Abstract
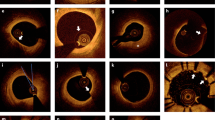
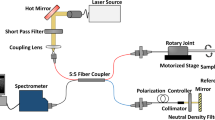
Progress in understanding, diagnosis, and treatment of coronary artery disease (CAD) has been hindered by our inability to observe cells and extracellular components associated with human coronary atherosclerosis in situ. The current standards for microstructural investigation, histology and electron microscopy are destructive and prone to artifacts. The highest-resolution intracoronary imaging modality, optical coherence tomography (OCT), has a resolution of ∼10 μm, which is too coarse for visualizing most cells. Here we report a new form of OCT, termed micro–optical coherence tomography (μOCT), whose resolution is improved by an order of magnitude. We show that μOCT images of cadaver coronary arteries provide clear pictures of cellular and subcellular features associated with atherogenesis, thrombosis and responses to interventional therapy. These results suggest that μOCT can complement existing diagnostic techniques for investigating atherosclerotic specimens, and that μOCT may eventually become a useful tool for cellular and subcellular characterization of the human coronary wall in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Virmani, R., Kolodgie, F.D., Burke, A.P., Farb, A. & Schwartz, S.M. Lessons from sudden coronary death—a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler. Thromb. Vasc. Biol. 20, 1262–1275 (2000).
Stary, H.C. et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis: a report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 92, 1355–1374 (1995).
Grønholdt, M.L.M., Dalager-Pedersen, S. & Falk, E. Coronary atherosclerosis: determinants of plaque rupture. Eur. Heart J. 19, C24–C29 (1998).
Davies, M.J. Acute coronary thrombosis—The role of plaque disruption and its initiation and prevention. Eur. Heart J. 16, 3–7 (1995).
Pasternak, R.C., Baughman, K.L., Fallon, J.T. & Block, P.C. Scanning electron microscopy after coronary transluminal angioplasty of normal canine coronary arteries. Am. J. Cardiol. 45, 591–598 (1980).
Bourassa, M.G., Cantin, M., Sandborn, E.B. & Pederson, E. Scanning electron microscopy of surface irregularities and thrombogenesis of polyurethane and polyethylene coronary catheters. Circulation 53, 992–996 (1976).
Abela, G.S. & Aziz, K. Cholesterol crystals rupture biological membranes and human plaques during acute cardiovascular events–a novel insight into plaque rupture by scanning electron microscopy. Scanning 28, 1–10 (2006).
Huang, D. et al. Optical coherence tomography. Science 254, 1178–1181 (1991).
Jang, I.K. et al. Visualization of coronary atherosclerotic plaques in patients using optical coherence tomography: comparison with intravascular ultrasound. J. Am. Coll. Cardiol. 39, 604–609 (2002).
Yun, S.H. et al. Comprehensive volumetric optical microscopy in vivo. Nat. Med. 12, 1429–1433 (2006).
Tearney, G.J. et al. Three-dimensional coronary artery microscopy by intracoronary optical frequency domain imaging. JACC Cardiovasc. Imaging 1, 752–761 (2008).
Boppart, S.A. et al. In vivo cellular optical coherence tomography imaging. Nat. Med. 4, 861–865 (1998).
Povazay, B. et al. Submicrometer axial resolution optical coherence tomography. Opt. Lett. 27, 1800–1802 (2002).
Ralston, T.S., Marks, D.L., Carney, P.S. & Boppart, S.A. Interferometric synthetic aperture microscopy. Nat. Phys. 3, 129–134 (2007).
Liu, L., Liu, C., Howe, W.C., Sheppard, C.J.R. & Chen, N.Q. Binary-phase spatial filter for real-time swept-source optical coherence microscopy. Opt. Lett. 32, 2375–2377 (2007).
Ding, Z., Ren, H.W., Zhao, Y.H., Nelson, J.S. & Chen, Z.P. High-resolution optical coherence tomography over a large depth range with an axicon lens. Opt. Lett. 27, 243–245 (2002).
Lee, K.S. & Rolland, L.P. Bessel beam spectral-domain high-resolution optical coherence tomography with micro-optic axicon providing extended focusing range. Opt. Lett. 33, 1696–1698 (2008).
