Abstract
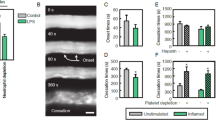
In contrast with many capillary beds, the glomerulus readily supports leukocyte recruitment. However, little is known regarding the actions of leukocytes following their recruitment to glomeruli. We used multiphoton confocal microscopy to examine leukocyte behavior in the glomerular microvasculature. In normal glomeruli, neutrophils and monocytes were retained in capillaries for several minutes, remaining static or migrating intravascularly. Induction of glomerular inflammation resulted in an increase in the duration of retention of static and migratory leukocytes. In response to immune complex deposition, both static and migratory neutrophils generated oxidants in inflamed glomeruli via a Mac-1–dependent mechanism. Our results describe a new paradigm for glomerular inflammation, suggesting that the major effect of acute inflammation is to increase the duration of leukocyte retention in the glomerulus. Moreover, these findings describe a previously unknown form of multicellular intravascular patrolling that involves both monocytes and neutrophils, which may underlie the susceptibility of the glomerulus to inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
12 August 2015
In the published article, in the Online Methods section, it is stated that the dose of DHE used is 20 mg/kg, when in fact DHE was administered at 2 mg/kg. The error has been corrected in the HTML and PDF versions of the article.
References
Cochrane, C.G., Unanue, E.R. & Dixon, F.J. A role of polymorphonuclear leukocytes and complement in nephrotoxic nephritis. J. Exp. Med. 122, 99–116 (1965).
Huang, X.R., Tipping, P.G., Shuo, L. & Holdsworth, S.R. Th1 responsiveness to nephritogenic antigens determines susceptibility to crescentic glomerulonephritis in mice. Kidney Int. 51, 94–103 (1997).
Tipping, P.G., Huang, X.R., Qi, M., Van, G.Y. & Tang, W.W. Crescentic glomerulonephritis in CD4- and CD8-deficient mice. Requirement for CD4 but not CD8 cells. Am. J. Pathol. 152, 1541–1548 (1998).
Duffield, J.S. et al. Conditional ablation of macrophages halts progression of crescentic glomerulonephritis. Am. J. Pathol. 167, 1207–1219 (2005).
Tipping, P.G., Huang, X.R., Berndt, M.C. & Holdsworth, S.R. A role for P selectin in complement-independent neutrophil-mediated glomerular injury. Kidney Int. 46, 79–88 (1994).
Tang, T. et al. A role for Mac-1 (CDIIb/CD18) in immune complex–stimulated neutrophil function in vivo: Mac-1 deficiency abrogates sustained Fcgamma receptor–dependent neutrophil adhesion and complement-dependent proteinuria in acute glomerulonephritis. J. Exp. Med. 186, 1853–1863 (1997).
Kuligowski, M.P., Kitching, A.R. & Hickey, M.J. Leukocyte recruitment to the inflamed glomerulus: a critical role for platelet-derived P-selectin in the absence of rolling. J. Immunol. 176, 6991–6999 (2006).
Massberg, S. et al. Platelet-endothelial cell interactions during ischemia/reperfusion: the role of P-selectin. Blood 92, 507–515 (1998).
Norman, M.U., Van De Velde, N.C., Timoshanko, J.R., Issekutz, A. & Hickey, M.J. Overlapping roles of endothelial selectins and vascular cell adhesion molecule 1 in immune complex–induced leukocyte recruitment in the cremasteric microvasculature. Am. J. Pathol. 163, 1491–1503 (2003).
Jung, U. & Ley, K. Regulation of E-selectin, P-selectin, and intercellular adhesion molecule 1 expression in mouse cremaster muscle vasculature. Microcirculation 4, 311–319 (1997).
Wong, J. et al. A minimal role for selectins in the recruitment of leukocytes into the inflamed liver microvasculature. J. Clin. Invest. 99, 2782–2790 (1997).
Downey, G.P., Worthen, G.S., Henson, P.M. & Hyde, D.M. Neutrophil sequestration and migration in localized pulmonary inflammation. Capillary localization and migration across the interalveolar septum. Am. Rev. Respir. Dis. 147, 168–176 (1993).
Kreisel, D. et al. In vivo two-photon imaging reveals monocyte-dependent neutrophil extravasation during pulmonary inflammation. Proc. Natl. Acad. Sci. USA 107, 18073–18078 (2010).
Mempel, T.R., Henrickson, S.E. & Von Andrian, U.H. T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004).
