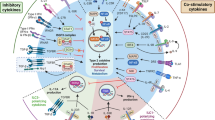
Abstract
The recognition that asthma is primarily an inflammatory disorder of the airways associated with T helper type 2 (TH2) cell-dependent promotion of IgE production and recruitment of mast cells and eosinophils has provided the rationale for disease control using inhaled corticosteroids and other anti-inflammatory drugs. As more has been discovered about the cytokine, chemokine and inflammatory pathways that are associated with TH2-driven adaptive immunity, attempts have been made to selectively inhibit these in the hope of discovering new therapeutics as predicted from animal models of allergic inflammation. The limited success of this approach, together with the recognition that asthma is more than allergic inflammation, has drawn attention to the innate immune response in this disease. Recent advances in our understanding of the sentinel role played by innate immunity provides new targets for disease prevention and treatment. These include pathways of innate stimulation by environmental or endogenous pathogen-associated molecular patterns (PAMPs) and danger-associated molecular patterns (DAMPs) to influence the activation and trafficking of DCs, innate sources of cytokines, and the identification of new T cell subsets and lymphoid cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bowen, H. et al. Control of cytokine gene transcription in TH1 and TH2 cells. Clin. Exp. Allergy 38, 1422–1431 (2008).
Das, J. et al. A critical role for NF-κB in GATA3 expression and TH2 differentiation in allergic airway inflammation. Nat. Immunol. 2, 45–50 (2001).
Alfvén, T. et al. Allergic diseases and atopic sensitization in children related to farming and anthroposophic lifestyle—the PARSIFAL study. Allergy 61, 414–421 (2006).
Ege, M.J. et al. Exposure to environmental microorganisms and childhood asthma. N. Engl. J. Med. 364, 701–709 (2011).
Ege, M.J. et al. Not all farming environments protect against the development of asthma and wheeze in children. J. Allergy Clin. Immunol. 119, 1140–1147 (2007).
Ege, M.J. et al. Prenatal exposure to a farm environment modifies atopic sensitization at birth. J. Allergy Clin. Immunol. 122, 407–412 (2008).
Sallmann, E. et al. High-affinity IgE receptors on dendritic cells exacerbate TH2-dependent inflammation. J. Immunol. 187, 164–171 (2011).
Nelson, D.J. et al. Development of the airway intraepithelial dendritic cell network in the rat from class II major histocompatibility (Ia)-negative precursors: differential regulation of Ia expression at different levels of the respiratory tract. J. Exp. Med. 179, 203–212 (1994).
McWilliam, A.S., Nelson, D., Thomas, J.A. & Holt, P.G. Rapid dendritic cell recruitment is a hallmark of the acute inflammatory response at mucosal surfaces. J. Exp. Med. 179, 1331–1336 (1994).
Hammad, H. et al. Inflammatory dendritic cells–not basophils–are necessary and sufficient for induction of Th2 immunity to inhaled house dust mite a llergen. J. Exp. Med. 207, 2097–2111 (2010).
Paul, W.E. & Zhu, J. How are TH2-type immune responses initiated and amplified? Nat. Rev. Immunol. 10, 225–235 (2010).
van Panhuys, N. et al. Basophils are the major producers of IL-4 during primary helminth infection. J. Immunol. 186, 2719–2728 (2011).
Trompette, A. et al. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature 457, 585–588 (2009).
Lu, T.X. et al. MicroRNA-21 limits in vivo immune response-mediated activation of the IL-12/IFN-γ pathway, Th1 polarization, and the severity of delayed-type hypersensitivity. J. Immunol. 187, 3362–3373 (2011).
Simpson, A. et al. Beyond atopy: multiple patterns of sensitization in relation to asthma in a birth cohort study. Am. J. Respir. Crit. Care Med. 181, 1200–1206 (2010).
Moffatt, M.F. et al. A large-scale, consortium-based genomewide association study of asthma. N. Engl. J. Med. 363, 1211–1221 (2010).
Granada, M. et al. A genome-wide association study of plasma total IgE concentrations in the Framingham Heart Study. J. Allergy Clin. Immunol. 129, 840–845 (2012).
Xiao, C. et al. Defective epithelial barrier function in asthma. J. Allergy Clin. Immunol. 128, 549–556 (2011).
