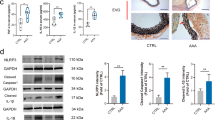
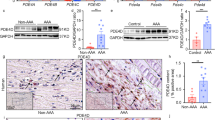
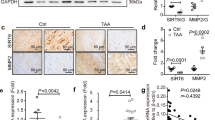
Abstract
Smoking is the only modifiable risk factor that is associated with the development, expansion and rupture of abdominal aortic aneurysm (AAA). However, the causative link between cigarette smoke and AAA is unknown. Here we report a causative link between smoking and AAA in vivo. Acute infusion of angiotensin II (AngII) or nicotine, a major component of cigarette smoke, markedly increased the incidence of AAA in apolipoprotein E (apoE) knockout (Apoe−/−) mice and in mice deficient in both apoE and the AMP-activated kinase α1 subunit (AMPK-α1) (Apoe−/−; Prkaa1−/− mice). In contrast, genetic deletion of AMPK-α2 (Apoe−/−; Prkaa2−/− mice) ablated nicotine- or AngII-triggered AAA in vivo. Mechanistically, we found that both nicotine and AngII activated AMPK-α2 in cultured vascular smooth muscle cells (VSMCs), resulting in the phosphorylation of activator protein 2α (AP-2α) and consequent matrix metallopeptidase 2 (MMP2) gene expression. We conclude that smoking (through nicotine) instigates AAA through AMPK-α2–mediated AP-2α–dependent MMP2 expression in VSMCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Vorp, D.A. & Vande Geest, J.P. Biomechanical determinants of abdominal aortic aneurysm rupture. Arterioscler. Thromb. Vasc. Biol. 25, 1558–1566 (2005).
Nordon, I.M., Hinchliffe, R.J., Loftus, I.M. & Thompson, M.M. Pathophysiology and epidemiology of abdominal aortic aneurysms. Nat. Rev. Cardiol. 8, 92–102 (2011).
Satoh, K. et al. Cyclophilin A enhances vascular oxidative stress and the development of angiotensin II–induced aortic aneurysms. Nat. Med. 15, 649–656 (2009).
Shah, P.K. Inflammation, metalloproteinases, and increased proteolysis: an emerging pathophysiological paradigm in aortic aneurysm. Circulation 96, 2115–2117 (1997).
Thompson, R.W., Geraghty, P.J. & Lee, J.K. Abdominal aortic aneurysms: basic mechanisms and clinical implications. Curr. Probl. Surg. 39, 110–230 (2002).
Rajagopalan, S. et al. Angiotensin II–mediated hypertension in the rat increases vascular superoxide production via membrane NADH/NADPH oxidase activation. Contribution to alterations of vasomotor tone. J. Clin. Invest. 97, 1916–1923 (1996).
Browatzki, M. et al. Angiotensin II stimulates matrix metalloproteinase secretion in human vascular smooth muscle cells via nuclear factor-κB and activator protein 1 in a redox-sensitive manner. J. Vasc. Res. 42, 415–423 (2005).
Thomas, M. et al. Deletion of p47phox attenuates angiotensin II–induced abdominal aortic aneurysm formation in apolipoprotein E–deficient mice. Circulation 114, 404–413 (2006).
Mu, J., Brozinick, J.T. Jr., Valladares, O., Bucan, M. & Birnbaum, M.J. A role for AMP-activated protein kinase in contraction- and hypoxia-regulated glucose transport in skeletal muscle. Mol. Cell 7, 1085–1094 (2001).
Choi, S.L. et al. The regulation of AMP-activated protein kinase by H(2)O(2). Biochem. Biophys. Res. Commun. 287, 92–97 (2001).
Han, Y., Wang, Q., Song, P., Zhu, Y. & Zou, M.H. Redox regulation of the AMP-activated protein kinase. PLoS ONE 5, e15420 (2010).
Zhang, M. et al. Thromboxane receptor activates the AMP-activated protein kinase in vascular smooth muscle cells via hydrogen peroxide. Circ. Res. 102, 328–337 (2008).
