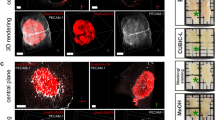
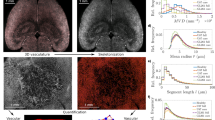
Abstract
Measurement of vessel caliber by magnetic resonance imaging (MRI) is a valuable technique for in vivo monitoring of hemodynamic status and vascular development, especially in the brain. Here, we introduce a new paradigm in MRI termed vessel architectural imaging (VAI) that exploits an overlooked temporal shift in the magnetic resonance signal, forming the basis for vessel caliber estimation, and show how this phenomenon can reveal new information on vessel type and function not assessed by any other noninvasive imaging technique. We also show how this biomarker can provide new biological insights into the treatment of patients with cancer. As an example, we demonstrate using VAI that anti-angiogenic therapy can improve microcirculation and oxygen saturation and reduce vessel calibers in patients with recurrent glioblastomas and, more crucially, that patients with these responses have prolonged survival. Thus, VAI has the potential to identify patients who would benefit from therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
20 September 2013
In the version of this article initially published, the labeling of Figure 2 was incorrect. The three top labels for arterioles should have read as follows (from bottom to top): “Rart = 7.5 μm,” “Rart = 10 μm” and “Rart = 20 μm,” respectively. The error has been corrected in the HTML and PDF versions of the article.
References
Carmeliet, P. & Jain, R.K. Molecular mechanisms and clinical applications of angiogenesis. Nature 473, 298–307 (2011).
Goel, S. et al. Normalization of the vasculature for treatment of cancer and other diseases. Physiol. Rev. 91, 1071–1121 (2011).
Willett, C.G. et al. Direct evidence that the VEGF-specific antibody bevacizumab has antivascular effects in human rectal cancer. Nat. Med. 10, 145–147 (2004).
Batchelor, T.T. et al. AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell 11, 83–95 (2007).
Sorensen, A.G. et al. Increased survival of glioblastoma patients who respond to antiangiogenic therapy with elevated blood perfusion. Cancer Res. 72, 402–407 (2012).
Dennie, J. et al. NMR imaging of changes in vascular morphology due to tumor angiogenesis. Magn. Reson. Med. 40, 793–799 (1998).
Boxerman, J.L., Hamberg, L.M., Rosen, B.R. & Weisskoff, R.M. MR contrast due to intravascular magnetic susceptibility perturbations. Magn. Reson. Med. 34, 555–566 (1995).
Weisskoff, R.M., Zuo, C.S., Boxerman, J.L. & Rosen, B.R. Microscopic susceptibility variation and transverse relaxation: theory and experiment. Magn. Reson. Med. 31, 601–610 (1994).
Kiselev, V.G., Strecker, R., Ziyeh, S., Speck, O. & Hennig, J. Vessel size imaging in humans. Magn. Reson. Med. 53, 553–563 (2005).
Kennan, R.P., Zhong, J. & Gore, J.C. Intravascular susceptibility contrast mechanisms in tissues. Magn. Reson. Med. 31, 9–21 (1994).
Fisel, C.R. et al. MR contrast due to microscopically heterogeneous magnetic susceptibility: numerical simulations and applications to cerebral physiology. Magn. Reson. Med. 17, 336–347 (1991).
Beaumont, M. et al. Characterization of tumor angiogenesis in rat brain using iron-based vessel size index MRI in combination with gadolinium-based dynamic contrast-enhanced MRI. J. Cereb. Blood Flow Metab. 29, 1714–1726 (2009).
Schmainda, K.M. et al. Characterization of a first-pass gradient-echo spin-echo method to predict brain tumor grade and angiogenesis. AJNR Am. J. Neuroradiol. 25, 1524–1532 (2004); erratum 26, 686 (2005).
Weisskoff, R.M., Chesler, D., Boxerman, J.L. & Rosen, B.R. Pitfalls in MR measurement of tissue blood flow with intravascular tracers: which mean transit time? Magn. Reson. Med. 29, 553–558 (1993).
Tsai, A.G., Johnson, P.C. & Intaglietta, M. Oxygen gradients in the microcirculation. Physiol. Rev. 83, 933–963 (2003).
Sharan, M., Jones, M.D. Jr., Koehler, R.C., Traystman, R.J. & Popel, A.S. A compartmental model for oxygen transport in brain microcirculation. Ann. Biomed. Eng. 17, 13–38 (1989).
