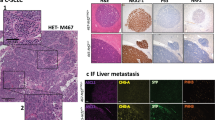
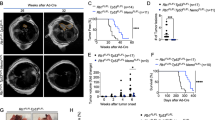
Abstract
Small-cell lung cancer (SCLC) is an aggressive neuroendocrine subtype of lung cancer for which there is no effective treatment1,2. Using a mouse model in which deletion of Rb1 and Trp53 in the lung epithelium of adult mice induces SCLC3,4, we found that the Hedgehog signaling pathway is activated in SCLC cells independently of the lung microenvironment. Constitutive activation of the Hedgehog signaling molecule Smoothened (Smo) promoted the clonogenicity of human SCLC in vitro and the initiation and progression of mouse SCLC in vivo. Reciprocally, deletion of Smo in Rb1 and Trp53-mutant lung epithelial cells strongly suppressed SCLC initiation and progression in mice. Furthermore, pharmacological blockade of Hedgehog signaling inhibited the growth of mouse and human SCLC, most notably following chemotherapy. These findings show a crucial cell-intrinsic role for Hedgehog signaling in the development and maintenance of SCLC and identify Hedgehog pathway inhibition as a therapeutic strategy to slow the progression of disease and delay cancer recurrence in individuals with SCLC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gustafsson, B.I., Kidd, M., Chan, A., Malfertheiner, M.V. & Modlin, I.M. Bronchopulmonary neuroendocrine tumors. Cancer 113, 5–21 (2008).
Rudin, C.M., Hann, C.L., Peacock, C.D. & Watkins, D.N. Novel systemic therapies for small cell lung cancer. J. Natl. Compr. Canc. Netw. 6, 315–322 (2008).
Meuwissen, R. et al. Induction of small cell lung cancer by somatic inactivation of both Trp53 and Rb1 in a conditional mouse model. Cancer Cell 4, 181–189 (2003).
Schaffer, B.E. et al. Loss of p130 accelerates tumor development in a mouse model for human small-cell lung carcinoma. Cancer Res. 70, 3877–3883 (2010).
Vestergaard, J. et al. Hedgehog signaling in small-cell lung cancer: frequent in vivo but a rare event in vitro. Lung Cancer 52, 281–290 (2006).
Chi, S. et al. Activation of the hedgehog pathway in a subset of lung cancers. Cancer Lett. 244, 53–60 (2006).
Watkins, D.N. et al. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422, 313–317 (2003).
Teglund, S. & Toftgard, R. Hedgehog beyond medulloblastoma and basal cell carcinoma. Biochim. Biophys. Acta 1805, 181–208 (2010).
Voortman, J. et al. Array comparative genomic hybridization-based characterization of genetic alterations in pulmonary neuroendocrine tumors. Proc. Natl. Acad. Sci. USA 107, 13040–13045 (2010).
Yauch, R.L. et al. A paracrine requirement for hedgehog signalling in cancer. Nature 455, 406–410 (2008).
Goodrich, L.V., Milenkovic, L., Higgins, K.M. & Scott, M.P. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 277, 1109–1113 (1997).
Jackson, E.L. et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 15, 3243–3248 (2001).
Sutherland, K.D. et al. Cell of origin of small cell lung cancer: inactivation of Trp53 and rb1 in distinct cell types of adult mouse lung. Cancer Cell 19, 754–764 (2011).
Park, K.S. et al. Characterization of the cell of origin for small cell lung cancer. Cell Cycle 10, 2806–2815 (2011).
Taipale, J. et al. Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine. Nature 406, 1005–1009 (2000).
Milenkovic, L., Scott, M.P. & Rohatgi, R. Lateral transport of Smoothened from the plasma membrane to the membrane of the cilium. J. Cell Biol. 187, 365–374 (2009).
Kim, J., Kato, M. & Beachy, P.A. Gli2 trafficking links Hedgehog-dependent activation of Smoothened in the primary cilium to transcriptional activation in the nucleus. Proc. Natl. Acad. Sci. USA 106, 21666–21671 (2009).
Mao, J. et al. A novel somatic mouse model to survey tumorigenic potential applied to the Hedgehog pathway. Cancer Res. 66, 10171–10178 (2006).
Stecca, B. & Ruiz i Altaba, A.A. GLI1-p53 inhibitory loop controls neural stem cell and tumour cell numbers. EMBO J. 28, 663–676 (2009).
