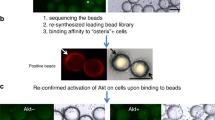
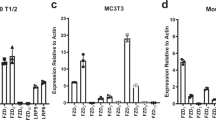
Abstract
Currently, major concerns about the safety and efficacy of RNA interference (RNAi)-based bone anabolic strategies still exist because of the lack of direct osteoblast-specific delivery systems for osteogenic siRNAs. Here we screened the aptamer CH6 by cell-SELEX, specifically targeting both rat and human osteoblasts, and then we developed CH6 aptamer–functionalized lipid nanoparticles (LNPs) encapsulating osteogenic pleckstrin homology domain-containing family O member 1 (Plekho1) siRNA (CH6-LNPs-siRNA). Our results showed that CH6 facilitated in vitro osteoblast-selective uptake of Plekho1 siRNA, mainly via macropinocytosis, and boosted in vivo osteoblast-specific Plekho1 gene silencing, which promoted bone formation, improved bone microarchitecture, increased bone mass and enhanced mechanical properties in both osteopenic and healthy rodents. These results indicate that osteoblast-specific aptamer-functionalized LNPs could act as a new RNAi-based bone anabolic strategy, advancing the targeted delivery selectivity of osteogenic siRNAs from the tissue level to the cellular level.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
López-Fraga, M., Martinez, T. & Jimenez, A. RNA interference technologies and therapeutics: from basic research to products. BioDrugs 23, 305–332 (2009).
Novina, C.D. & Sharp, P.A. The RNAi revolution. Nature 430, 161–164 (2004).
Wang, Y. & Grainger, D.W. RNA therapeutics targeting osteoclast-mediated excessive bone resorption. Adv. Drug Deliv. Rev. 64, 1341–1357 (2012).
Lu, K. et al. Targeting WW domains linker of HECT-type ubiquitin ligase Smurf1 for activation by CKIP-1. Nat. Cell Biol. 10, 994–1002 (2008).
Zhang, G. et al. A delivery system targeting bone formation surfaces to facilitate RNAi-based anabolic therapy. Nat. Med. 18, 307–314 (2012).
Wang, D., Miller, S.C., Kopeckova, P. & Kopecek, J. Bone-targeting macromolecular therapeutics. Adv. Drug Deliv. Rev. 57, 1049–1076 (2005).
Wang, X. et al. miR-214 targets ATF4 to inhibit bone formation. Nat. Med. 19, 93–100 (2013).
Li, S.D. & Huang, L. Pharmacokinetics and biodistribution of nanoparticles. Mol. Pharm. 5, 496–504 (2008).
Zamboni, W.C. Liposomal, nanoparticle, and conjugated formulations of anticancer agents. Clin. Cancer Res. 11, 8230–8234 (2005).
Wang, G., Kucharski, C., Lin, X. & Uludag, H. Bisphosphonate-coated BSA nanoparticles lack bone targeting after systemic administration. J. Drug Target. 18, 611–626 (2010).
Akhtar, S. & Benter, I.F. Nonviral delivery of synthetic siRNAs in vivo. J. Clin. Invest. 117, 3623–3632 (2007).
Ye, M. et al. Generating aptamers by cell-SELEX for applications in molecular medicine. Int. J. Mol. Sci. 13, 3341–3353 (2012).
Sefah, K., Shangguan, D., Xiong, X., O'Donoghue, M.B. & Tan, W. Development of DNA aptamers using cell-SELEX. Nat. Protoc. 5, 1169–1185 (2010).
Tam, Y.Y., Chen, S. & Cullis, P.R. Advances in lipid nanoparticles for siRNA delivery. Pharmaceutics 5, 498–507 (2013).
Auguste, D.T. et al. Triggered release of siRNA from poly(ethylene glycol)-protected, pH-dependent liposomes. J. Control. Release 130, 266–274 (2008).
Mui, B.L. et al. Influence of polyethylene glycol lipid desorption rates on pharmacokinetics and pharmacodynamics of siRNA lipid nanoparticles. Mol. Ther. Nucleic Acids 2, e139 (2013).
Rockey, W.M. et al. Rational truncation of an RNA aptamer to prostate-specific membrane antigen using computational structural modeling. Nucleic Acid Ther. 21, 299–314 (2011).
Keefe, A.D., Pai, S. & Ellington, A. Aptamers as therapeutics. Nat. Rev. Drug Discov. 9, 537–550 (2010).
Gilbert, J.C. et al. First-in-human evaluation of anti von Willebrand factor therapeutic aptamer ARC1779 in healthy volunteers. Circulation 116, 2678–2686 (2007).
Cao, H., Chen, J., Awoniyi, M., Henley, J.R. & McNiven, M.A. Dynamin 2 mediates fluid-phase micropinocytosis in epithelial cells. J. Cell Sci. 120, 4167–4177 (2007).
Vercauteren, D. et al. The use of inhibitors to study endocytic pathways of gene carriers: optimization and pitfalls. Mol. Ther. 18, 561–569 (2010).
Lim, J.P. & Gleeson, P.A. Macropinocytosis: an endocytic pathway for internalising large gulps. Immunol. Cell Biol. 89, 836–843 (2011).
Zernik, J., Twarog, K. & Upholt, W.B. Regulation of alkaline phosphatase and alpha 2(I) procollagen synthesis during early intramembranous bone formation in the rat mandible. Differentiation 44, 207–215 (1990).
Fukushima, N., Hiraoka, K., Shirachi, I., Kojima, M. & Nagata, K. Isolation and characterization of a novel peptide, osteoblast activating peptide (OBAP), associated with osteoblast differentiation and bone formation. Biochem. Biophys. Res. Commun. 400, 157–163 (2010).