Jacquet, A. Interactions of airway epithelium with protease allergens in the allergic response. Clin. Exp. Allergy 41, 305–311 (2011).
Hackett, T.L. et al. Intrinsic phenotypic differences of asthmatic epithelium and its inflammatory responses to RSV and air pollution. Am. J. Respir. Cell Mol. Biol. 45, 1090–1100 (2011).
Sajjan, U. et al. Rhinovirus disrupts the barrier function of polarized airway epithelial cells. Am. J. Respir. Crit. Care Med. 178, 1271–1281 (2008).
Groschwitz, K.R. & Hogan, S.P. Intestinal barrier function: molecular regulation and disease pathogenesis. J. Allergy Clin. Immunol. 124, 3–20 (2009).
Tieu, D.D. et al. Evidence for diminished levels of epithelial psoriasin and calprotectin in chronic rhinosinusitis. J. Allergy Clin. Immunol. 125, 667–675 (2010).
Hughes, J.L. et al. Reduced structural proteins in the conjunctival epithelium in allergic eye disease. Allergy 61, 1268–1274 (2006).
O'Regan, G.M., Sandilands, A., McLean, W.H. & Irvine, A.D. Filaggrin in atopic dermatitis. J. Allergy Clin. Immunol. 124 (suppl. 2), R2–R6 (2009).
McLean, W.H. The allergy gene: how a mutation in a skin protein revealed a link between eczema and asthma. F1000 Med. Rep. 3, 2 (2011).
Zoltán Veres, T. et al. Spatiotemporal and functional behavior of airway dendritic cells visualized by two-photon microscopy. Am. J. Pathol. 179, 603–609 (2011).
Blank, F. et al. Macrophages and dendritic cells express tight junction proteins and exchange particles in an in vitro model of the human airway wall. Immunobiology 216, 86–95 (2011).
Lambrecht, B.N. & Hammad, H. Biology of lung dendritic cells at the origin of asthma. Immunity 31, 412–424 (2009).
Kassianos, A.J., Jongbloed, S.L., Hart, D.N. & Radford, K.J. Isolation of human blood DC subtypes. Methods Mol. Biol. 595, 45–54 (2010).
Dua, B. et al. Myeloid and plasmacytoid dendritic cells in induced sputum after allergen inhalation in subjects with asthma. J. Allergy Clin. Immunol. 126, 133–139 (2010).
Sha, Q. et al. Activation of airway epithelial cells by Toll-like receptor agonists. Am. J. Respir. Cell Mol. Biol. 31, 358–364 (2004).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Willart, M.A. & Lambrecht, B.N. The danger within: endogenous danger signals, atopy and asthma. Clin. Exp. Allergy 39, 12–19 (2009).
Lam, D. et al. Airway house dust extract exposures modify allergen-induced airway hypersensitivity responses by TLR4-dependent and independent pathways. J. Immunol. 181, 2925–2932 (2008).
Perros, F., Hoogsteden, H.C., Coyle, A.J., Lambrecht, B.N. & Hammad, H. Blockade of CCR4 in a humanized model of asthma reveals a critical role for DC-derived CCL17 and CCL22 in attracting TH2 cells and inducing airway inflammation. Allergy 64, 995–1002 (2009).
Jackson, D.J. et al. Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am. J. Respir. Crit. Care Med. 178, 667–672 (2008).
Jartti, T. et al. Allergic sensitization is associated with rhinovirus-, but not other virus-, induced wheezing in children. Pediatr. Allergy Immunol. 21, 1008–1014 (2010).
Jackson, D.J. et al. Evidence for a causal relationship between allergic sensitization and rhinovirus wheezing in early life. Am. J. Respir. Crit. Care Med. 185, 281–285 (2012).
Wark, P.A. et al. Asthmatic bronchial epithelial cells have a deficient innate immune response to infection with rhinovirus. J. Exp. Med. 201, 937–947 (2005).
Contoli, M. et al. Role of deficient type III interferon-λ production in asthma exacerbations. Nat. Med. 12, 1023–1026 (2006).
Jartti, T. & Korppi, M. Rhinovirus-induced bronchiolitis and asthma development. Pediatr. Allergy Immunol. 22, 350–355 (2011).
Bosco, A. et al. Decreased activation of inflammatory networks during acute asthma exacerbations is associated with chronic airflow obstruction. Mucosal Immunol. 3, 399–409 (2010).
