Abstract
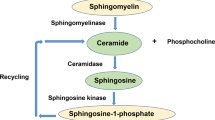
Major depression is a highly prevalent severe mood disorder that is treated with antidepressants. The molecular targets of antidepressants require definition. We investigated the role of the acid sphingomyelinase (Asm)-ceramide system as a target for antidepressants. Therapeutic concentrations of the antidepressants amitriptyline and fluoxetine reduced Asm activity and ceramide concentrations in the hippocampus, increased neuronal proliferation, maturation and survival and improved behavior in mouse models of stress-induced depression. Genetic Asm deficiency abrogated these effects. Mice overexpressing Asm, heterozygous for acid ceramidase, treated with blockers of ceramide metabolism or directly injected with C16 ceramide in the hippocampus had higher ceramide concentrations and lower rates of neuronal proliferation, maturation and survival compared with controls and showed depression-like behavior even in the absence of stress. The decrease of ceramide abundance achieved by antidepressant-mediated inhibition of Asm normalized these effects. Lowering ceramide abundance may thus be a central goal for the future development of antidepressants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Belmaker, R.H. & Agam, G. Major depressive disorder. N. Engl. J. Med. 358, 55–68 (2008).
Howren, M.B., Lamkin, D.M. & Suls, J. Associations of depression with C-reactive protein, IL-1, and IL-6: a meta-analysis. Psychosom. Med. 71, 171–186 (2009).
Dowlati, Y. et al. A meta-analysis of cytokines in major depression. Biol. Psychiatry 67, 446–457 (2010).
Krishnan, V. & Nestler, E.J. The molecular neurobiology of depression. Nature 455, 894–902 (2008).
Brink, C.B., Harvey, B.H. & Brand, L. Tianeptine: a novel atypical antidepressant that may provide new insights into the biomolecular basis of depression. Recent Pat. CNS Drug Discov. 1, 29–41 (2006).
Santarelli, L. et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 301, 805–809 (2003).
Koo, J.W. & Duman, R.S. IL-1β is an essential mediator of the antineurogenic and anhedonic effects of stress. Proc. Natl. Acad. Sci. USA 105, 751–756 (2008).
David, D.J. et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron 62, 479–493 (2009).
Warner-Schmidt, J.L. & Duman, R.S. Hippocampal neurogenesis: opposing effects of stress and antidepressant treatment. Hippocampus 16, 239–249 (2006).
Gulbins, E. & Kolesnick, R. Raft ceramide in molecular medicine. Oncogene 22, 7070–7077 (2003).
Grassmé, H. et al. CD95 signaling via ceramide-rich membrane rafts. J. Biol. Chem. 276, 20589–20596 (2001).
Perrotta, C. et al. Syntaxin 4 is required for acid sphingomyelinase activity and apoptotic function. J. Biol. Chem. 285, 40240–40251 (2010).
Grassmé, H. et al. Host defense against Pseudomonas aeruginosa requires ceramide-rich membrane rafts. Nat. Med. 9, 322–330 (2003).
Baumann, P. et al. The AGNP-TDM Expert Group Consensus Guidelines: focus on therapeutic monitoring of antidepressants. Dialogues Clin. Neurosci. 7, 231–247 (2005).
Kölzer, M., Werth, N. & Sandhoff, K. Interactions of acid sphingomyelinase and lipid bilayers in the presence of the tricyclic antidepressant desipramine. FEBS Lett. 559, 96–98 (2004).
Kornhuber, J. et al. Identification of new functional inhibitors of acid sphingomyelinase using a structure-property-activity relation model. J. Med. Chem. 51, 219–237 (2008).
Ranganathan, R., Sawin, E.R., Trent, C. & Horvitz, H.R. Mutations in the Caenorhabditis elegans serotonin reuptake transporter MOD-5 reveal serotonin-dependent and -independent activities of fluoxetine. J. Neurosci. 21, 5871–5884 (2001).
Dempsey, C.M., Mackenzie, S.M., Gargus, A., Blanco, G. & Sze, J.Y. Serotonin (5HT), fluoxetine, imipramine and dopamine target distinct 5HT receptor signaling to modulate Caenorhabditis elegans egg-laying behavior. Genetics 169, 1425–1436 (2005).
De Stefanis, D. et al. Increase in ceramide level alters the lysosomal targeting of cathepsin D prior to onset of apoptosis in HT-29 colon cancer cells. Biol. Chem. 383, 989–999 (2002).
Hisaki, H. et al. In vivo influence of ceramide accumulation induced by treatment with a glucosylceramide synthase inhibitor on ischemic neuronal cell death. Brain Res. 1018, 73–77 (2004).
Peltier, J., O'Neill, A. & Schaffer, D.V. PI3K and CREB regulate adult neural hippocampal progenitor proliferation and differentiation. Dev. Neurobiol. 67, 1348–1361 (2007).
Zhang, Y., Li, X., Carpinteiro, A. & Gulbins, E. Acid sphingomyelinase amplifies redox signaling in Pseudomonas aeruginosa–induced macrophage apoptosis. J. Immunol. 181, 4247–4254 (2008).
Mathias, S. et al. Activation of the sphingomyelin signaling pathway in intact EL4 cells and in a cell-free system by IL-1 β. Science 259, 519–522 (1993).
Wiegmann, K., Schütze, S., Machleidt, T., Witte, D. & Krönke, M. Functional dichotomy of neutral and acidic sphingomyelinases in tumor necrosis factor signaling. Cell 78, 1005–1015 (1994).