Matheu, M.P. et al. Imaging of effector memory T cells during a delayed-type hypersensitivity reaction and suppression by Kv1.3 channel block. Immunity 29, 602–614 (2008).
Geissmann, F. et al. Intravascular immune surveillance by CXCR6+ NKT cells patrolling liver sinusoids. PLoS Biol. 3, e113 (2005).
Auffray, C. et al. Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior. Science 317, 666–670 (2007).
Audoy-Rémus, J. et al. Rod-Shaped monocytes patrol the brain vasculature and give rise to perivascular macrophages under the influence of proinflammatory cytokines and angiopoietin-2. J. Neurosci. 28, 10187–10199 (2008).
Lee, W.Y. et al. An intravascular immune response to Borrelia burgdorferi involves Kupffer cells and iNKT cells. Nat. Immunol. 11, 295–302 (2010).
Kuligowski, M.P. et al. Antimyeloperoxidase antibodies rapidly induce alpha-4-integrin–dependent glomerular neutrophil adhesion. Blood 113, 6485–6494 (2009).
Devi, S. et al. Platelet recruitment to the inflamed glomerulus occurs via an alphaIIbbeta3/GPVI-dependent pathway. Am. J. Pathol. 177, 1131–1142 (2010).
Chiang, E.Y., Hidalgo, A., Chang, J. & Frenette, P.S. Imaging receptor microdomains on leukocyte subsets in live mice. Nat. Methods 4, 219–222 (2007).
Pippin, J.W. et al. Inducible rodent models of acquired podocyte diseases. Am. J. Physiol. Renal Physiol. 296, F213–F229 (2009).
Koshikawa, M. et al. Role of p38 mitogen-activated protein kinase activation in podocyte injury and proteinuria in experimental nephrotic syndrome. J. Am. Soc. Nephrol. 16, 2690–2701 (2005).
Kinugasa, S. et al. Selective albuminuria via podocyte albumin transport in puromycin nephrotic rats is attenuated by an inhibitor of NADPH oxidase. Kidney Int. 80, 1328–1338 (2011).
Smith, M.L., Olson, T.S. & Ley, K. CXCR2- and E-selectin–induced neutrophil arrest during inflammation in vivo. J. Exp. Med. 200, 935–939 (2004).
Sipos, A., Toma, I., Kang, J.J., Rosivall, L. & Peti-Peterdi, J. Advances in renal (patho)physiology using multiphoton microscopy. Kidney Int. 72, 1188–1191 (2007).
Boyce, N.W. & Holdsworth, S.R. Hydroxyl radical mediation of immune renal injury by desferrioxamine. Kidney Int. 30, 813–817 (1986).
Odobasic, D., Kitching, A.R., Semple, T.J. & Holdsworth, S.R. Endogenous myeloperoxidase promotes neutrophil-mediated renal injury, but attenuates T cell immunity inducing crescentic glomerulonephritis. J. Am. Soc. Nephrol. 18, 760–770 (2007).
Jackman, K.A. et al. Reduction of cerebral infarct volume by apocynin requires pretreatment and is absent in Nox2-deficient mice. Br. J. Pharmacol. 156, 680–688 (2009).
Chen, Z. et al. Decreased neointimal formation in Nox2-deficient mice reveals a direct role for NADPH oxidase in the response to arterial injury. Proc. Natl. Acad. Sci. USA 101, 13014–13019 (2004).
Phillipson, M. et al. Intraluminal crawling of neutrophils to emigration sites: a molecularly distinct process from adhesion in the recruitment cascade. J. Exp. Med. 203, 2569–2575 (2006).
McDonald, B. et al. Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science 330, 362–366 (2010).
Kitching, A.R., Holdsworth, S.R. & Tipping, P.G. Crescentic glomerulonephritis—a manifestation of a nephritogenic Th1 response? Histol. Histopathol. 15, 993–1003 (2000).
Summers, S.A. et al. Th1 and Th17 cells induce proliferative glomerulonephritis. J. Am. Soc. Nephrol. 20, 2518–2524 (2009).
Dapino, P., Dallegri, F., Ottonello, L. & Sacchetti, C. Induction of neutrophil respiratory burst by tumour necrosis factor-alpha; priming effect of solid-phase fibronectin and intervention of CD11b-CD18 integrins. Clin. Exp. Immunol. 94, 533–538 (1993).
