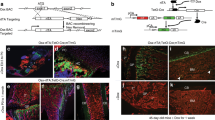
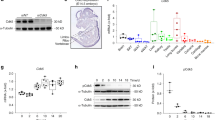
Abstract
The WNT16 locus is a major determinant of cortical bone thickness and nonvertebral fracture risk in humans. The disability, mortality and costs caused by osteoporosis-induced nonvertebral fractures are enormous. We demonstrate here that Wnt16-deficient mice develop spontaneous fractures as a result of low cortical thickness and high cortical porosity. In contrast, trabecular bone volume is not altered in these mice. Mechanistic studies revealed that WNT16 is osteoblast derived and inhibits human and mouse osteoclastogenesis both directly by acting on osteoclast progenitors and indirectly by increasing expression of osteoprotegerin (Opg) in osteoblasts. The signaling pathway activated by WNT16 in osteoclast progenitors is noncanonical, whereas the pathway activated in osteoblasts is both canonical and noncanonical. Conditional Wnt16 inactivation revealed that osteoblast-lineage cells are the principal source of WNT16, and its targeted deletion in osteoblasts increases fracture susceptibility. Thus, osteoblast-derived WNT16 is a previously unreported key regulator of osteoclastogenesis and fracture susceptibility. These findings open new avenues for the specific prevention or treatment of nonvertebral fractures, a substantial unmet medical need.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baron, R. & Hesse, E. Update on bone anabolics in osteoporosis treatment: rationale, current status, and perspectives. J. Clin. Endocrinol. Metab. 97, 311–325 (2012).
Zebaze, R.M. et al. Intracortical remodelling and porosity in the distal radius and post-mortem femurs of women: a cross-sectional study. Lancet 375, 1729–1736 (2010).
Holzer, G., von Skrbensky, G., Holzer, L.A. & Pichl, W. Hip fractures and the contribution of cortical versus trabecular bone to femoral neck strength. J. Bone Miner. Res. 24, 468–474 (2009).
Zheng, H.F. et al. WNT16 influences bone mineral density, cortical bone thickness, bone strength, and osteoporotic fracture risk. PLoS Genet. 8, e1002745 (2012).
Baron, R. & Kneissel, M. WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat. Med. 19, 179–192 (2013).
Cadigan, K.M. & Peifer, M. Wnt signaling from development to disease: insights from model systems. Cold Spring Harb. Perspect. Biol. 1, a002881 (2009).
Jenny, A. Planar cell polarity signaling in the Drosophila eye. Curr. Top. Dev. Biol. 93, 189–227 (2010).
Kohn, A.D. & Moon, R.T. Wnt and calcium signaling: β-catenin–independent pathways. Cell Calcium 38, 439–446 (2005).
Gong, Y. et al. LDL receptor–related protein 5 (LRP5) affects bone accrual and eye development. Cell 107, 513–523 (2001).
Little, R.D. et al. A mutation in the LDL receptor–related protein 5 gene results in the autosomal dominant high–bone-mass trait. Am. J. Hum. Genet. 70, 11–19 (2002).
Boyden, L.M. et al. High bone density due to a mutation in LDL-receptor–related protein 5. N. Engl. J. Med. 346, 1513–1521 (2002).
Brunkow, M.E. et al. Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein. Am. J. Hum. Genet. 68, 577–589 (2001).
Balemans, W. et al. Identification of a 52 kb deletion downstream of the SOST gene in patients with van Buchem disease. J. Med. Genet. 39, 91–97 (2002).
Kramer, I. et al. Osteocyte Wnt/β-catenin signaling is required for normal bone homeostasis. Mol. Cell. Biol. 30, 3071–3085 (2010).
Glass, D.A. II et al. Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev. Cell 8, 751–764 (2005).
Miclea, R.L. et al. Adenomatous polyposis coli–mediated control of β-catenin is essential for both chondrogenic and osteogenic differentiation of skeletal precursors. BMC Dev. Biol. 9, 26 (2009).
Bennett, C.N. et al. Wnt10b increases postnatal bone formation by enhancing osteoblast differentiation. J. Bone Miner. Res. 22, 1924–1932 (2007).
Kato, M. et al. Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J. Cell Biol. 157, 303–314 (2002).
Bonewald, L.F. & Johnson, M.L. Osteocytes, mechanosensing and Wnt signaling. Bone 42, 606–615 (2008).