McWilliam, A.S. et al. Dendritic cells are recruited into the airway epithelium during the inflammatory response to a broad spectrum of stimuli. J. Exp. Med. 184, 2429–2432 (1996).
Uller, L. et al. Double-stranded RNA induces disproportionate expression of thymic stromal lymphopoietin versus interferon-ββ in bronchial epithelial cells from donors with asthma. Thorax 65, 626–632 (2010).
Cakebread, J.A. et al. Exogenous IFN-β has antiviral and anti-inflammatory properties in primary bronchial epithelial cells from asthmatic subjects exposed to rhinovirus. J. Allergy Clin. Immunol. 127, 1148–1154 (2011).
von Mutius, E. & Vercelli, D. Farm living: effects on childhood asthma and allergy. Nat. Rev. Immunol. 10, 861–868 (2010).
Schaub, B. et al. Maternal farm exposure modulates neonatal immune mechanisms through regulatory T cells. J. Allergy Clin. Immunol. 123, 774–782 (2009).
Gern, J.E. Barnyard microbes and childhood asthma. N. Engl. J. Med. 364, 769–770 (2011).
Haahtela, T. Allergy is rare where butterflies flourish in a biodiverse environment. Allergy 64, 1799–1803 (2009).
Rodriguez, B. et al. Germ-free status and altered caecal subdominant microbiota are associated with a high susceptibility to cow's milk allergy in mice. FEMS Microbiol. Ecol. 76, 133–144 (2011).
Hilty, M. et al. Disordered microbial communities in asthmatic airways. PLoS ONE 5, e8578 (2010).
Bisgaard, H. et al. Childhood asthma after bacterial colonization of the airway in neonates. N. Engl. J. Med. 357, 1487–1495 (2007).
Gern, J.E. Barnyard microbes and childhood asthma. N. Engl. J. Med. 364, 769–770 (2011).
Rabinovitch, N. et al. Importance of the personal endotoxin cloud in school-age children with asthma. J. Allergy Clin. Immunol. 116, 1053–1057 (2005).
Platts-Mills, J.A. et al. Airborne endotoxin in homes with domestic animals: implications for cat-specific tolerance. J. Allergy Clin. Immunol. 116, 384–389 (2005).
Shalev, I. et al. Making sense of regulatory T cell suppressive function. Semin. Immunol. 23, 282–292 (2011).
Akdis, C.A. & Akdis, M. Mechanisms of allergen-specific immunotherapy. J. Allergy Clin. Immunol. 127, 18–27 (2011).
Cady, C.T. et al. IgG antibodies produced during subcutaneous allergen immunotherapy mediate inhibition of basophil activation via a mechanism involving both FcγRIIA and FcγRIIB. Immunol. Lett. 130, 57–65 (2010).
Schaub, B. et al. Maternal farm exposure modulates neonatal immune mechanisms through regulatory T cells. J. Allergy Clin. Immunol. 123, 774–782 (2009).
Baru, A.M. et al. Selective depletion of Foxp3+ Treg during sensitization phase aggravates experimental allergic airway inflammation. Eur. J. Immunol. 40, 2259–2266 (2010).
Mold, J.E. et al. Maternal alloantigens promote the development of tolerogenic fetal regulatory T cells in utero. Science 322, 1562–1565 (2008).
Andersson, J. et al. CD4+ FoxP3+ regulatory T cells confer infectious tolerance in a TGF-β–dependent manner. J. Exp. Med. 205, 1975–1981 (2008).
Conrad, M.L. et al. Maternal TLR signaling is required for prenatal asthma protection by the nonpathogenic microbe Acinetobacter lwoffii F78. J. Exp. Med. 206, 2869–2877 (2009).
Brand, S. et al. Epigenetic regulation in murine offspring as a novel mechanism for transmaternal asthma protection induced by microbes. J. Allergy Clin. Immunol. 128, 618–625 (2011).
Rudd, C.E., Taylor, A. & Schneider, H. CD28 and CTLA-4 coreceptor expression and signal transduction. Immunol. Rev. 229, 12–26 (2009).
Kato, A. et al. TLR3- and TH2-cytokine–dependent production of thymic stromal lymphopoietin in human airway epithelial cells. J. Immunol. 179, 1080–1087 (2007).
