Abstract
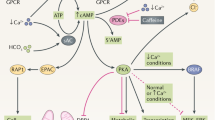
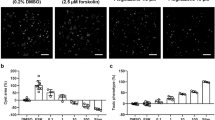
Autosomal dominant polycystic kidney disease (ADPKD) is a common genetic disorder characterized by bilateral renal cyst formation1. Recent identification of signaling cascades deregulated in ADPKD has led to the initiation of several clinical trials, but an approved therapy is still lacking2,3. Using a metabolomic approach, we identify a pathogenic pathway in this disease that can be safely targeted for therapy. We show that mutation of PKD1 results in enhanced glycolysis in cells in a mouse model of PKD and in kidneys from humans with ADPKD. Glucose deprivation resulted in lower proliferation and higher apoptotic rates in PKD1-mutant cells than in nondeprived cells. Notably, two distinct PKD mouse models treated with 2-deoxyglucose (2DG), to inhibit glycolysis, had lower kidney weight, volume, cystic index and proliferation rates as compared to nontreated mice. These metabolic alterations depend on the extracellular signal-related kinase (ERK) pathway acting in a dual manner by inhibiting the liver kinase B1 (LKB1)–AMP-activated protein kinase (AMPK) axis on the one hand while activating the mTOR complex 1 (mTORC1)-glycolytic cascade on the other. Enhanced metabolic rates further inhibit AMPK. Forced activation of AMPK acts in a negative feedback loop, restoring normal ERK activity. Taken together, these data indicate that defective glucose metabolism is intimately involved in the pathobiology of ADPKD. Our findings provide a strong rationale for a new therapeutic strategy using existing drugs, either individually or in combination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Torres, V.E., Harris, P.C. & Pirson, Y. Autosomal dominant polycystic kidney disease. Lancet 369, 1287–1301 (2007).
Harris, P.C. & Torres, V.E. Polycystic kidney disease. Annu. Rev. Med. 60, 321–337 (2009).
Takiar, V. & Caplan, M.J. Polycystic kidney disease: pathogenesis and potential therapies. Biochim. Biophys. Acta 1812, 1337–1343 (2011).
Grantham, J.J., Geiser, J.L. & Evan, A.P. Cyst formation and growth in autosomal dominant polycystic kidney disease. Kidney Int. 31, 1145–1152 (1987).
Torres, V.E. et al. Prospects for mTOR inhibitor use in patients with polycystic kidney disease and hamartomatous diseases. Clin. J. Am. Soc. Nephrol. 5, 1312–1329 (2010).
Distefano, G. et al. Polycystin-1 regulates extracellular signal-regulated kinase-dependent phosphorylation of tuberin to control cell size through mTOR and its downstream effectors S6K and 4EBP1. Mol. Cell Biol. 29, 2359–2371 (2009).
Forseth, R.R. & Schroeder, F.C. NMR-spectroscopic analysis of mixtures: from structure to function. Curr. Opin. Chem. Biol. 15, 38–47 (2011).
Garcia-Manteiga, J.M. et al. Metabolomics of B to plasma cell differentiation. J. Proteome Res. 10, 4165–4176 (2011).
Vander Heiden, M.G., Cantley, L.C. & Thompson, C.B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Düvel, K. et al. Activation of a metabolic gene regulatory network downstream of mTOR complex 1. Mol. Cell 39, 171–183 (2010).
Yecies, J.L. & Manning, B.D. Transcriptional control of cellular metabolism by mTOR signaling. Cancer Res. 71, 2815–2820 (2011).
Chiaradonna, F. et al. From cancer metabolism to new biomarkers and drug targets. Biotechnol. Adv. 30, 30–51 (2012).
Woo, D. Apoptosis and loss of renal tissue in polycystic kidney diseases. N. Engl. J. Med. 333, 18–25 (1995).
Merrick, D. et al. The γ-secretase cleavage product of polycystin-1 regulates TCF and CHOP-mediated transcriptional activation through a p300-dependent mechanism. Dev. Cell 22, 197–210 (2012).
Choo, A.Y. et al. Glucose addiction of TSC null cells is caused by failed mTORC1-dependent balancing of metabolic demand with supply. Mol. Cell 38, 487–499 (2010).
Zheng, B. et al. Oncogenic B-RAF negatively regulates the tumor suppressor LKB1 to promote melanoma cell proliferation. Mol. Cell 33, 237–247 (2009).
Shibazaki, S. et al. Cyst formation and activation of the extracellular regulated kinase pathway after kidney specific inactivation of Pkd1. Hum. Mol. Genet. 17, 1505–1516 (2008).
Song, X. et al. Systems biology of autosomal dominant polycystic kidney disease (ADPKD): computational identification of gene expression pathways and integrated regulatory networks. Hum. Mol. Genet. 18, 2328–2343 (2009).
Foxall, P.J. et al. High resolution proton magnetic resonance spectroscopy of cyst fluids from patients with polycystic kidney disease. Biochim. Biophys. Acta 1138, 305–314 (1992).
Yu, S. et al. Essential role of cleavage of polycystin-1 at G protein-coupled receptor proteolytic site for kidney tubular structure. Proc. Natl. Acad. Sci. USA 104, 18688–18693 (2007).
Csibi, A. & Blenis, J. Appetite for destruction: the inhibition of glycolysis as a therapy for tuberous sclerosis complex-related tumors. BMC Biol. 9, 69 (2011).
Shillingford, J.M. et al. The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease. Proc. Natl. Acad. Sci. USA 103, 5466–5471 (2006).