Nagata, D. et al. AMP-activated protein kinase inhibits angiotensin II–stimulated vascular smooth muscle cell proliferation. Circulation 110, 444–451 (2004).
Song, P. et al. AMPKα2 deletion exacerbates neointima formation by upregulating Skp2 in vascular smooth muscle cells. Circ. Res. 109, 1230–1239 (2011).
Henningfield, J.E., Stapleton, J.M., Benowitz, N.L., Grayson, R.F. & London, E.D. Higher levels of nicotine in arterial than in venous blood after cigarette smoking. Drug Alcohol Depend. 33, 23–29 (1993).
Benowitz, N.L. Cigarette smoking and cardiovascular disease: pathophysiology and implications for treatment. Prog. Cardiovasc. Dis. 46, 91–111 (2003).
Lu, H., Rateri, D.L., Cassis, L.A. & Daugherty, A. The role of the renin-angiotensin system in aortic aneurysmal diseases. Curr. Hypertens. Rep. 10, 99–106 (2008).
Daugherty, A. & Cassis, L.A. Mouse models of abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 24, 429–434 (2004).
Wang, Y.X. et al. Fasudil, a Rho-kinase inhibitor, attenuates angiotensin II–induced abdominal aortic aneurysm in apolipoprotein E–deficient mice by inhibiting apoptosis and proteolysis. Circulation 111, 2219–2226 (2005).
Daugherty, A. et al. Angiotensin II infusion promotes ascending aortic aneurysms: attenuation by CCR2 deficiency in apoE−/− mice. Clin. Sci. (Lond.) 118, 681–689 (2010).
Gavrila, D. et al. Vitamin E inhibits abdominal aortic aneurysm formation in angiotensin II–infused apolipoprotein E–deficient mice. Arterioscler. Thromb. Vasc. Biol. 25, 1671–1677 (2005).
Rateri, D.L. et al. Endothelial cell-specific deficiency of Ang II type 1a receptors attenuates Ang II–induced ascending aortic aneurysms in LDL receptor–/– mice. Circ. Res. 108, 574–581 (2011).
Pearce, W.H. & Shively, V.P. Abdominal aortic aneurysm as a complex multifactorial disease: interactions of polymorphisms of inflammatory genes, features of autoimmunity, and current status of MMPs. Ann. NY Acad. Sci. 1085, 117–132 (2006).
Aziz, F. & Kuivaniemi, H. Role of matrix metalloproteinase inhibitors in preventing abdominal aortic aneurysm. Ann. Vasc. Surg. 21, 392–401 (2007).
Longo, G.M. et al. Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J. Clin. Invest. 110, 625–632 (2002).
Pyo, R. et al. Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms. J. Clin. Invest. 105, 1641–1649 (2000).
Wang, S. et al. AMPKα2 deletion causes aberrant expression and activation of NAD(P)H oxidase and consequent endothelial dysfunction in vivo: role of 26S proteasomes. Circ. Res. 106, 1117–1128 (2010).
Park, M.J. et al. Nerve growth factor induces endothelial cell invasion and cord formation by promoting matrix metalloproteinase-2 expression through the phosphatidylinositol 3-kinase/Akt signaling pathway and AP-2 transcription factor. J. Biol. Chem. 282, 30485–30496 (2007).
Zhang, J. et al. Neural tube, skeletal and body wall defects in mice lacking transcription factor AP-2. Nature 381, 238–241 (1996).
Schorle, H., Meier, P., Buchert, M., Jaenisch, R. & Mitchell, P.J. Transcription factor AP-2 essential for cranial closure and craniofacial development. Nature 381, 235–238 (1996).
García, M.A., Campillos, M., Marina, A., Valdivieso, F. & Vazquez, J. Transcription factor AP-2 activity is modulated by protein kinase A–mediated phosphorylation. FEBS Lett. 444, 27–31 (1999).