Liew, F.Y., Pitman, N.I. & McInnes, I.B. Disease-associated functions of IL-33: the new kid in the IL-1 family. Nat. Rev. Immunol. 10, 103–110 (2010).
Wang, Y.H. et al. IL-25 augments type 2 immune responses by enhancing the expansion and functions of TSLP-DC–activated TH2 memory cells. J. Exp. Med. 204, 1837–1847 (2007).
Corrigan, C.J. et al. Allergen-induced expression of IL-25 and IL-25 receptor in atopic asthmatic airways and late-phase cutaneous responses. J. Allergy Clin. Immunol. 128, 116–124 (2011).
Iwakura, Y. et al. Functional specialization of interleukin-17 family members. Immunity 34, 149–162 (2011).
Saenz, S.A. et al. IL25 elicits a multipotent progenitor cell population that promotes TH2 cytokine responses. Nature 464, 1362–1366 (2010).
Neill, D.R. et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464, 1367–1370 (2010).
Barlow, J.L. et al. Innate IL-13–producing nuocytes arise during allergic lung inflammation and contribute to airways hyperreactivity. J. Allergy Clin. Immunol. 129, 191–198 (2012).
Yasuda, K. et al. Contribution of IL-33–activated type II innate lymphoid cells to pulmonary eosinophilia in intestinal nematode-infected mice. Proc. Natl. Acad. Sci. USA 109, 3451–3456 (2012).
Veldhoen, M. et al. Transforming growth factor-β 'reprograms' the differentiation of T helper 2 cells and promotes an interleukin 9–producing subset. Nat. Immunol. 9, 1341–1346 (2008).
Houssiau, F.A. et al. A cascade of cytokines is responsible for IL-9 expression in human T cells. Involvement of IL-2, IL-4, and IL-10. J. Immunol. 154, 2624–2630 (1995).
Goswami, R. et al. STAT6-dependent regulation of TH9 development. J. Immunol. 188, 968–975 (2012).
Faulkner, H., Renauld, J.C., Van Snick, J. & Grencis, R.K. Interleukin-9 enhances resistance to the intestinal nematode Trichuris muris. Infect. Immun. 66, 3832–3840 (1998).
Shimbara, A. et al. IL-9 and its receptor in allergic and nonallergic lung disease: increased expression in asthma. J. Allergy Clin. Immunol. 105, 108–115 (2000).
Erpenbeck, V.J. et al. Segmental allergen challenge in patients with atopic asthma leads to increased IL-9 expression in bronchoalveolar lavage fluid lymphocytes. J. Allergy Clin. Immunol. 111, 1319–1327 (2003).
Bryce, P.J. Revolution 9: the backwards and forwards evidence surrounding interleukin-9. Am. J. Respir. Crit. Care Med. 183, 834–835 (2011).
Steenwinckel, V. et al. IL-13 mediates in vivo IL-9 activities on lung epithelial cells but not on hematopoietic cells. J. Immunol. 178, 3244–3251 (2007).
Kearley, J. et al. IL-9 governs allergen-induced mast cell numbers in the lung and chronic remodeling of the airways. Am. J. Respir. Crit. Care Med. 183, 865–875 (2011).
Parker, J.M. et al. Safety profile and clinical activity of multiple subcutaneous doses of MEDI-528, a humanized anti–interleukin-9 monoclonal antibody, in two randomized phase 2a studies in subjects with asthma. BMC Pulm. Med. 11, 14 (2011).
Harrington, L.E. et al. Interleukin 17–producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 6, 1123–1132 (2005).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Nembrini, C., Marsland, B.J. & Kopf, M. IL-17–producing T cells in lung immunity and inflammation. J. Allergy Clin. Immunol. 123, 986–994 (2009).
Louten, J., Boniface, K. & de Waal Malefyt, R. Development and function of TH17 cells in health and disease. J. Allergy Clin. Immunol. 123, 1004–1011 (2009).
Wilson, R.H. et al. Allergic sensitization through the airway primes TH17-dependent neutrophilia and airway hyperresponsiveness. Am. J. Respir. Crit. Care Med. 180, 720–730 (2009).
Yang, X.O. et al. Regulation of inflammatory responses by IL-17F. J. Exp. Med. 205, 1063–1075 (2008).