Ishikawa, S. et al. Involvement of FcRgamma in signal transduction of osteoclast-associated receptor (OSCAR). Int. Immunol. 16, 1019–1025 (2004).
Herman, S. et al. Induction of osteoclast-associated receptor, a key osteoclast costimulation molecule, in rheumatoid arthritis. Arthritis Rheum. 58, 3041–3050 (2008).
Lotinun, S. et al. Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J. Clin. Invest. 123, 666–681 (2013).
Guo, B. et al. Therapeutic RNA interference targeting CKIP-1 with a cross-species sequence to stimulate bone formation. Bone 59, 76–88 (2014).
Sanguineti, R., Storace, D., Monacelli, F., Federici, A. & Odetti, P. Pentosidine effects on human osteoblasts in vitro. Ann. NY Acad. Sci. 1126, 166–172 (2008).
Kishimoto, Y. et al. Gene expression relevant to osteoclastogenesis in the synovium and bone marrow of mature rats with collagen-induced arthritis. Rheumatology (Oxford) 43, 1496–1503 (2004).
Susa, M., Luong-Nguyen, N.H., Cappellen, D., Zamurovic, N. & Gamse, R. Human primary osteoclasts: in vitro generation and applications as pharmacological and clinical assay. J. Transl. Med. 2, 6 (2004).
Yang, M. et al. Developing aptamer probes for acute myelogenous leukemia detection and surface protein biomarker discovery. J.Hematol. Oncol. 7, 5 (2014).
Wu, H. et al. Preparation of aptamer-functionalized lipid nanoparticles (LNPs) encapsulating siRNAs. Protocol Exchange doi:10.1038/protex.2014.053 (2014).
Hao, Y.J. et al. Changes of microstructure and mineralized tissue in the middle and late phase of osteoporotic fracture healing in rats. Bone 41, 631–638 (2007).
Semple, S.C. et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 28, 172–176 (2010).
Acknowledgements
We thank the academic staff (L. Qin) from the Chinese University of Hong Kong and H.Y.S. Cheung from the Institute for Advancing Translational Medicine in Bone & Joint Diseases, Hong Kong Baptist University for providing critical comments and technical support. This study was supported by the Ministry of Science and Technology of China (2013ZX09301307 to A.L.), the Hong Kong General Research Fund (HKBU479111 to G.Z., HKBU478312 to G.Z., HKBU12102914 to G.Z. and HKBU261113 to A.L.), the Natural Science Foundation Council of China (81272045 to G.Z., 81228013 to J.X. and 21221003 to W.T.), the Research Grants Council & Natural Science Foundation Council of China (N_HKBU435/12 to G.Z.), the Interdisciplinary Research Matching Scheme (IRMS) of Hong Kong Baptist University (RC-IRMS/12-13/02 to A.L. and RC-IRMS/13-14/02 to G.Z.), the Hong Kong Baptist University Strategic Development Fund (SDF) (SDF13-1209-P01 to A.L.), the Hong Kong Research Grants Council (RGC) Early Career Scheme (ECS) (489213 to B.-T.Z.), the Faculty Research Grant of Hong Kong Baptist University (FRG1/13-14/024 and FRG2/12-13/027 to G.Z.), the China Academy of Chinese Medical Sciences (Z0252 and Z0293 to A.L.), the Chinese National High-Tech Research and Development Programme (2012AA022501 to N.S.), the National Key Technologies R&D Programs for New Drugs of China (2012ZX09301003-001-001 and 2014ZX09J14106-04C to L.Z.), the Collaborative Research Programme (CRP)-International Centre for Genetic Engineering and Biotechnology (ICGEB) grant (CRP/CHN13-02 to L.Z.), the Chinese National Natural Science Foundation Project (81261160503 to L.Z.), the National Key Scientific Program of China (2011CB911000 to W.T.), the National Institutes of Health (GM079359 to W.T.) and the Beijing Natural Science Foundation (7131012 to L.Z.). The statistical analysis was performed by a contract service from Bioinformedicine (http://www.bioinformedicine.com/index.php).
Author information
Authors and Affiliations
Contributions
G.Z., A.L. and L.Z. supervised the whole project. C. Liang, B.G. and H.W. performed the major research and wrote the manuscript in equal contribution. D.L., J.L., C.W., W.K.L., Changwei Lu, Y.C. and L.D. provided the technical support. X.H., D.W.T.A., Cheng Lu, H.L., S.L., B.-T.Z., N.S., Z.Y., X.P., H.Z., K.Y., A.Q., P.S., J.X., L.X., Z.L., Z.B., F.H. and W.T. provided their professional expertise.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1–4 (PDF 3497 kb)
Rights and permissions
About this article
Cite this article
Liang, C., Guo, B., Wu, H. et al. Aptamer-functionalized lipid nanoparticles targeting osteoblasts as a novel RNA interference–based bone anabolic strategy. Nat Med 21, 288–294 (2015). https://doi.org/10.1038/nm.3791
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3791
This article is cited by
-
Peptide-modified PAMAM-based bone-targeting RNA delivery system
Future Journal of Pharmaceutical Sciences (2023)
-
Cell unit-inspired natural nano-based biomaterials as versatile building blocks for bone/cartilage regeneration
Journal of Nanobiotechnology (2023)
-
A novel Anti-ROS osteoblast-specific delivery system for ankylosing spondylitis treatment via suppression of both inflammation and pathological new bone formation
Journal of Nanobiotechnology (2023)
-
Targeting strategies for bone diseases: signaling pathways and clinical studies
Signal Transduction and Targeted Therapy (2023)
-
Advantages of Material Biofunctionalization Using Nucleic Acid Aptamers in Tissue Engineering and Regenerative Medicine
Molecular Biotechnology (2023)