Gwinn, D.M. et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol. Cell 30, 214–226 (2008).
Bungard, D. et al. Signaling kinase AMPK activates stress-promoted transcription via histone H2B phosphorylation. Science 329, 1201–1205 (2010).
Egan, D.F. et al. Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science 331, 456–461 (2011).
Kim, J., Kundu, M., Viollet, B. & Guan, K.L. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol. 13, 132–141 (2011).
Golledge, J. et al. Reduced expansion rate of abdominal aortic aneurysms in patients with diabetes may be related to aberrant monocyte-matrix interactions. Eur. Heart J. 29, 665–672 (2008).
Wang, S. et al. Acute inhibition of guanosine triphosphate cyclohydrolase 1 uncouples endothelial nitric oxide synthase and elevates blood pressure. Hypertension 52, 484–490 (2008).
Choi, H.C. et al. Reactive nitrogen species is required for the activation of the AMP-activated protein kinase by statin in vivo. J. Biol. Chem. 283, 20186–20197 (2008).
Miyama, N. et al. Hyperglycemia limits experimental aortic aneurysm progression. J. Vasc. Surg. 52, 975–983 (2010).
Dua, M.M. et al. Hyperglycemia modulates plasminogen activator inhibitor-1 expression and aortic diameter in experimental aortic aneurysm disease. Surgery 148, 429–435 (2010).
Nishikawa, K., Toker, A., Johannes, F.J., Songyang, Z. & Cantley, L.C. Determination of the specific substrate sequence motifs of protein kinase C isozymes. J. Biol. Chem. 272, 952–960 (1997).
Maegdefessel, L. et al. MicroRNA-21 blocks abdominal aortic aneurysm development and nicotine-augmented expansion. Sci. Transl. Med. 4, 122ra22 (2012).
An, Z. et al. Nicotine-induced activation of AMP-activated protein kinase inhibits fatty acid synthase in 3T3L1 adipocytes: a role for oxidant stress. J. Biol. Chem. 282, 26793–26801 (2007).
Dong, Y. et al. Reduction of AMP-activated protein kinase α2 increases endoplasmic reticulum stress and atherosclerosis in vivo. Circulation 121, 792–803 (2010).
Thompson, R.W. et al. Production and localization of 92-kilodalton gelatinase in abdominal aortic aneurysms. An elastolytic metalloproteinase expressed by aneurysm-infiltrating macrophages. J. Clin. Invest. 96, 318–326 (1995).
Corey, S. & Moran et al. Association of osteoprotegerin with human abdominal aortic aneurysm progression. Circulation. 111, 3119–3125 (2005).
Golledge, J., Muller, J., Daugherty, A. & Norman, P. Abdominal aortic aneurysm: pathogenesis and implications for management. Arterioscler. Thromb. Vasc. Biol. 26, 2605–2613 (2006).
Dallas, S.L., Rosser, J.L., Mundy, G.R. & Bonewald, L.F. Proteolysis of latent transforming growth factor-β (TGF-β)-binding protein-1 by osteoclasts. A cellular mechanism for release of TGF-β from bone matrix. J. Biol. Chem. 277, 21352–21360 (2002).
ten Dijke, P. & Arthur, H.M. Extracellular control of TGFβ signalling in vascular development and disease. Nat. Rev. Mol. Cell Biol. 8, 857–869 (2007).
Tundulawessa, Y., Yongchaiyud, P., Chutrthong, W. & Tundulawessa, K. The bioequivalent and effect of nicotine formulation gum on smoking cessation. J. Med. Assoc. Thai. 93, 574–579 (2010).
Viollet, B. et al. The AMP-activated protein kinase α2 catalytic subunit controls whole-body insulin sensitivity. J. Clin. Invest. 111, 91–98 (2003).
Sparks, A.R., Johnson, P.L. & Meyer, M.C. Imaging of abdominal aortic aneurysms. Am. Fam. Physician 65, 1565–1570 (2002).