Leitgeb, R.A., Villiger, M., Bachmann, A.H., Steinmann, L. & Lasser, T. Extended focus depth for Fourier domain optical coherence microscopy. Opt. Lett. 31, 2450–2452 (2006).
Vakhtin, A.B., Kane, D.J., Wood, W.R. & Peterson, K.A. Common-path interferometer for frequency-domain optical coherence tomography. Appl. Opt. 42, 6953–6958 (2003).
Hausler, G. & Lindner, M.W. “Coherence radar” and “Spectral radar”—new tools for dermatological diagnosis. J. Biomed. Opt. 3, 21–31 (1998).
Finn, A.V. et al. Pathological correlates of late drug-eluting stent thrombosis—strut coverage as a marker of endothelialization. Circulation 115, 2435–2441 (2007).
Libby, P. Changing concepts of atherogenesis. J. Intern. Med. 247, 349–358 (2000).
Lusis, A.J. Atherosclerosis. Nature 407, 233–241 (2000).
Virmani, R., Burke, A.P., Farb, A. & Kolodgie, F.D. Pathology of the vulnerable plaque. J. Am. Coll. Cardiol. 47, C13–C18 (2006).
Vengrenyuk, Y., Cardoso, L. & Weinbaum, S. μCT-based analysis of a new paradigm for vulnerable plaque rupture: cellular microcalcifications in fibrous caps. Mol. Cell. Biomech. 5, 37–47 (2008).
Serruys, P.W. et al. A bioabsorbable everolimus-eluting coronary stent system (ABSORB): 2-year outcomes and results from multiple imaging methods. Lancet 373, 897–910 (2009).
Rasband, W.S. ImageJ. (US National Institutes of Health, Bethesda, Maryland, 1997–2011).
Acknowledgements
We wish to acknowledge contributions from D. Winsor-Hines of Boston Scientific for providing the stents that were used to create Supplementary Figure 4 , G. Veytsman of Capital Biosciences for providing explanted human hearts, and J. Zhao and the Wellman Center Photopathology Laboratory staff for expert histology processing. Swine arterial tissue was obtained from the Massachusetts General Hospital Knight Surgical Laboratory. Human tissue was provided by the National Disease Research Interchange and Capital Biosciences. Endothelial cells were supplied by the Schepens Eye Research Institute. This research was supported in part by the US National Institutes of Health (contracts R01HL076398 and R01HL093717) and the Cystic Fibrosis Foundation (contract TEARNE07XX0).
Author information
Authors and Affiliations
Contributions
L.L. developed the μOCT system and participated in conducting the imaging studies and writing the manuscript. J.A.G. was responsible for procuring and preparing specimens, preparing the specimens for histopathology and organizing all digital histopathology data. S.K.N. and J.D.T. prepared the endothelial cell cultures. Y.Y. digitized the histopathology slides using her full-slide scanning systems. L.L. and G.J.T. analyzed and processed the data. B.E.B. contributed to the study design and participated in the analysis of the data. G.J.T. supervised the overall project and contributed to the design of experiments, interpretation of the μOCT image data and preparation of the manuscript. All authors read and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
Massachusetts General Hospital has licensed OFDI technology to Terumo Corporation. B.E.B. and G.J.T. receive sponsored research relating to OFDI technology development from Terumo Corporation. B.E.B. and G.J.T. also have the right to receive royalty payments as part of this licensing arrangement.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Methods (PDF 1109 kb)
Rights and permissions
About this article
Cite this article
Liu, L., Gardecki, J., Nadkarni, S. et al. Imaging the subcellular structure of human coronary atherosclerosis using micro–optical coherence tomography. Nat Med 17, 1010–1014 (2011). https://doi.org/10.1038/nm.2409
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2409
This article is cited by
-
Ultra-thin light-weight laser-induced-graphene (LIG) diffractive optics
Light: Science & Applications (2023)
-
Skin pore imaging using spectral-domain optical coherence tomography: a case report
Biomedical Engineering Letters (2023)
-
Metasurface-based bijective illumination collection imaging provides high-resolution tomography in three dimensions
Nature Photonics (2022)
-
Excess comorbidities in gout: the causal paradigm and pleiotropic approaches to care
Nature Reviews Rheumatology (2022)
-
Imaging Technologies for Microfluidic Biochips
BioChip Journal (2022)