Shillingford, J.M., Piontek, K.B., Germino, G.G. & Weimbs, T. Rapamycin ameliorates PKD resulting from conditional inactivation of Pkd1. J. Am. Soc. Nephrol. 21, 489–497 (2010).
Serra, A.L. et al. Clinical proof-of-concept trial to assess the therapeutic effect of sirolimus in patients with autosomal dominant polycystic kidney disease: SUISSE ADPKD study. BMC Nephrol. 8, 13 (2007).
Walz, G. et al. Everolimus in patients with autosomal dominant polycystic kidney disease. N. Engl. J. Med. 363, 830–840 (2010).
Serra, A.L. et al. Sirolimus and kidney growth in autosomal dominant polycystic kidney disease. N. Engl. J. Med. 363, 820–829 (2010).
McCarty, M.F., Barroso-Aranda, J. & Contreras, F. Activation of AMP-activated kinase as a strategy for managing autosomal dominant polycystic kidney disease. Med. Hypotheses 73, 1008–1010 (2009).
Takiar, V. et al. Activating AMP-activated protein kinase (AMPK) slows renal cystogenesis. Proc. Natl. Acad. Sci. USA 108, 2462–2467 (2011).
Cheong, J.H. et al. Dual inhibition of tumor energy pathway by 2-deoxyglucose and metformin is effective against a broad spectrum of preclinical cancer models. Mol. Cancer Ther. 10, 2350–2362 (2011).
Bastos, A.P. et al. Pkd1 haploinsufficiency increases renal damage and induces microcyst formation following ischemia/reperfusion. J. Am. Soc. Nephrol. 20, 2389–2402 (2009).
Wodarczyk, C. et al. Nephrocystin-1 forms a complex with polycystin-1 via a polyproline motif/SH3 domain interaction and regulates the apoptotic response in mammals. PLoS ONE 5, e12719 (2010).
Wodarczyk, C. et al. A novel mouse model reveals that polycystin-1 deficiency in ependyma and choroid plexus results in dysfunctional cilia and hydrocephalus. PLoS ONE 4, e7137 (2009).
Shao, X., Somlo, S. & Igarashi, P. Epithelial-specific Cre/lox recombination in the developing kidney and genitourinary tract. J. Am. Soc. Nephrol. 13, 1837–1846 (2002).
Acknowledgements
We thank other members of the lab Boletta and G. Di Grigoli for helpful discussion, R.M. Moresco for helpful suggestions and critically reading the manuscript, G. Casari and L. Cassina for help with the experiments on mitochondria, M. Giorgio for analysis of oxygen consumption and the San Raffaele microscopy facility (Alembic) for the electron microscopy studies (M.C. Panzeri) and tetramethylrhodamine (TMRM) analysis (M. Ascagni). V.M. is a student in the PhD Program of Biochemical, Nutritional and Metabolic Sciences, University of Milan. This work was supported by Telethon-Italy (TCR05007 to A.B. and TCP99035 to G.M.), US National Institutes of Health grants DK62199 (to F.Q.) and DK090868 (Johns Hopkins Polycystic Kidney Disease Research and Clinical Core Center, P30) and the Canadian Institutes of Health Research grant MOP123429 (to Y.P.). A.B. and G.M. are Associate Telethon Scientists. The lab Boletta is especially indebted to S. Bramani for her continuous, intelligent and motivating support.
Author information
Authors and Affiliations
Contributions
I.R. designed and performed the experiments, interpreted them and wrote the manuscript. M.C. designed and performed the in vivo experiments with 2DG treatment and 13-C glucose injections and interpreted the results. V.U. performed experiments in vitro on autophagy and signaling. M.P. generated the Ksp-Cre; Pkd1 mice, analyzed the kidneys biochemically and performed quantitative RT-PCR. A.B. designed the studies, supervised the work and collaborations and wrote the manuscript. V.M. prepared samples for metabolomic analysis, analyzed NMR spectra and performed statistical analysis. G.Q. acquired NMR spectra and performed statistical analysis. S.M. prepared samples for metabolomic analysis, acquired and analyzed NMR spectra and performed statistical analysis. G.M. supervised metabolomic analysis and discussed results. X.W.S. and Y.P. designed, performed and interpreted the human PKD1 renal cyst microarray experiment. H.X. and F.Q. designed and carried out the 2DG treatment experiment of Pkd1V/V mice and analyzed and interpreted the resulting data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Table 1 and Supplementary Methods (PDF 6337 kb)
Supplementary Table 2
Glycolytic gene expression in cystic as compared to minimally cuystic or normal renal tissues (XLS 62 kb)
Rights and permissions
About this article
Cite this article
Rowe, I., Chiaravalli, M., Mannella, V. et al. Defective glucose metabolism in polycystic kidney disease identifies a new therapeutic strategy. Nat Med 19, 488–493 (2013). https://doi.org/10.1038/nm.3092
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3092
This article is cited by
-
Deletion of Aurora kinase A prevents the development of polycystic kidney disease in mice
Nature Communications (2024)
-
The C-terminal tail of polycystin-1 suppresses cystic disease in a mitochondrial enzyme-dependent fashion
Nature Communications (2023)
-
Primary cilia sense glutamine availability and respond via asparagine synthetase
Nature Metabolism (2023)
-
Nutrient-sensing mTORC1 and AMPK pathways in chronic kidney diseases
Nature Reviews Nephrology (2023)
-
Fibrocystin/Polyductin releases a C-terminal fragment that translocates into mitochondria and suppresses cystogenesis
Nature Communications (2023)