Holmen, S.L. et al. Essential role of β-catenin in postnatal bone acquisition. J. Biol. Chem. 280, 21162–21168 (2005).
Day, T.F., Guo, X., Garrett-Beal, L. & Yang, Y. Wnt/β-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev. Cell 8, 739–750 (2005).
Hill, T.P., Spater, D., Taketo, M.M., Birchmeier, W. & Hartmann, C. Canonical Wnt/β-catenin signaling prevents osteoblasts from differentiating into chondrocytes. Dev. Cell 8, 727–738 (2005).
Wei, W. et al. Biphasic and dosage-dependent regulation of osteoclastogenesis by β-catenin. Mol. Cell. Biol. 31, 4706–4719 (2011).
Maeda, K. et al. Wnt5a-Ror2 signaling between osteoblast-lineage cells and osteoclast precursors enhances osteoclastogenesis. Nat. Med. 18, 405–412 (2012).
Estrada, K. et al. Genome-wide meta-analysis identifies 56 bone mineral density loci and reveals 14 loci associated with risk of fracture. Nat. Genet. 44, 491–501 (2012).
Medina-Gomez, C. et al. Meta-analysis of genome-wide scans for total body BMD in children and adults reveals allelic heterogeneity and age-specific effects at the WNT16 locus. PLoS Genet. 8, e1002718 (2012).
Koller, D.L. et al. Meta-analysis of genome-wide studies identifies WNT16 and ESR1 SNPs associated with bone mineral density in premenopausal women. J. Bone Miner. Res. 28, 547–558 (2013).
García-Ibarbia, C. et al. Missense polymorphisms of the WNT16 gene are associated with bone mass, hip geometry and fractures. Osteoporos. Int. 24, 2449–2454 (2013).
Hendrickx, G. et al. Variation in the Kozak sequence of WNT16 results in an increased translation and is associated with osteoporosis related parameters. Bone 59, 57–65 (2014).
Brommage, R., Liu, J., Revelli, J., Kirkpatrick, L. & Powwell, D. Gene knockouts of Wnt10b and Wnt16 in mice result in low bone mass. Bone 40, S187 (2007).
Huang, S.M. et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 461, 614–620 (2009).
Teh, M.T. et al. Role for WNT16B in human epidermal keratinocyte proliferation and differentiation. J. Cell Sci. 120, 330–339 (2007).
Liu, Y., Ross, J.F., Bodine, P.V. & Billiard, J. Homodimerization of Ror2 tyrosine kinase receptor induces 14–3-3β phosphorylation and promotes osteoblast differentiation and bone formation. Mol. Endocrinol. 21, 3050–3061 (2007).
Liu, Y., Rubin, B., Bodine, P.V. & Billiard, J. Wnt5a induces homodimerization and activation of Ror2 receptor tyrosine kinase. J. Cell. Biochem. 105, 497–502 (2008).
Rauch, A. et al. Glucocorticoids suppress bone formation by attenuating osteoblast differentiation via the monomeric glucocorticoid receptor. Cell Metab. 11, 517–531 (2010).
Lu, Y. et al. DMP1-targeted Cre expression in odontoblasts and osteocytes. J. Dent. Res. 86, 320–325 (2007).
Kim, J. et al. Lipoproteins are an important bacterial component responsible for bone destruction through the induction of osteoclast differentiation and activation. J. Bone Miner. Res. 28, 2381–2391 (2013).
Wermelin, K., Suska, F., Tengvall, P., Thomsen, P. & Aspenberg, P. Stainless steel screws coated with bisphosphonates gave stronger fixation and more surrounding bone. Histomorphometry in rats. Bone 42, 365–371 (2008).
Zhong, Z. et al. Wntless functions in mature osteoblasts to regulate bone mass. Proc. Natl. Acad. Sci. USA 109, E2197–E2204 (2012).
Bennett, C.N. et al. Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc. Natl. Acad. Sci. USA 102, 3324–3329 (2005).
Stevens, J.R. et al. Wnt10b deficiency results in age-dependent loss of bone mass and progressive reduction of mesenchymal progenitor cells. J. Bone Miner. Res. 25, 2138–2147 (2010).
Green, J.L. et al. Use of a molecular genetic platform technology to produce human Wnt proteins reveals distinct local and distal signaling abilities. PLoS ONE 8, e58395 (2013).