Kim, M.Y., Linardic, C., Obeid, L. & Hannun, Y. Identification of sphingomyelin turnover as an effector mechanism for the action of tumor necrosis factor α and γ-interferon. Specific role in cell differentiation. J. Biol. Chem. 266, 484–489 (1991).
Brenner, B. et al. Fas- or ceramide-induced apoptosis is mediated by a Rac1-regulated activation of Jun N-terminal kinase/p38 kinases and GADD153. J. Biol. Chem. 272, 22173–22181 (1997).
Lepple-Wienhues, A. et al. Stimulation of CD95 (Fas) blocks T lymphocyte calcium channels through sphingomyelinase and sphingolipids. Proc. Natl. Acad. Sci. USA 96, 13795–13800 (1999).
Müller, N. COX-2 inhibitors as antidepressants and antipsychotics: clinical evidence. Curr. Opin. Investig. Drugs 11, 31–42 (2010).
Walker, J.R. et al. Psychiatric disorders in patients with immune-mediated inflammatory diseases: prevalence, association with disease activity, and overall patient well-being. J. Rheumatol. Suppl. 88, 31–35 (2011).
Rudisch, B. & Nemeroff, C.B. Epidemiology of comorbid coronary artery disease and depression. Biol. Psychiatry 54, 227–240 (2003).
Kojima, M. et al. Depression, inflammation, and pain in patients with rheumatoid arthritis. Arthritis Rheum. 61, 1018–1024 (2009).
Tabas, I. Sphingolipids and atherosclerosis: a mechanistic connection? A therapeutic opportunity? Circulation 110, 3400–3401 (2004).
Kornhuber, J. et al. High activity of acid sphingomyelinase in major depression. J. Neural Transm. 112, 1583–1590 (2005).
Horinouchi, K. et al. Acid sphingomyelinase deficient mice: a model of types A and B Niemann-Pick disease. Nat. Genet. 10, 288–293 (1995).
Lozano, J. et al. Niemann-Pick disease versus acid sphingomyelinase deficiency. Cell Death Differ. 8, 100–103 (2001).
Lakso, M. et al. Efficient in vivo manipulation of mouse genomic sequences at the zygote stage. Proc. Natl. Acad. Sci. USA 93, 5860–5865 (1996).
Amato, D., Müller, C.P. & Badiani, A. Increased drinking after intra-striatal injection of the dopamine D2/D3 receptor agonist quinpirole in the rat. Psychopharmacology (Berl.) 223, 457–463 (2012).
Franklin, K.B.J. & Paxinos, G. The Mouse Brain in Stereotaxic Coordinates 3rd edn., Figure 48 (Academic Press, San Diego, 2007).
Acknowledgements
Asm-deficient mice and asm-1–deficient worms were provided by R. Kolesnick, Memorial Sloan Kettering Cancer Hospital, and E2A-Cre mice by R. Waldschütz, University Hospital Essen. The G4 antibody against Asm was provided by K. Sandhoff, University of Bonn. We thank S. Harde, B. Wilker, C. Sehl, S. Keitsch, M. Schäfer, S. Müller and E. Naschberger for excellent technical help and F. Lang for valuable discussion. Parts of the work were supported by funding from Deutsche Forschungsgemeinschaft grants GU 335/23-1, KO 947/11-1 and GRK 1302.
Author information
Authors and Affiliations
Contributions
E.G. and J.K. initiated the studies, designed experiments and supervised research. E.G., J.K. and M.W. wrote the manuscript. E.G. also performed most mouse studies. E.G., K.A.B. and G.T. performed the histological studies and developed the polyclonal Asm-specific antibody. M.P. and C.B. performed the C. elegans studies. A.L. and B.K. measured ceramide concentrations by mass spectrometry. M.W. designed some experiments and participated in BrdU stainings. H.G. performed the confocal microscopy studies. J.K., P.T. and S.S. performed experiments on the concentration-dependent inhibition of ASM by antidepressant drugs. M.R. and M.P. performed experiments on 5-HT uptake in cultured hippocampal neurons. M.R. and J.K. performed experiments on 5-HT uptake in mouse brain synaptosomes. C.H.T. and T.W.G. designed and performed synapse staining and confocal analyses. T.F.A., U.E.L. and E.G. performed behavioral experiments. C.P.M., D.A., M.R. and J.K. designed and performed hippocampal injection and microdialysis studies. C.A., J.v.B., M.R. and J.K. designed and performed electrophysiological studies in hippocampal slices. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13, Supplementary Notes 1–3 and Supplementary Methods (PDF 21987 kb)
Rights and permissions
About this article
Cite this article
Gulbins, E., Palmada, M., Reichel, M. et al. Acid sphingomyelinase–ceramide system mediates effects of antidepressant drugs. Nat Med 19, 934–938 (2013). https://doi.org/10.1038/nm.3214
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3214
This article is cited by
-
The Acid Sphingomyelinase Inhibitor Amitriptyline Ameliorates TNF-α-Induced Endothelial Dysfunction
Cardiovascular Drugs and Therapy (2024)
-
Repetitive and compulsive behavior after Early-Life-Pain associated with reduced long-chain sphingolipid species
Cell & Bioscience (2023)
-
Association of fluvoxamine with mortality and symptom resolution among inpatients with COVID-19 in Uganda: a prospective interventional open-label cohort study
Molecular Psychiatry (2023)
-
The pharmacological bases for repurposing statins in depression: a review of mechanistic studies
Translational Psychiatry (2023)
-
Overview of the potential use of fluvoxamine for COVID-19 and long COVID
Discover Mental Health (2023)