Schnyder-Candrian, S. et al. Interleukin-17 is a negative regulator of established allergic asthma. J. Exp. Med. 203, 2715–2725 (2006).
Barlow, J.L. et al. Reciprocal expression of IL-25 and IL-17A is important for allergic airways hyperreactivity. Clin. Exp. Allergy 41, 1447–1455 (2011).
Kudo, M. et al. IL-17A produced by αβ T cells drives airway hyper-responsiveness in mice and enhances mouse and human airway smooth muscle contraction. Nat. Med. 18, 547–554 (2012).
Dunkelberger, J.R. & Song, W.C. Role and mechanism of action of complement in regulating T cell immunity. Mol. Immunol. 47, 2176–2186 (2010).
Humbles, A.A. et al. A role for the C3a anaphylatoxin receptor in the effector phase of asthma. Nature 406, 998–1001 (2000).
Drouin, S.M. et al. Absence of the complement anaphylatoxin C3a receptor suppresses TH2 effector functions in a murine model of pulmonary allergy. J. Immunol. 169, 5926–5933 (2002).
Baelder, R. et al. Pharmacological targeting of anaphylatoxin receptors during the effector phase of allergic asthma suppresses airway hyperresponsiveness and airway inflammation. J. Immunol. 174, 783–789 (2005).
Wills-Karp, M. Complement activation pathways: a bridge between innate and adaptive immune responses in asthma. Proc. Am. Thorac. Soc. 4, 247–251 (2007).
Cosmi, L. et al. Identification of a novel subset of human circulating memory CD4+ T cells that produce both IL-17A and IL-4. J. Allergy Clin. Immunol. 125, 222–230 (2010).
Chakir, J. et al. Airway remodeling-associated mediators in moderate to severe asthma: effect of steroids on TGF-β, IL-11, IL-17, and type I and type III collagen expression. J. Allergy Clin. Immunol. 111, 1293–1298 (2003).
Doe, C. et al. Expression of the T helper 17–associated cytokines IL-17A and IL-17F in asthma and COPD. Chest 138, 1140–1147 (2010).
Chaudhuri, R. et al. Effects of smoking cessation on lung function and airway inflammation in smokers with asthma. Am. J. Respir. Crit. Care Med. 174, 127–133 (2006).
Uddin, M. et al. Enhancement of neutrophil function by the bronchial epithelium stimulated by epidermal growth factor. Eur. Respir. J. 31, 714–724 (2008).
Capone, M. et al. Human invariant V α 24-J α Q TCR supports the development of CD1d-dependent NK1.1+ and NK1.1− T cells in transgenic mice. J. Immunol. 170, 2390–2398 (2003).
Brennan, P.J. et al. Invariant natural killer T cells recognize lipid self antigen induced by microbial danger signals. Nat. Immunol. 12, 1202–1211 (2011).
Meyer, E.H. et al. Glycolipid activation of invariant T cell receptor+ NK T cells is sufficient to induce airway hyperreactivity independent of conventional CD4+ T cells. Proc. Natl. Acad. Sci. USA 103, 2782–2787 (2006).
Akbari, O. et al. CD4+ invariant T-cell-receptor+ natural killer T cells in bronchial asthma. N. Engl. J. Med. 354, 1117–1129 (2006).
Vijayanand, P. et al. Invariant natural killer T cells in asthma and chronic obstructive pulmonary disease. N. Engl. J. Med. 356, 1410–1422 (2007).
Wingender, G. et al. Invariant NKT cells are required for airway inflammation induced by environmental antigens. J. Exp. Med. 208, 1151–1162 (2011).
Altin, J., Shen, C. & Liston, A. Understanding the genetic regulation of IgE production. Blood Rev. 24, 163–169 (2010).
Xiong, H. et al. Sequential class switching is required for the generation of high affinity IgE antibodies. J. Exp. Med. 209, 353–364 (2012).
Acharya, M. et al. CD23/FcɛRII: molecular multi-tasking. Clin. Exp. Immunol. 162, 12–23 (2010).
Ying, S. et al. Local expression of epsilon germline gene transcripts and RNA for the ɛ heavy chain of IgE in the bronchial mucosa in atopic and nonatopic asthma. J. Allergy Clin. Immunol. 107, 686–692 (2001).
