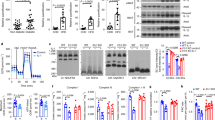
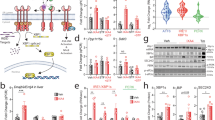
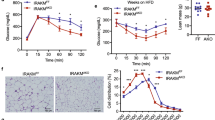
Abstract
Insulin resistance results from several pathophysiologic mechanisms, including chronic tissue inflammation and defective insulin signaling. We found that liver, muscle and adipose tissue exhibit higher levels of the chemotactic eicosanoid LTB4 in obese high-fat diet (HFD)–fed mice. Inhibition of the LTB4 receptor Ltb4r1, through either genetic or pharmacologic loss of function, led to an anti-inflammatory phenotype with protection from insulin resistance and hepatic steatosis. In vitro treatment with LTB4 directly enhanced macrophage chemotaxis, stimulated inflammatory pathways, reduced insulin-stimulated glucose uptake in L6 myocytes, and impaired insulin-mediated suppression of hepatic glucose output in primary mouse hepatocytes. This was accompanied by lower insulin-stimulated Akt phosphorylation and higher Irs-1/2 serine phosphorylation, and all of these events were dependent on Gαi and Jnk1, two downstream mediators of Ltb4r1 signaling. These observations elucidate a novel role of the LTB4–Ltb4r1 signaling pathway in hepatocyte and myocyte insulin resistance, and they show that in vivo inhibition of Ltb4r1 leads to robust insulin-sensitizing effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Haffner, S. & Taegtmeyer, H. Epidemic obesity and the metabolic syndrome. Circulation 108, 1541–1545 (2003).
Olefsky, J.M. & Glass, C.K. Macrophages, inflammation, and insulin resistance. Annu. Rev. Physiol. 72, 219–246 (2010).
Reaven, G.M. The insulin-resistance syndrome: definition and dietary approaches to treatment. Annu. Rev. Nutr. 25, 391–406 (2005).
Olefsky, J.M. et al. Cellular mechanisms of insulin resistance in non-insulin-dependent (type II) diabetes. Am. J. Med. 85, 86–105 (1988).
Kahn, S.E., Hull, R.L. & Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444, 840–846 (2006).
Samuel, V.T., Petersen, K.F. & Shulman, G.I. Lipid-induced insulin resistance: unravelling the mechanism. Lancet 375, 2267–2277 (2010).
Hotamisligil, G.S., Shargill, N.S. & Spiegelman, B.M. Adipose expression of tumor necrosis factor-α: direct role in obesity-linked insulin resistance. Science 259, 87–91 (1993).
Shoelson, S.E., Lee, J. & Yuan, M. Inflammation and the IKK beta/I kappa B/NF-kappa B axis in obesity- and diet-induced insulin resistance. Int. J. Obes. Relat. Metab. Disord. 27 (suppl. 3), S49–S52 (2003).
Mathis, D. Immunological goings-on in visceral adipose tissue. Cell Metab. 17, 851–859 (2013).
Weisberg, S.P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Xu, H. et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Invest. 112, 1821–1830 (2003).
Lanthier, N. et al. Kupffer cell activation is a causal factor for hepatic insulin resistance. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G107–G116 (2010).
Pillon, N.J., Bilan, P.J., Fink, L.N. & Klip, A. Cross-talk between skeletal muscle and immune cells: muscle-derived mediators and metabolic implications. Am. J. Physiol. Endocrinol. Metab. 304, E453–E465 (2013).
Chawla, A., Nguyen, K.D. & Goh, Y.P. Macrophage-mediated inflammation in metabolic disease. Nat. Rev. Immunol. 11, 738–749 (2011).
Kintscher, U. et al. T-lymphocyte infiltration in visceral adipose tissue: a primary event in adipose tissue inflammation and the development of obesity-mediated insulin resistance. Arterioscler. Thromb. Vasc. Biol. 28, 1304–1310 (2008).
Wu, D. et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 332, 243–247 (2011).
Yang, H. et al. Obesity increases the production of proinflammatory mediators from adipose tissue T cells and compromises TCR repertoire diversity: implications for systemic inflammation and insulin resistance. J. Immunol. 185, 1836–1845 (2010).