Guo, X. et al. Wnt/β-catenin signaling is sufficient and necessary for synovial joint formation. Genes Dev. 18, 2404–2417 (2004).
Lu, D. et al. Activation of the Wnt signaling pathway in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 101, 3118–3123 (2004).
Sugimura, R. et al. Noncanonical Wnt signaling maintains hematopoietic stem cells in the niche. Cell 150, 351–365 (2012).
Jiang, Z., Von den Hoff, J.W., Torensma, R., Meng, L. & Bian, Z. Wnt16 is involved in intramembranous ossification and suppresses osteoblast differentiation through the Wnt/β-catenin pathway. J. Cell. Physiol. 229, 384–392 (2014).
Santiago, F., Oguma, J., Brown, A.M. & Laurence, J. Noncanonical Wnt signaling promotes osteoclast differentiation and is facilitated by the human immunodeficiency virus protease inhibitor ritonavir. Biochem. Biophys. Res. Commun. 417, 223–230 (2012).
van Amerongen, R. & Nusse, R. Towards an integrated view of Wnt signaling in development. Development 136, 3205–3214 (2009).
Valenzuela, D.M. et al. High-throughput engineering of the mouse genome coupled with high-resolution expression analysis. Nat. Biotechnol. 21, 652–659 (2003).
Windahl, S.H. et al. Estrogen receptor-α in osteocytes is important for trabecular bone formation in male mice. Proc. Natl. Acad. Sci. USA 110, 2294–2299 (2013).
Maes, C. et al. Increased skeletal VEGF enhances β-catenin activity and results in excessively ossified bones. EMBO J. 29, 424–441 (2010).
Windahl, S.H., Vidal, O., Andersson, G., Gustafsson, J.A. & Ohlsson, C. Increased cortical bone mineral content but unchanged trabecular bone mineral density in female ERβ−/− mice. J. Clin. Invest. 104, 895–901 (1999).
Vidal, O. et al. Estrogen receptor specificity in the regulation of skeletal growth and maturation in male mice. Proc. Natl. Acad. Sci. USA 97, 5474–5479 (2000).
Movérare, S. et al. Differential effects on bone of estrogen receptor α and androgen receptor activation in orchidectomized adult male mice. Proc. Natl. Acad. Sci. USA 100, 13573–13578 (2003).
Waarsing, J.H., Day, J.S. & Weinans, H. An improved segmentation method for in vivo microCT imaging. J. Bone Miner. Res. 19, 1640–1650 (2004).
Hildebrand, T. & Ruegsegger, P. Quantification of bone microarchitecture with the structure model index. Comput. Methods Biomech. Biomed. Engin. 1, 15–23 (1997).
Parfitt, A.M. et al. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 2, 595–610 (1987).
Baron, R., Vignery, A., Neff, L., Silverglate,, A. & Santa Maria, A. Processing of undecalcified bone specimens for bone histomorphometry. in Bone Histomorphometry: Techniques and Interpretation (ed. Recker, R.) 13–35 (CRC Press Inc., Boca Raton, 1983).
Eriksen, E., Axelrod, D. & Melsen, F. Bone Histomorphometry (Raven, New York, 1994).
Hayman, A.R., Macary, P., Lehner, P.J. & Cox, T.M. Tartrate-resistant acid phosphatase (Acp 5): identification in diverse human tissues and dendritic cells. J. Histochem. Cytochem. 49, 675–684 (2001).
Granholm, S., Henning, P., Lindholm, C. & Lerner, U.H. Osteoclast progenitor cells present in significant amounts in mouse calvarial osteoblast isolations and osteoclastogenesis increased by BMP-2. Bone 52, 83–92 (2013).
Stanford, C.M., Jacobson, P.A., Eanes, E.D., Lembke, L.A. & Midura, R.J. Rapidly forming apatitic mineral in an osteoblastic cell line (UMR 106–01 BSP). J. Biol. Chem. 270, 9420–9428 (1995).
Granholm, S., Lundberg, P. & Lerner, U.H. Calcitonin inhibits osteoclast formation in mouse haematopoetic cells independently of transcriptional regulation by receptor activator of NF-κB and c-Fms. J. Endocrinol. 195, 415–427 (2007).
Takeshita, S., Kaji, K. & Kudo, A. Identification and characterization of the new osteoclast progenitor with macrophage phenotypes being able to differentiate into mature osteoclasts. J. Bone Miner. Res. 15, 1477–1488 (2000).