Barnes, P.J. Intrinsic asthma: not so different from allergic asthma but driven by superantigens? Clin. Exp. Allergy 39, 1145–1151 (2009).
Bel, E. & ten Brinke, A. A rational approach to the management of severe refractory asthma. Treat. Respir. Med. 4, 365–379 (2005).
Kon, O.M. et al. Randomised, dose-ranging, placebo-controlled study of chimeric antibody to CD4 (keliximab) in chronic severe asthma. Lancet 352, 1109–1113 (1998).
Kon, O.M. et al. The effects of an anti-CD4 monoclonal antibody, keliximab, on peripheral blood CD4+ T cells in asthma. Eur. Respir. J. 18, 45–52 (2001).
Wang, X.F. et al. Interleukin-2 receptor antagonists in liver transplantation: a meta-analysis of randomized trials. Transplant. Proc. 42, 4567–4572 (2010).
Busse, W.W. et al. Daclizumab improves asthma control in patients with moderate to severe persistent asthma: a randomized, controlled trial. Am. J. Respir. Crit. Care Med. 178, 1002–1008 (2008).
Panina-Bordignon, P. et al. The C–C chemokine receptors CCR4 and CCR8 identify airway T cells of allergen-challenged atopic asthmatics. J. Clin. Invest. 107, 1357–1364 (2001).
Vijayanand, P. et al. Chemokine receptor 4 plays a key role in T cell recruitment into the airways of asthmatic patients. J. Immunol. 184, 4568–4574 (2010).
Reyes, C.L. Americas Antibody Congress 2009. 21–23 September 2009, Washington DC, USA. IDrugs 12, 692–694 (2009).
Yano, H. et al. Defucosylated anti CC chemokine receptor 4 monoclonal antibody combined with immunomodulatory cytokines: a novel immunotherapy for aggressive/refractory Mycosis fungoides and Sezary syndrome. Clin. Cancer Res. 13, 6494–6500 (2007).
Matsuoka, T. et al. Prostaglandin D2 as a mediator of allergic asthma. Science 287, 2013–2017 (2000).
Arima, M. & Fukuda, T. Prostaglandin D2 receptors DP and CRTH2 in the pathogenesis of asthma. Curr. Mol. Med. 8, 365–375 (2008).
Gervais, F.G. et al. Pharmacological characterization of MK-7246, a potent and selective CRTH2 (chemoattractant receptor-homologous molecule expressed on T-helper type 2 cells) antagonist. Mol. Pharmacol. 79, 69–76 (2011).
Barnes, N. et al. A randomized, double-blind, placebo-controlled study of the CRTH2 antagonist OC000459 in moderate persistent asthma. Clin. Exp. Allergy 42, 38–48 (2012).
Karman, J. et al. Ligation of cytotoxic T lymphocyte antigen-4 to the TCR inhibits T cell activation and directs differentiation into FOXP3+ regulatory T cells. J. Biol. Chem. published online (15 February 2012), doi:10.1074/jbc.M111.283705.
Álvarez-Quiroga, C. et al. CTLA-4-Ig therapy diminishes the frequency but enhances the function of Treg cells in patients with rheumatoid arthritis. J. Clin. Immunol. 31, 588–595 (2011).
Bour-Jordan, H. et al. Costimulation controls diabetes by altering the balance of pathogenic and regulatory T cells. J. Clin. Invest. 114, 979–987 (2004).
Jaffar, Z.H. et al. Essential role for both CD80 and CD86 costimulation, but not CD40 interactions, in allergen-induced TH2 cytokine production from asthmatic bronchial tissue: role for αβ, but not γδ, T cells. J. Immunol. 163, 6283–6291 (1999).
Hidi, R. et al. Role of B7–CD28/CTLA-4 costimulation and NF-κB in allergen-induced T cell chemotaxis by IL-16 and RANTES. J. Immunol. 164, 412–418 (2000).
Lordan, J.L. et al. The role of CD28–B7 costimulation in allergen-induced cytokine release by bronchial mucosa from patients with moderately severe asthma. J. Allergy Clin. Immunol. 108, 976–981 (2001).
Holgate, S.T. Pathophysiology of asthma: what has our current understanding taught us about new therapeutic approaches? J. Allergy Clin. Immunol. 128, 495–505 (2011).