Samuelsson, B., Dahlen, S.E., Lindgren, J.A., Rouzer, C.A. & Serhan, C.N. Leukotrienes and lipoxins: structures, biosynthesis, and biological effects. Science 237, 1171–1176 (1987).
Kim, N. & Luster, A.D. Regulation of immune cells by eicosanoid receptors. ScientificWorldJournal 7, 1307–1328 (2007).
Tager, A.M. & Luster, A.D. BLT1 and BLT2: the leukotriene B(4) receptors. Prostaglandins Leukot. Essent. Fatty Acids 69, 123–134 (2003).
Toda, A., Yokomizo, T. & Shimizu, T. Leukotriene B4 receptors. Prostaglandins Other Lipid Mediat. 68–69, 575–585 (2002).
Chou, R.C. et al. Lipid-cytokine-chemokine cascade drives neutrophil recruitment in a murine model of inflammatory arthritis. Immunity 33, 266–278 (2010).
Haribabu, B. et al. Targeted disruption of the leukotriene B(4) receptor in mice reveals its role in inflammation and platelet-activating factor-induced anaphylaxis. J. Exp. Med. 192, 433–438 (2000).
Subbarao, K. et al. Role of leukotriene B4 receptors in the development of atherosclerosis: potential mechanisms. Arterioscler. Thromb. Vasc. Biol. 24, 369–375 (2004).
Spite, M. et al. Deficiency of the leukotriene B4 receptor, BLT-1, protects against systemic insulin resistance in diet-induced obesity. J. Immunol. 187, 1942–1949 (2011).
Yokomizo, T., Kato, K., Hagiya, H., Izumi, T. & Shimizu, T. Hydroxyeicosanoids bind to and activate the low affinity leukotriene B4 receptor, BLT2. J. Biol. Chem. 276, 12454–12459 (2001).
Liston, T.E. et al. Pharmacokinetics and pharmacodynamics of the leukotriene B4 receptor antagonist CP-105,696 in man following single oral administration. Br. J. Clin. Pharmacol. 45, 115–121 (1998).
Aiello, R.J. et al. Leukotriene B4 receptor antagonism reduces monocytic foam cells in mice. Arterioscler. Thromb. Vasc. Biol. 22, 443–449 (2002).
Showell, H.J., Breslow, R., Conklyn, M.J., Hingorani, G.P. & Koch, K. Characterization of the pharmacological profile of the potent LTB4 antagonist CP-105,696 on murine LTB4 receptors in vitro. Br. J. Pharmacol. 117, 1127–1132 (1996).
Hicks, A., Monkarsh, S.P., Hoffman, A.F. & Goodnow, R. Jr. Leukotriene B4 receptor antagonists as therapeutics for inflammatory disease: preclinical and clinical developments. Expert Opin. Investig. Drugs 16, 1909–1920 (2007).
Oh, D.Y., Morinaga, H., Talukdar, S., Bae, E.J. & Olefsky, J.M. Increased macrophage migration into adipose tissue in obese mice. Diabetes 61, 346–354 (2012).
Lumeng, C.N., Bodzin, J.L. & Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117, 175–184 (2007).
Birkenfeld, A.L. & Shulman, G.I. Nonalcoholic fatty liver disease, hepatic insulin resistance and type 2 diabetes. Hepatology 59, 713–723 (2014).
Samuel, V.T. et al. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J. Biol. Chem. 279, 32345–32353 (2004).
Kumashiro, N. et al. Cellular mechanism of insulin resistance in nonalcoholic fatty liver disease. Proc. Natl. Acad. Sci. USA 108, 16381–16385 (2011).
Jornayvaz, F.R. & Shulman, G.I. Diacylglycerol activation of protein kinase Cɛ and hepatic insulin resistance. Cell Metab. 15, 574–584 (2012).
Holland, W.L. et al. Lipid-induced insulin resistance mediated by the proinflammatory receptor TLR4 requires saturated fatty acid-induced ceramide biosynthesis in mice. J. Clin. Invest. 121, 1858–1870 (2011).