Cooper, M.K., Porter, J.A., Young, K.E. & Beachy, P.A. Teratogen-mediated inhibition of target tissue response to Shh signaling. Science 280, 1603–1607 (1998).
Hyman, J.M. et al. Small-molecule inhibitors reveal multiple strategies for Hedgehog pathway blockade. Proc. Natl. Acad. Sci. USA 106, 14132–14137 (2009).
Lauth, M., Bergstrom, A., Shimokawa, T. & Toftgard, R. Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc. Natl. Acad. Sci. USA 104, 8455–8460 (2007).
Buonamici, S. et al. Interfering with resistance to smoothened antagonists by inhibition of the PI3K pathway in medulloblastoma. Sci. Transl. Med. 2, 51ra70 (2010).
Pan, S. et al. Discovery of NVP-LDE225, a potent and selective Smoothened antagonist. ACS Med Chem Lett 1, 130–134 (2010).
Daniel, V.C. et al. A primary xenograft model of small-cell lung cancer reveals irreversible changes in gene expression imposed by culture in vitro. Cancer Res. 69, 3364–3373 (2009).
Peacock, C.D. et al. Hedgehog signaling maintains a tumor stem cell compartment in multiple myeloma. Proc. Natl. Acad. Sci. USA 104, 4048–4053 (2007).
Scales, S.J. & de Sauvage, F.J. Mechanisms of Hedgehog pathway activation in cancer and implications for therapy. Trends Pharmacol. Sci. 30, 303–312 (2009).
Berman, D.M. et al. Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 297, 1559–1561 (2002).
Rudin, C.M. et al. Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. N. Engl. J. Med. 361, 1173–1178 (2009).
Sanchez, P. et al. Inhibition of prostate cancer proliferation by interference with SONIC HEDGEHOG-GLI1 signaling. Proc. Natl. Acad. Sci. USA 101, 12561–12566 (2004).
Stecca, B. et al. Melanomas require HEDGEHOG-GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. Proc. Natl. Acad. Sci. USA 104, 5895–5900 (2007).
Clement, V., Sanchez, P., de Tribolet, N., Radovanovic, I. & Ruiz i Altaba, A. HEDGEHOG-GLI1 signaling regulates human glioma growth, cancer stem cell self-renewal, and tumorigenicity. Curr. Biol. 17, 165–172 (2007).
Varnat, F. et al. Human colon cancer epithelial cells harbour active HEDGEHOG-GLI signalling that is essential for tumour growth, recurrence, metastasis and stem cell survival and expansion. EMBO Mol. Med. 1, 338–351 (2009).
Arimura, S. et al. Reduced level of smoothened suppresses intestinal tumorigenesis by down-regulation of Wnt signaling. Gastroenterology 137, 629–638 (2009).
Thayer, S.P. et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425, 851–856 (2003).
Yuan, Z. et al. Frequent requirement of hedgehog signaling in non-small cell lung carcinoma. Oncogene 26, 1046–1055 (2007).
Singh, S. et al. Hedgehog-producing cancer cells respond to and require autocrine Hedgehog activity. Cancer Res. 71, 4454–4463 (2011).
Sage, J., Miller, A.L., Perez-Mancera, P.A., Wysocki, J.M. & Jacks, T. Acute mutation of retinoblastoma gene function is sufficient for cell cycle re-entry. Nature 424, 223–228 (2003).
Long, F., Zhang, X.M., Karp, S., Yang, Y. & McMahon, A.P. Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation. Development 128, 5099–5108 (2001).
Humke, E.W., Dorn, K.V., Milenkovic, L., Scott, M.P. & Rohatgi, R. The output of Hedgehog signaling is controlled by the dynamic association between Suppressor of Fused and the Gli proteins. Genes Dev. 24, 670–682 (2010).