Lukasova, M., Malaval, C., Gille, A., Kero, J. & Offermanns, S. Nicotinic acid inhibits progression of atherosclerosis in mice through its receptor GPR109A expressed by immune cells. J. Clin. Invest. 121, 1163–1173 (2011).
Matute-Bello, G. et al. Optimal timing to repopulation of resident alveolar macrophages with donor cells following total body irradiation and bone marrow transplantation in mice. J. Immunol. Methods 292, 25–34 (2004).
Xie, Z. et al. Identification of the serine 307 of LKB1 as a novel phosphorylation site essential for its nucleocytoplasmic transport and endothelial cell angiogenesis. Mol. Cell. Biol. 29, 3582–3596 (2009).
Wang, S., Peng, Q., Zhang, J. & Liu, L. Na+/H+ exchanger is required for hyperglycaemia-induced endothelial dysfunction via calcium-dependent calpain. Cardiovasc. Res. 80, 255–262 (2008).
Wang, S., Xu, J., Song, P., Viollet, B. & Zou, M.H. In vivo activation of AMP-activated protein kinase attenuates diabetes-enhanced degradation of GTP cyclohydrolase I. Diabetes 58, 1893–1901 (2009).
Zhang, J. et al. Identification of nitric oxide as an endogenous activator of the AMP-activated protein kinase in vascular endothelial cells. J. Biol. Chem. 283, 27452–27461 (2008).
Acknowledgements
We are grateful for helpful discussions with J. Du, X.L. Wang and C. Yan. We also thank D. Wang and M. McDaniel for technical support. This work was supported by US National Institutes of Health (NIH) grants (HL079584, HL074399, HL080499, HL089920, HL096032, HL105157 and HL110488 to M.-H.Z.). L.X.'s lab is supported by NIH grants (HL085607 and RR018758). This work was also supported by the National High-tech Research and Development Program of China (No.2012AA02A510) awarded to C.Z. and the National 973 Basic Research Program of China (number 2011CB503906) and the State Program of National Natural Science Foundation of China for Innovative Research Group (number 81021001) awarded to Y.Z. M.-H.Z. is a recipient of the National Established Investigator Award of the American Heart Association.
Author information
Authors and Affiliations
Contributions
S.W. designed and conducted the experiments, analyzed data and wrote the manuscript. C.Z. and Y.Z. did the clinical experiments. M.Z. and L.X. did bone morrow transplantation. B.L. partially performed the pathological experiments. H.Z. and J.L. generated series mutants. B.V. provided the AMPK-deleted mice. M.-H.Z. conceived of the project, designed the experiments, analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12 and Supplementary Tables 1–10 (PDF 5097 kb)
Rights and permissions
About this article
Cite this article
Wang, S., Zhang, C., Zhang, M. et al. Activation of AMP-activated protein kinase α2 by nicotine instigates formation of abdominal aortic aneurysms in mice in vivo. Nat Med 18, 902–910 (2012). https://doi.org/10.1038/nm.2711
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2711
This article is cited by
-
S-nitrosylation of AMPKγ impairs coronary collateral circulation and disrupts VSMC reprogramming
EMBO Reports (2023)
-
Phosphodiesterase 4D contributes to angiotensin II-induced abdominal aortic aneurysm through smooth muscle cell apoptosis
Experimental & Molecular Medicine (2022)
-
An intersegmental single-cell profile reveals aortic heterogeneity and identifies a novel Malat1+ vascular smooth muscle subtype involved in abdominal aortic aneurysm formation
Signal Transduction and Targeted Therapy (2022)
-
A positive feedback loop between Periostin and TGFβ1 induces and maintains the stemness of hepatocellular carcinoma cells via AP-2α activation
Journal of Experimental & Clinical Cancer Research (2021)
-
Systems toxicology study reveals reduced impact of heated tobacco product aerosol extract relative to cigarette smoke on premature aging and exacerbation effects in aged aortic cells in vitro
Archives of Toxicology (2021)