Pagadala, M., Kasumov, T., McCullough, A.J., Zein, N.N. & Kirwan, J.P. Role of ceramides in nonalcoholic fatty liver disease. Trends Endocrinol. Metab. 23, 365–371 (2012).
Chavez, J.A. & Summers, S.A. A ceramide-centric view of insulin resistance. Cell Metab. 15, 585–594 (2012).
Islam, S.A. et al. The leukotriene B4 lipid chemoattractant receptor BLT1 defines antigen-primed T cells in humans. Blood 107, 444–453 (2006).
Weller, C.L. et al. Leukotriene B4, an activation product of mast cells, is a chemoattractant for their progenitors. J. Exp. Med. 201, 1961–1971 (2005).
Afonso, P.V. et al. LTB4 is a signal-relay molecule during neutrophil chemotaxis. Dev. Cell 22, 1079–1091 (2012).
Yamaoka, K.A., Claesson, H.E. & Rosen, A. Leukotriene B4 enhances activation, proliferation, and differentiation of human B lymphocytes. J. Immunol. 143, 1996–2000 (1989).
Horrillo, R. et al. 5-lipoxygenase activating protein signals adipose tissue inflammation and lipid dysfunction in experimental obesity. J. Immunol. 184, 3978–3987 (2010).
Li, P. et al. Functional heterogeneity of CD11c-positive adipose tissue macrophages in diet-induced obese mice. J. Biol. Chem. 285, 15333–15345 (2010).
Lee, Y.S. et al. Increased adipocyte O2 consumption triggers HIF-1α, causing inflammation and insulin resistance in obesity. Cell 157, 1339–1352 (2014).
Taniguchi, C.M., Emanuelli, B. & Kahn, C.R. Critical nodes in signalling pathways: insights into insulin action. Nat. Rev. Mol. Cell Biol. 7, 85–96 (2006).
Osborn, O. & Olefsky, J.M. The cellular and signaling networks linking the immune system and metabolism in disease. Nat. Med. 18, 363–374 (2012).
Donath, M.Y., Dalmas, E., Sauter, N.S. & Boni-Schnetzler, M. Inflammation in obesity and diabetes: islet dysfunction and therapeutic opportunity. Cell Metab. 17, 860–872 (2013).
Larsen, C.M. et al. Interleukin-1-receptor antagonist in type 2 diabetes mellitus. N. Engl. J. Med. 356, 1517–1526 (2007).
Stanley, T.L. et al. TNF-α antagonism with etanercept decreases glucose and increases the proportion of high molecular weight adiponectin in obese subjects with features of the metabolic syndrome. J. Clin. Endocrinol. Metab. 96, E146–E150 (2011).
Kiortsis, D.N., Mavridis, A.K., Vasakos, S., Nikas, S.N. & Drosos, A.A. Effects of infliximab treatment on insulin resistance in patients with rheumatoid arthritis and ankylosing spondylitis. Ann. Rheum. Dis. 64, 765–766 (2005).
Ofei, F., Hurel, S., Newkirk, J., Sopwith, M. & Taylor, R. Effects of an engineered human anti-TNF-α antibody (CDP571) on insulin sensitivity and glycemic control in patients with NIDDM. Diabetes 45, 881–885 (1996).
Goldfine, A.B. et al. Targeting inflammation using salsalate in patients with type 2 diabetes (TINSAL): effects on flow-mediated dilation. Diabetes Care 36, 4132–4139 (2013).
Goldfine, A.B. et al. A randomised trial of salsalate for insulin resistance and cardiovascular risk factors in persons with abnormal glucose tolerance. Diabetologia 56, 714–723 (2013).
Goldfine, A.B. et al. Salicylate (salsalate) in patients with type 2 diabetes: a randomized trial. Ann. Intern. Med. 159, 1–12 (2013).
Cavelti-Weder, C. et al. Effects of gevokizumab on glycemia and inflammatory markers in type 2 diabetes. Diabetes Care 35, 1654–1662 (2012).