Hansbro, P.M., Kaiko, G.E. & Foster, P.S. Cytokine/anti-cytokine therapy—novel treatments for asthma? Br. J. Pharmacol. 163, 81–95 (2011).
Berry, M.A. et al. Sputum and bronchial submucosal IL-13 expression in asthma and eosinophilic bronchitis. J. Allergy Clin. Immunol. 114, 1106–1109 (2004).
Woodruff, P.G. et al. T-helper type 2–driven inflammation defines major subphenotypes of asthma. Am. J. Respir. Crit. Care Med. 180, 388–395 (2009).
Corren, J. et al. Lebrikizumab treatment in adults with asthma. N. Engl. J. Med. 365, 1088–1098 (2011).
Nair, P. et al. Mepolizumab for prednisone-dependent asthma with sputum eosinophilia. N. Engl. J. Med. 360, 985–993 (2009).
Haldar, P. et al. Mepolizumab and exacerbations of refractory eosinophilic asthma. N. Engl. J. Med. 360, 973–984 (2009).
Abonia, J.P. & Putnam, P.E. Mepolizumab in eosinophilic disorders. Expert Rev. Clin. Immunol. 7, 411–417 (2011).
Castro, M. et al. Reslizumab for poorly controlled, eosinophilic asthma: a randomized, placebo-controlled study. Am. J. Respir. Crit. Care Med. 184, 1125–1132 (2011).
Spergel, J.M. et al. Reslizumab in children and adolescents with eosinophilic esophagitis: Results of a double-blind, randomized, placebo-controlled trial. J. Allergy Clin. Immunol. 129, 456–463 (2012).
Gevaert, P. et al. Nasal IL-5 levels determine the response to anti–IL-5 treatment in patients with nasal polyps. J. Allergy Clin. Immunol. 118, 1133–1141 (2006).
Kolbeck, R. et al. MEDI-563, a humanized anti–IL-5 receptor α mAb with enhanced antibody-dependent cell-mediated cytotoxicity function. J. Allergy Clin. Immunol. 125, 1344–1353 (2010).
Busse, W.W. et al. Safety profile, pharmacokinetics, and biologic activity of MEDI-563, an anti–IL-5 receptor α antibody, in a phase I study of subjects with mild asthma. J. Allergy Clin. Immunol. 125, 1237–1244 (2010).
Wenzel, S.E. et al. A randomized, double-blind, placebo-controlled study of tumor necrosis factor-α blockade in severe persistent asthma. Am. J. Respir. Crit. Care Med. 179, 549–558 (2009).
Silvestri, M. et al. High serum levels of tumour necrosis factor-α and interleukin-8 in severe asthma: markers of systemic inflammation? Clin. Exp. Allergy 36, 1373–1381 (2006).
Baines, K.J. et al. Differential gene expression and cytokine production from neutrophils in asthma phenotypes. Eur. Respir. J. 35, 522–531 (2010).
Holmes, A.M., Solari, R. & Holgate, S.T. Animal models of asthma: value, limitations and opportunities for alternative approaches. Drug Discov. Today 16, 659–670 (2011).
Acknowledgements
The author is a current UK Medical Research Council program grant holder.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
S.T.H. is a consultant to and nonexecutive director of Synairgen and an occasional consultant to Novartis, MSD and Amgen.
Rights and permissions
About this article
Cite this article
Holgate, S. Innate and adaptive immune responses in asthma. Nat Med 18, 673–683 (2012). https://doi.org/10.1038/nm.2731
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2731
This article is cited by
-
Increased expression of ficolin-1 is associated with airway obstruction in asthma
BMC Pulmonary Medicine (2023)
-
Magnetic-activated cell sorting identifies a unique lung microbiome community
Microbiome (2023)
-
Isthmin-1 attenuates allergic Asthma by stimulating adiponectin expression and alveolar macrophage efferocytosis in mice
Respiratory Research (2023)
-
Therapeutic properties of Helicobacter pylori-derived vacuolating cytotoxin A in an animal model of chronic allergic airway disease
Respiratory Research (2023)
-
Bei Mu Gua Lou San facilitates mucus expectoration by increasing surface area and hydration levels of airway mucus in an air-liquid-interface cell culture model of the respiratory epithelium
BMC Complementary Medicine and Therapies (2023)