Batchelor, T.T. et al. Phase II study of cediranib, an oral pan–vascular endothelial growth factor receptor tyrosine kinase inhibitor, in patients with recurrent glioblastoma. J. Clin. Oncol. 28, 2817–2823 (2010).
Less, J.R., Skalak, T.C., Sevick, E.M. & Jain, R.K. Microvascular architecture in a mammary carcinoma: branching patterns and vessel dimensions. Cancer Res. 51, 265–273 (1991).
Helmlinger, G., Yuan, F., Dellian, M. & Jain, R.K. Interstitial pH and pO2 gradients in solid tumors in vivo: high-resolution measurements reveal a lack of correlation. Nat. Med. 3, 177–182 (1997).
Wilson, W.R. & Hay, M.P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 11, 393–410 (2011).
Dvorak, H.F., Brown, L.F., Detmar, M. & Dvorak, A.M. Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am. J. Pathol. 146, 1029–1039 (1995).
Stylianopoulos, T. et al. Causes, consequences, and remedies for growth-induced solid stress in murine and human tumors. Proc. Natl. Acad. Sci. USA 109, 15101–15108 (2012).
Pries, A.R., Hopfner, M., le Noble, F., Dewhirst, M.W. & Secomb, T.W. The shunt problem: control of functional shunting in normal and tumour vasculature. Nat. Rev. Cancer 10, 587–593 (2010).
Bulnes, S., Bilbao, J. & Lafuente, J.V. Microvascular adaptive changes in experimental endogenous brain gliomas. Histol. Histopathol. 24, 693–706 (2009).
Zwick, S. et al. Assessment of vascular remodeling under antiangiogenic therapy using DCE-MRI and vessel size imaging. J. Magn. Reson. Imaging 29, 1125–1133 (2009).
Kamoun, W.S. et al. Edema control by cediranib, a vascular endothelial growth factor receptor–targeted kinase inhibitor, prolongs survival despite persistent brain tumor growth in mice. J. Clin. Oncol. 27, 2542–2552 (2009).
Huang, Y. et al. Vascular normalizing doses of antiangiogenic treatment reprogram the immunosuppressive tumor microenvironment and enhance immunotherapy. Proc. Natl. Acad. Sci. USA 109, 17561–17566 (2012).
Bjørnerud, A., Briley-Saebo, K., Johansson, L.O. & Kellar, K.E. Effect of NC100150 injection on the 1H NMR linewidth of human whole blood ex vivo: dependency on blood oxygen tension. Magn. Reson. Med. 44, 803–807 (2000).
Thulborn, K.R., Waterton, J.C., Matthews, P.M. & Radda, G.K. Oxygenation dependence of the transverse relaxation time of water protons in whole blood at high field. Biochim. Biophys. Acta 714, 265–270 (1982).
Larsen, O.A. & Lassen, N.A. Cerebral hematocrit in normal man. J. Appl. Physiol. 19, 571–574 (1964).
Yuan, F. et al. Vascular permeability and microcirculation of gliomas and mammary carcinomas transplanted in rat and mouse cranial windows. Cancer Res. 54, 4564–4568 (1994).
Duvernoy, H., Delon, S. & Vannson, J.L. The vascularization of the human cerebellar cortex. Brain Res. Bull. 11, 419–480 (1983).
Barboriak, D.P., MacFall, J.R., Viglianti, B.L. & Dewhirst Dvm, M.W. Comparison of three physiologically-based pharmacokinetic models for the prediction of contrast agent distribution measured by dynamic MR imaging. J. Magn. Reson. Imaging 27, 1388–1398 (2008).
Jensen, J.H. & Chandra, R. MR imaging of microvasculature. Magn. Reson. Med. 44, 224–230 (2000).
Macdonald, D.R., Cascino, T.L., Schold, S.C. Jr. & Cairncross, J.G. Response criteria for phase II studies of supratentorial malignant glioma. J. Clin. Oncol. 8, 1277–1280 (1990).
Bjørnerud, A. & Emblem, K.E. A fully automated method for quantitative cerebral hemodynamic analysis using DSC-MRI. J. Cereb. Blood Flow Metab. 30, 1066–1078 (2010).