Acknowledgements
We would like to thank A. Berns (The Netherlands Cancer Institute) and M. Scott (Stanford University) for the Trp53lox and Ptch1lacZ/+ mice, respectively, J. Whitsett (Cincinnati Children's Hospital) for the antibodies to surfactant protein C, C. Janke (Orsay Curie Institute) for antibodies to polyglutamylated tubulin and C.-M. Fan (Carnegie Institution of Washington) for the adenovirally expressed SmoM2 virus, as well as T. Oro, James Kim, Jynho Kim and P. Beachy for helpful discussions throughout the course of this study. We thank R. Toftgård (Karolinska Institute) and K. McGovern at Infinity Pharmaceuticals for their generous gift of the GANT-61 and cyclopamine, respectively, R. Rohatgi for help with the immunoblot analysis and B. Schaffer for help with the cell culture. This work was supported by the Lucile Packard Foundation for Children's Health (J.S.), the Damon Runyon Cancer Research Foundation (J.S.), the American Lung Association (J.S. and K.-S.P.), the Francis Family Foundation (K.-S.P.), the American Cancer Society (J.S.), the Tobacco-Related Disease Research Program of California (J.F.C.), US National Institutes of Health (NIH) National Cancer Institute R01 CA136574 (J.K.C.), the Flight Attendant Medical Research Institute (YCSA 072033) (C.D.P.), NIH 5T32 CA009302-33 (C.O.), the National Health and Medical Research Council of Australia project grants 546024 and 546098 and the Victorian Cancer Agency (D.N.W., L.G.M. and A.S.), the Deutsche Krebshilfe (107954) (R.K.T.), the German Ministry of Science and Education as part of the Nationales Genomforschungsnetz (NGFN-plus) program (01GS08100) (R.K.T.), the Max Planck Society (MIFA NEUR8061) (R.K.T.), the Deutsche Forschungsgemeinschaft (DFG) through Sonderforschungsbereiche (TP6) (R.K.T.), the Ministry for Innovation, Science, Research and Technology of the State of Nordrhein-Westfalen (MIWT, 4000-12 09) (R.K.T.) and by an anonymous foundation to R.K.T.
Author information
Authors and Affiliations
Contributions
A.N.K. analyzed the histopathology of all mouse lung tumors and edited the manuscript. A.S. performed and analyzed the immunohistochemistry. D.N.W. and L.G.M. edited the manuscript and analyzed the immunohistochemistry. L.G.M. performed experiments with Smo inhibitors and chemotherapy in culture and in xenografts. C.A.O. and J.K.C. generated HPI-1 for cell culture experiments. J.K.C. edited the manuscript. M.R.M. and T.S. designed, performed and analyzed the experiments related to the primary cilia in mouse cells. M.P., M.L.S. and R.K.T. designed and performed the experiments related to the genomic analysis of mouse and human tumors. K.-S.P. and K.B. quantified the proliferation and survival phenotypes in tumors treated with cyclopamine. K.-S.P. and J.F.C. analyzed gene and protein expression levels in tumor cells. K.-S.P. performed all the other experiments involving mouse cells. K.-S.P. and J.S. designed the experiments for the analysis of mouse SCLC cells in culture and in vivo, and generated the corresponding figures. C.D.P. designed and analyzed the research performed by A.M. and W.L.D. on the human SCLC cells in vitro. J.F., S.Buonamici, S. Bennett, J.Y., R.G., B.O., M.D., A.M., W.L.D. and T.J.B. designed and performed in vivo xenograft experiments and analyzed the data. K.-S.P., J.S., C.D.P. and S.B. wrote and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
D.N.W. is a co-inventor on a patent application for the use of Smo antagonists in SCLC. J.K.C. has consulted for Fate Therapeutics, and J.S. has consulted for Infinity Pharmaceuticals. R.K.T. received consulting and lecture fees (from Sequenom, Sanofi-Aventis, Merck, Roche, Infinity, Boehringer, AstraZeneca and ATLAS Biolabs) as well as research support (from Novartis and AstraZeneca). S.B., J.Y., B.O. and R.G. are employees of Novartis Institute of BioMedical Research. M.D. is an employee of Sanofi-aventis.
Supplementary information
Supplementary Text and Figures
Supplementary Discussion, Supplementary Methods, Supplementary Figures 1–8 and Supplementary Tables 1 and 2 (PDF 1786 kb)
Rights and permissions
About this article
Cite this article
Park, KS., Martelotto, L., Peifer, M. et al. A crucial requirement for Hedgehog signaling in small cell lung cancer. Nat Med 17, 1504–1508 (2011). https://doi.org/10.1038/nm.2473
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2473
This article is cited by
-
Genetically-engineered mouse models of small cell lung cancer: the next generation
Oncogene (2024)
-
Orchestrating smart therapeutics to achieve optimal treatment in small cell lung cancer: recent progress and future directions
Journal of Translational Medicine (2023)
-
Ligand-dependent hedgehog signaling maintains an undifferentiated, malignant osteosarcoma phenotype
Oncogene (2023)
-
Signal pathways and precision therapy of small-cell lung cancer
Signal Transduction and Targeted Therapy (2022)
-
Drug resistance and Cancer stem cells
Cell Communication and Signaling (2021)