Sloan-Lancaster, J. et al. Double-blind, randomized study evaluating the glycemic and anti-inflammatory effects of subcutaneous LY2189102, a neutralizing IL-1β antibody, in patients with type 2 diabetes. Diabetes Care 36, 2239–2246 (2013).
Quehenberger, O. et al. Lipidomics reveals a remarkable diversity of lipids in human plasma. J. Lipid Res. 51, 3299–3305 (2010).
He, W. et al. Adipose-specific peroxisome proliferator-activated receptor gamma knockout causes insulin resistance in fat and liver but not in muscle. Proc. Natl. Acad. Sci. USA 100, 15712–15717 (2003).
Lu, M. et al. Inducible nitric oxide synthase deficiency in myeloid cells does not prevent diet-induced insulin resistance. Mol. Endocrinol. 24, 1413–1422 (2010).
Oh, D.Y. et al. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 142, 687–698 (2010).
Li, P. et al. NCoR repression of LXRs restricts macrophage biosynthesis of insulin-sensitizing omega 3 fatty acids. Cell 155, 200–214 (2013).
Lee, Y.S. et al. Inflammation is necessary for long-term but not short-term high-fat diet-induced insulin resistance. Diabetes 60, 2474–2483 (2011).
Bodennec, J., Brichon, G., Koul, O., El Babili, M. & Zwingelstein, G. A two-dimensional thin-layer chromatography procedure for simultaneous separation of ceramide and diacylglycerol species. J. Lipid Res. 38, 1702–1706 (1997).
Bielawska, A., Perry, D.K. & Hannun, Y.A. Determination of ceramides and diglycerides by the diglyceride kinase assay. Anal. Biochem. 298, 141–150 (2001).
Bektas, M., Jolly, P.S., Milstien, S. & Spiegel, S. A specific ceramide kinase assay to measure cellular levels of ceramide. Anal. Biochem. 320, 259–265 (2003).
Acknowledgements
We thank A.D. Luster from Harvard Medical School for providing the Ltb4r1-KO mice. We thank A. Klip at The Hospital for Sick Children in Canada for providing L6 myocytes. This study was funded in part by grants to J.M. Olefsky from the US National Institute of Diabetes and Digestive and Kidney Diseases (DK033651, DK074868, DK063491, DK09062), the Eunice Kennedy Shriver National Institute of Child Health and Human Development/National Institutes of Health through a Cooperative Centers Program in Reproduction and Infertility Research (J.M. Olefsky), and a grant to J.M.Olefsky from Merck, Inc.
Author information
Authors and Affiliations
Contributions
P.L. designed the studies and performed most of the experiments; D.Y.O. performed macrophage signaling, chemotaxis, and FACS analysis; G.B. did glucose uptake and gluconeogenesis assay and western blot. W.S.L., M.L. and S. Talukdar assisted with hyperinsulinemic-euglycemic clamps; A.J., M.M., O.O. and R.M. assisted with collecting tissues and gene expression measurements; H.C. performed the GTT in female ovariectomized mice; J.M. Ofrecio and S. Taguchi assisted with genotyping; P.L. and J.M. Olefsky analyzed and interpreted the data, supervised the project, and co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 7836 kb)
Rights and permissions
About this article
Cite this article
Li, P., Oh, D., Bandyopadhyay, G. et al. LTB4 promotes insulin resistance in obese mice by acting on macrophages, hepatocytes and myocytes. Nat Med 21, 239–247 (2015). https://doi.org/10.1038/nm.3800
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3800
This article is cited by
-
Emerging mechanisms of obesity-associated immune dysfunction
Nature Reviews Endocrinology (2024)
-
The relationship of tongue fat content and efficacy of uvulopalatopharyngoplasty in Chinese patients with obstructive sleep apnea
BMC Surgery (2023)
-
Clinical response to EPA supplementation in patients with major depressive disorder is associated with higher plasma concentrations of pro-resolving lipid mediators
Neuropsychopharmacology (2023)
-
The role of captopril in leukotriene deficient type 1 diabetic mice
Scientific Reports (2023)
-
Chromogranin A-derived peptides pancreastatin and catestatin: emerging therapeutic target for diabetes
Amino Acids (2023)