Acknowledgements
The authors thank T. Stylianopoulos (Department of Mechanical and Manufacturing Engineering, University of Cyprus) and J.W. Baish (Department of Mechanical and Biomedical Engineering, Bucknell University) for critical reading of this manuscript. This work was funded by the US Public Health Service (grants R21CA117079, S10RR023401, S10RR019307, S10RR019254, S10RR023043, S10RR021110, R01CA137254, R01CA129371, 5R01NS060918, K24CA125440, P01CA80124 and K25AG029415), by the US National Cancer Institute (http://www.clinicaltrials.gov/, NCT00035656); SAIC-Frederick Inc. grant 26XS263; Norwegian Research Council grant 191088/F20; South-Eastern Norway Regional Health Authority Grant 2013069; the Danish Research Foundation (Center of Functionally Integrative Neuroscience); the Ministry of Science, Innovation and Technology–Denmark (Investment Capital for University Research); the Sigrid Juselius Foundation; the Instrumentarium Research Foundation; the Academy of Finland; the Paulo Foundation; and the Finnish Medical Foundation. This work was conducted with support from Harvard Catalyst–Harvard Clinical and Translational Science Center (US National Institutes of Health Award nos. UL1 RR 025758 and M01-RR-01066 and financial contributions from Harvard University and its affiliated academic healthcare centers). The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University and its affiliated academic health care centers, the US National Center for Research Resources or the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
K.E.E. wrote the manuscript. K.E.E., K.M., A.B., P.Y.W., P.I., T.T.B., B.R.R., R.K.J. and A.G.S. designed the study. D.J., B.R.R. and A.G.S. acquired all of the MRI data. P.Y.W. and T.T.B. acquired all of the clinical data. K.E.E. and A.B. performed the MatLab simulations. K.E.E., K.M., A.B., C.T.F., D.J., R.J.H.B., B.R.R., R.K.J. and A.G.S. analyzed and interpreted the simulations and human data. K.E.E. performed the statistical analysis. K.E.E., K.M., A.B., C.T.F., D.J., R.J.H.B., P.Y.W., P.I., T.T.B., B.R.R., R.K.J. and A.G.S. revised the manuscript critically. K.E.E., K.M., A.B., C.T.F., D.J., R.J.H.B., P.Y.W., P.I., T.T.B., B.R.R., R.K.J. and A.G.S. approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
K.E.E. and A.B. have licensed patents with NordicNeuroLab AS. A.B. is a board member at NordicNeuroLab AS. P.Y.W. received research support from Merck, Sanofi-Aventis, Genentech, Novartis, Medimmune, AstraZeneca, Amgen, Vascular Biogenics and Genzyme. T.T.B. is on the consultant-advisory boards of Merck, Roche, Kirin Pharmaceuticals, Champions Biotechnology and Advance Medical and received grants from Pfizer, AstraZeneca and Millennium. B.R. is on the consultant-advisory board of Siemens Medical. R.K.J. received grant support from Dyax, MedImmune and Roche; is on the consultant-advisory board of Dyax, Noxxon Pharma and SynDevRx; and is on the board of directors of Xtuit. A.G.S. is the chief executive officer of Siemens HealthCare USA; received grant support from Sanofi-Aventis, Exelixis Inc., Schering-Plough and Takeda Pharmaceutical Company Ltd.; and is on the consultant-advisory board of Sanofi-Aventis, Bayer AG, Mitsubishi and Biogen Idec Inc.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 1824 kb)
Rights and permissions
About this article
Cite this article
Emblem, K., Mouridsen, K., Bjornerud, A. et al. Vessel architectural imaging identifies cancer patient responders to anti-angiogenic therapy. Nat Med 19, 1178–1183 (2013). https://doi.org/10.1038/nm.3289
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3289
This article is cited by
-
Hematopoietic stem-cell gene therapy is associated with restored white matter microvascular function in cerebral adrenoleukodystrophy
Nature Communications (2023)
-
Antiangiogenic Therapy for Malignant Brain Tumors: Does It Still Matter?
Current Oncology Reports (2023)
-
Simultaneous quantification of perfusion, permeability, and leakage effects in brain gliomas using dynamic spin-and-gradient-echo echoplanar imaging MRI
European Radiology (2023)
-
Differentiation of Cerebral Neoplasms with Vessel Size Imaging (VSI)
Clinical Neuroradiology (2022)
-
Vessel size and perfusion-derived vascular habitat refines prediction of treatment failure to bevacizumab in recurrent glioblastomas: validation in a prospective cohort
European Radiology (2022)