Zhou, M.J. & Brown, E.J. CR3 (Mac-1, alpha M beta 2, CD11b/CD18) and Fc gamma RIII cooperate in generation of a neutrophil respiratory burst: requirement for Fc gamma RIII and tyrosine phosphorylation. J. Cell Biol. 125, 1407–1416 (1994).
Faust, N., Varas, F., Kelly, L.M., Heck, S. & Graf, T. Insertion of enhanced green fluorescent protein into the lysozyme gene creates mice with green fluorescent granulocytes and macrophages. Blood 96, 719–726 (2000).
Snelgrove, S.L. et al. Renal dendritic cells adopt a pro-inflammatory phenotype in obstructive uropathy to activate T cells, but do not directly contribute to fibrosis. Am. J. Pathol. 180, 91–103 (2011).
Bu, X. et al. Systemic administration of naked plasmid encoding HGF attenuates puromycin aminonucleoside–induced damage of murine glomerular podocytes. Am. J. Physiol. Renal Physiol. 301, F784–F792 (2011).
Dunne, J.L., Ballantyne, C.M., Beaudet, A.L. & Ley, K. Control of leukocyte rolling velocity in TNF-alpha–induced inflammation by LFA-1 and Mac-1. Blood 99, 336–341 (2002).
Acknowledgements
The authors gratefully acknowledge the assistance of J. Peti-Peterdi for advice on intravital imaging of the kidney, T. Graf (Center for Genomic Regulation) for LysM-eGFP mice, M. Tymms and E. Williams for assistance with fluorochrome labeling, D.-B. Borza (Vanderbilt University School of Medicine) for providing 8D1 monoclonal antibody, and C. Lo for technical assistance. This work was supported by a National Health and Medical Research Council (Australia) Program grant (#334067), a National Health and Medical Research Council (Australia) Project grant (#606564) and a Genzyme Renal Innovations Program Project grant. M.J.H. is a National Health and Medical Research Council (Australia) Senior Research Fellow.
Author information
Authors and Affiliations
Contributions
S.D. designed and performed the experiments and wrote the paper. A.L., C.L.V.W., L.D.A., S.L.S., P.H. and J.D.O. performed the experiments. C.Y.L. and C.G.S. contributed analytic tools. A.R.K. and M.J.H. designed the study and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 375 kb)
Supplementary Video 1
Neutrophil adhesion and migration in an uninflamed glomerulus. Video of a glomerulus in a hydronephrotic kidney of an untreated wild-type mouse. Gr-1+ neutrophils (red) can be seen undergoing periods of transient retention and migration within glomerular capillaries (glomerular capillaries stained with Alexa 488-conjugated isolectin GS-IB4). (Duration in real time = 10 min). (MOV 345 kb)
Supplementary Video 2
Retention and migration of GFP+ leukocytes in an uninflamed glomerulus of a CX3CR1GFP/+ mouse. Video of a glomerulus in a hydronephrotic kidney of an untreated CX3CR1GFP/+ mouse. A GFP+ monocyte can be seen undergoing prolonged migration in the glomerulus (capillaries labelled with rhodamine dextran) in the absence of an inflammatory stimulus. (Duration in real time = 35 min). (MOV 150 kb)
Supplementary Video 3
Neutrophil adhesion and migration in a control (NSG-treated) mouse. Video of a glomerulus of a wild-type mouse in the first hour after receiving non-immune sheep globulin (NSG) as control for anti-GBM Ab. Gr-1+ neutrophils (red) can be seen undergoing periods of transient retention and migration within glomerular capillaries (glomerular capillaries stained with Alexa 488-conjugated isolectin GS-IB4), consistent with the glomerular leukocyte behaviour seen in untreated mice. (Duration in real time = 20 min 30 s). (AVI 3742 kb)
Supplementary Video 4
Neutrophil adhesion and migration in an inflamed glomerulus. Video of a glomerulus of a wild-type mouse in the first hour following administration of anti-GBM Ab. Gr-1+ neutrophils (red) can be seen undergoing sustained interactions within glomerular capillaries (glomerular capillaries stained with Alexa 488-conjugated isolectin GS-IB4), either remaining static, or migrating within glomerular capillaries. (Duration in real time = 25 min). (AVI 4412 kb)
Supplementary Video 5
Leukocyte retention in capillaries of inflamed skeletal muscle. Multiphoton imaging of the cremaster muscle microvasculature following local TNF injection. Gr-1+ neutrophils (red) are visible moving within muscle capillaries (stained with FITC-conjugated isolectin GS-IB4). (Duration in real time = 10 min). (AVI 4437 kb)