Morrell, N.T. et al. Liposomal packaging generates Wnt protein with in vivo biological activity. PLoS ONE 3, e2930 (2008).
Agholme, F., Li, X., Isaksson, H., Ke, H.Z. & Aspenberg, P. Sclerostin antibody treatment enhances metaphyseal bone healing in rats. J. Bone Miner. Res. 25, 2412–2418 (2010).
Acknowledgements
We are grateful to C. Uggla, M. Petersson, A. Hansevi, A. Lie, I. Lundgren, B. Aleksic and the staff at the Turku Center for Disease Modeling (TCDM) for technical assistance. We thank M. Johansson at Bergman Labora for help with photographing the embryos. We thank R. Brommage (Lexicon Pharmaceuticals Inc.) for providing the Wnt16−/−, exon 1−3 mice. This study was supported by the Swedish Research Council, the Swedish Foundation for Strategic Research, COMBINE, the Avtal om Läkarutbildning och Forskning/Läkarutbildningsavtalet (ALF/LUA) research grant in Gothenburg, Linköping University, Swedish National Graduate School in Odontological Sciences, the Lundberg Foundation, the Torsten and Ragnar Söderberg's Foundation, the Swedish Rheumatism Association, the Royal 80 Year Fund of King Gustav V, the Novo Nordisk Foundation and the German Research Foundation (SPP 1468 Tu220/6, Immunobone). The work at TCDM was supported by funding provided by University of Turku and Biocenter Finland. This work was supported in part by a grant to R.B. from the US National Institutes of Health (R01AR064724). The HSDM micro-CT core facility performed the micro-CT analyses of the Wnt16−/−, exon 1−3 mice.
Author information
Authors and Affiliations
Contributions
S.M.-S., P.H. and X.L. contributed equally to this work. S.M.-S. was responsible for the breeding, phenotyping and treatment of the Wnt16−/−, exon 1−4 and conditional Wnt16flox/flox mouse strains. P.H. conducted the in vitro culture experiments and analyses of primary osteoblasts, cocultures and osteoclasts. X.L. was responsible for the breeding and phenotyping of the Wnt16−/−, exon 1−3 mouse colony and conducted in vitro and ex vivo experiments. H.S. assisted with animal experiments and performed initial bone histomorphometry analyses. K.N. performed bone histomorphometry analyses. A.E.B. assisted with animal experiments and performed micro-CT analysis. K.S., S.H.W., H.F., H.I. and C.E. assisted with animal experiments. B.K. performed the FACS analysis. A. Koskela and J. Tuukkanen performed the three-point bending experiments. F.-P.Z. and M.P. generated the conditional Wnt16flox/flox mice. E.E.E., F.Z. and L.S. performed the immunohistochemistry. A.H. performed western blots. M.B. produced the WNT16 liposomes. A. Kassem and C.L. performed the inflammation-induced calvarial bone loss model. O.S. and P.A. performed the rat metaphyseal WNT16 injections. J.Q.F. provided the Dpm1-cre mice. J. Tuckermann provided the Runx2-cre mice. S.M.-S., P.H., R.B., U.H.L., F.G. and C.O. designed and supervised the project and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Tables 1–10 (PDF 1013 kb)
Rights and permissions
About this article
Cite this article
Movérare-Skrtic, S., Henning, P., Liu, X. et al. Osteoblast-derived WNT16 represses osteoclastogenesis and prevents cortical bone fragility fractures. Nat Med 20, 1279–1288 (2014). https://doi.org/10.1038/nm.3654
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3654
This article is cited by
-
Oncostatin M: Dual Regulator of the Skeletal and Hematopoietic Systems
Current Osteoporosis Reports (2024)
-
The role of wnt signaling in diabetes-induced osteoporosis
Diabetology & Metabolic Syndrome (2023)
-
Rspo2 exacerbates rheumatoid arthritis by targeting aggressive phenotype of fibroblast-like synoviocytes and disrupting chondrocyte homeostasis via Wnt/β-catenin pathway
Arthritis Research & Therapy (2023)
-
CircFam190a: a critical positive regulator of osteoclast differentiation via enhancement of the AKT1/HSP90β complex
Experimental & Molecular Medicine (2023)
-
Unraveling the prevalence of various signalling pathways in non-small-cell lung cancer: a review
Molecular and Cellular Biochemistry (2023)