 709–711 (2014).
Ristow, M. & Schmeisser, K. Mitohormesis: promoting health and lifespan by increased levels of reactive oxygen species (ROS). Dose Response 12, 288–341 (2014).
Durieux, J., Wolff, S. & Dillin, A. The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell 144, 79–91 (2011).
Owusu-Ansah, E., Song, W. & Perrimon, N. Muscle mitohormesis promotes longevity via systemic repression of insulin signaling. Cell 155, 699–712 (2013).
Jovaisaite, V. & Auwerx, J. The mitochondrial unfolded protein response-synchronizing genomes. Curr. Opin. Cell Biol. 33, 74–81 (2015).
Nargund, A.M., Pellegrino, M.W., Fiorese, C.J., Baker, B.M. & Haynes, C.M. Mitochondrial import efficiency of ATFS-1 regulates mitochondrial UPR activation. Science 337, 587–590 (2012).
Liu, Y., Samuel, B.S., Breen, P.C. & Ruvkun, G. Caenorhabditis elegans pathways that surveil and defend mitochondria. Nature 508, 406–410 (2014).
Pellegrino, M.W. et al. Mitochondrial UPR–regulated innate immunity provides resistance to pathogen infection. Nature 516, 414–417 (2014).
Houtkooper, R.H. et al. Mitonuclear protein imbalance as a conserved longevity mechanism. Nature 497, 451–457 (2013).
López-Lluch, G. et al. Calorie restriction induces mitochondrial biogenesis and bioenergetic efficiency. Proc. Natl. Acad. Sci. USA 103, 1768–1773 (2006).
Finley, L.W. et al. Skeletal muscle transcriptional coactivator PGC-1α mediates mitochondrial, but not metabolic, changes during calorie restriction. Proc. Natl. Acad. Sci. USA 109, 2931–2936 (2012).
Gomes, A.P. et al. Declining NAD+ induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell 155, 1624–1638 (2013).
Cuervo, A.M. et al. Autophagy and aging: the importance of maintaining “clean” cells. Autophagy 1, 131–140 (2005).
Palikaras, K., Lionaki, E. & Tavernarakis, N. Coordination of mitophagy and mitochondrial biogenesis during ageing in C. elegans. Nature 521, 525–528 (2015).
Rana, A., Rera, M. & Walker, D.W. Parkin overexpression during aging reduces proteotoxicity, alters mitochondrial dynamics, and extends lifespan. Proc. Natl. Acad. Sci. USA 110, 8638–8643 (2013).
Howitz, K.T. et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 425, 191–196 (2003).
Dang, W. The controversial world of sirtuins. Drug Discov. Today. Technol. 12, e9–e17 (2014).
Lagouge, M. et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 127, 1109–1122 (2006).
Pearson, K.J. et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab. 8, 157–168 (2008).
Jimenez-Gomez, Y. et al. Resveratrol improves adipose insulin signaling and reduces the inflammatory response in adipose tissue of rhesus monkeys on high-fat, high-sugar diet. Cell Metab. 18, 533–545 (2013).
Mattison, J.A. et al. Resveratrol prevents high fat/sucrose diet–induced central arterial wall inflammation and stiffening in nonhuman primates. Cell Metab. 20, 183–190 (2014).
Timmers, S. et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 14, 612–622 (2011).
Mitchell, S.J. et al. The SIRT1 activator SRT1720 extends lifespan and improves health of mice fed a standard diet. Cell Rep. 6, 836–843 (2014).
Harrison, D.E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
De Haes, W. et al. Metformin promotes lifespan through mitohormesis via the peroxiredoxin PRDX-2. Proc. Natl. Acad. Sci. USA 111, E2501–E2509 (2014).
Martin-Montalvo, A. et al. Metformin improves healthspan and lifespan in mice. Nat. Commun. 4, 2192 (2013).
Kasznicki, J., Sliwinska, A. & Drzewoski, J. Metformin in cancer prevention and therapy. Ann. Transl. Med. 2, 57 (2014).
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009).
Gupta, V.K. et al. Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nat. Neurosci. 16, 1453–1460 (2013).
Cantó, C., Menzies, K.J. & Auwerx, J. NAD+ metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metab. 22, 31–53 (2015).
Mouchiroud, L. et al. The NAD+/sirtuin pathway modulates longevity through activation of mitochondrial UPR and FOXO signaling. Cell 154, 430–441 (2013).
Yoshino, J., Mills, K.F., Yoon, M.J. & Imai, S. Nicotinamide mononucleotide, a key NAD+ intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 14, 528–536 (2011).
Cantó, C. et al. The NAD+ precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell Metab. 15, 838–847 (2012).
Imai, S. & Guarente, L. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 24, 464–471 (2014).
Austad, S.N. Methusaleh's zoo: how nature provides us with clues for extending human health span. J. Comp. Pathol. 142 (suppl. 1), S10–S21 (2010).
Wall, C.E. et al. High-fat diet and FGF21 cooperatively promote aerobic thermogenesis in mtDNA mutator mice. Proc. Natl. Acad. Sci. USA 112, 8714–8719 (2015).
Zhang, Y. et al. The starvation hormone, fibroblast growth factor-21, extends lifespan in mice. Elife 1, e00065 (2012).
Acknowledgements
I am grateful to members of my laboratory for their helpful suggestions, and in particular to I. Rovira for help with the manuscript. This work is supported by funds from the US National Institutes of Health Intramural Program and from The Leducq Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Finkel, T. The metabolic regulation of aging. Nat Med 21, 1416–1423 (2015). https://doi.org/10.1038/nm.3998
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3998
This article is cited by
-
Network pharmacology and molecular docking analysis on Shenfu Qiangxin indicate mTOR is a potential target to treat heart failure
European Journal of Medical Research (2024)
-
Targeted metabolomics reveals plasma biomarkers and metabolic alterations of the aging process in healthy young and older adults
GeroScience (2023)
-
Roles of NOLC1 in cancers and viral infection
Journal of Cancer Research and Clinical Oncology (2023)
-
The metabolic contribution of SKN-1/Nrf2 to the lifespan of Caenorhabditis elegans
Metabolomics (2023)
-
Immunometabolism in biofilm infection: lessons from cancer
Molecular Medicine (2022)