Supplementary Video 6
Neutrophil adhesion and migration in an uninflamed glomerulus of an intact kidney. Video of a glomerulus in an intact (non-hydronephrotic) kidney of a wild-type mouse treated with NSG. Gr-1+ neutrophils (green) are visible undergoing periods of transient retention and migration within glomerular capillaries (vasculature labelled with rhodamine dextran). (Duration in real time = 20 min). (AVI 4217 kb)
Supplementary Video 7
Neutrophil adhesion and migration in an inflamed glomerulus of an intact kidney. Video of a glomerulus in an intact kidney of a wild-type mouse treated with anti-GBM Ab. Gr-1+ neutrophils (green) are visible undergoing lengthy periods of retention and migration within glomerular capillaries (vasculature labelled with rhodamine dextran). (Duration in real time = 20 min). (AVI 3883 kb)
Supplementary Video 8
Monocyte migration in an uninflamed glomerulus of an intact kidney. Video of a glomerulus in an intact kidney of an untreated CX3CR1GFP/+ mouse. A GFP+ monocyte can be seen undergoing prolonged migration in the glomerulus (capillaries labelled with rhodamine dextran). (Duration in real time = 11 min 30 s). (MOV 229 kb)
Supplementary Video 9
Neutrophil oxidant generation in uninflamed glomeruli detected by DHE. Video shows a glomerulus of a wild-type mouse 1–2 hrs after receiving NSG, and DHE (to facilitate detection of oxidant generation). Gr-1+ neutrophils (green) can be seen undergoing periods of transient retention and migration within glomerular capillaries (labelled with Pacific blue dextran), while minimal DHE-associated fluorescence (red) is detectable. Autofluorescent cells are present adjacent to the glomerulus. (Duration in real time = 5 min). (MOV 274 kb)
Supplementary Video 10
Neutrophil oxidant generation in inflamed glomeruli detected by DHE. Video shows a glomerulus of a wild-type mouse 1–2 hrs after receiving anti-GBM Ab, and DHE (to facilitate detection of oxidant generation). Gr-1+ neutrophils (green) can be seen undergoing sustained interactions within glomerular capillaries (labelled with Pacific blue dextran). Some neutrophils also display intracellular DHE-associated fluorescence (red). DHE+ neutrophils either remain static, or migrate within glomerular capillaries. Autofluorescent cells are present adjacent to the glomerulus. (Duration in real time = 5 min). (MOV 281 kb)
Supplementary Video 11
Neutrophil oxidant generation in Nox2-deficient mouse. Video shows a glomerulus of a Cybb−/− mouse 1–2 hrs after receiving anti-GBM Ab, and DHE, to facilitate detection of oxidant generation. Gr-1+ neutrophils (green) can be seen undergoing sustained static or migratory interactions within glomerular capillaries (labelled with Pacific blue dextran). However, no neutrophils show detectable DHE-associated fluorescence. (Duration in real time = 20 min). (MOV 907 kb)
Supplementary Video 12
Neutrophil behaviour in glomeruli of anti-GBM Ab-treated mouse following Mac-1 inhibition. Video of a glomerulus in an anti-GBM Ab-treated wild-type mouse following pretreatment with anti-Mac-1. 1–2 hrs after administration of anti-GBM Ab, interactions of Gr-1+ neutrophils in glomerular capillaries are predominantly transient, with cells subsequently exiting the glomerulus. (Duration in real time = 20 min). (AVI 3426 kb)
Rights and permissions
About this article
Cite this article
Devi, S., Li, A., Westhorpe, C. et al. Multiphoton imaging reveals a new leukocyte recruitment paradigm in the glomerulus. Nat Med 19, 107–112 (2013). https://doi.org/10.1038/nm.3024
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3024
This article is cited by
-
Renal microvascular endothelial cell responses in sepsis-induced acute kidney injury
Nature Reviews Nephrology (2022)
-
Immune cell behaviour and dynamics in the kidney — insights from in vivo imaging
Nature Reviews Nephrology (2022)
-
MEK inhibition exerts temporal and myeloid cell-specific effects in the pathogenesis of neurofibromatosis type 1 arteriopathy
Scientific Reports (2021)
-
Advances in fluorescence microscopy techniques to study kidney function
Nature Reviews Nephrology (2021)
-
Parietal epithelial cell dysfunction in crescentic glomerulonephritis
Cell and Tissue Research (2021)