Abstract
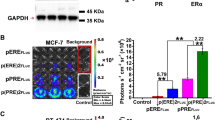
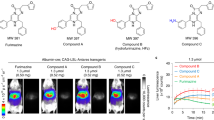
Molecular-genetic imaging is advancing from a valuable preclinical tool to a guide for patient management. The strategy involves pairing an imaging reporter gene with a complementary imaging agent in a system that can be used to measure gene expression or protein interaction or track gene-tagged cells in vivo. Tissue-specific promoters can be used to delineate gene expression in certain tissues, particularly when coupled with an appropriate amplification mechanism. Here we show that the progression elevated gene-3 (PEG-3) promoter, derived from a rodent gene mediating tumor progression and metastatic phenotypes, can be used to drive imaging reporters selectively to enable detection of micrometastatic disease in mouse models of human melanoma and breast cancer using bioluminescence and radionuclide-based molecular imaging techniques. Because of its strong promoter activity, tumor specificity and capacity for clinical translation, PEG-3 promoter–driven gene expression may represent a practical, new system for facilitating cancer imaging and therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Blasberg, R.G. & Tjuvajev, J.G. Molecular-genetic imaging: current and future perspectives. J. Clin. Invest. 111, 1620–1629 (2003).
Zhang, Y. et al. ABCG2/BCRP expression modulates D-luciferin based bioluminescence imaging. Cancer Res. 67, 9389–9397 (2007).
Uhrbom, L., Nerio, E. & Holland, E.C. Dissecting tumor maintenance requirements using bioluminescence imaging of cell proliferation in a mouse glioma model. Nat. Med. 10, 1257–1260 (2004).
Kishimoto, H. et al. In vivo imaging of lymph node metastasis with telomerase-specific replication-selective adenovirus. Nat. Med. 12, 1213–1219 (2006).
Padmanabhan, P. et al. Visualization of telomerase reverse transcriptase (hTERT) promoter activity using a trimodality fusion reporter construct. J. Nucl. Med. 47, 270–277 (2006).
Freytag, S.O. et al. Phase I trial of replication-competent adenovirus–mediated suicide gene therapy combined with IMRT for prostate cancer. Mol. Ther. 15, 1016–1023 (2007).
Yaghoubi, S.S. et al. Noninvasive detection of therapeutic cytolytic T cells with 18F-FHBG PET in a patient with glioma. Nat. Clin. Pract. Oncol. 6, 53–58 (2009).
Su, Z.Z., Shi, Y. & Fisher, P.B. Subtraction hybridization identifies a transformation progression-associated gene PEG-3 with sequence homology to a growth arrest and DNA damage–inducible gene. Proc. Natl. Acad. Sci. USA 94, 9125–9130 (1997).
Su, Z.Z. et al. Targeting gene expression selectively in cancer cells by using the progression-elevated gene-3 promoter. Proc. Natl. Acad. Sci. USA 102, 1059–1064 (2005).
Sarkar, D. et al. Eradication of therapy-resistant human prostate tumors using a cancer terminator virus. Cancer Res. 67, 5434–5442 (2007).
Sarkar, D. et al. Targeted virus replication plus immunotherapy eradicates primary and distant pancreatic tumors in nude mice. Cancer Res. 65, 9056–9063 (2005).
Sarkar, D. et al. A cancer terminator virus eradicates both primary and distant human melanomas. Cancer Gene Ther. 15, 293–302 (2008).
Su, Z., Shi, Y. & Fisher, P.B. Cooperation between AP1 and PEA3 sites within the progression elevated gene-3 (PEG-3) promoter regulate basal and differential expression of PEG-3 during progression of the oncogenic phenotype in transformed rat embryo cells. Oncogene 19, 3411–3421 (2000).
Sarkar, D. et al. Dual cancer-specific targeting strategy cures primary and distant breast carcinomas in nude mice. Proc. Natl. Acad. Sci. USA 102, 14034–14039 (2005).
Wood, M. et al. Biodistribution of an adenoviral vector carrying the luciferase reporter gene following intravesical or intravenous administration to a mouse. Cancer Gene Ther. 6, 367–372 (1999).
Peng, K.W. et al. Organ distribution of gene expression after intravenous infusion of targeted and untargeted lentiviral vectors. Gene Ther. 8, 1456–1463 (2001).
Evans, K.D., Tulloss, T.A. & Hall, N. 18FDG uptake in brown fat: potential for false positives. Radiol. Technol. 78, 361–366 (2007).
Shreve, P.D., Anzai, Y. & Wahl, R.L. Pitfalls in oncologic diagnosis with FDG PET imaging: physiologic and benign variants. Radiographics 19, 61–77, quiz 150–151 (1999).
Ray, S. et al. Noninvasive imaging of therapeutic gene expression using a bidirectional transcriptional amplification strategy. Mol. Ther. 16, 1848–1856 (2008).
Qiao, J. et al. Tumor-specific transcriptional targeting of suicide gene therapy. Gene Ther. 9, 168–175 (2002).
Iyer, M. et al. Two-step transcriptional amplification as a method for imaging reporter gene expression using weak promoters. Proc. Natl. Acad. Sci. USA 98, 14595–14600 (2001).
Sadowski, I., Ma, J., Triezenberg, S. & Ptashne, M. GAL4–VP16 is an unusually potent transcriptional activator. Nature 335, 563–564 (1988).
Burton, J.B. et al. Adenovirus-mediated gene expression imaging to directly detect sentinel lymph node metastasis of prostate cancer. Nat. Med. 14, 882–888 (2008).
Iyer, M. et al. Noninvasive imaging of enhanced prostate-specific gene expression using a two-step transcriptional amplification-based lentivirus vector. Mol. Ther. 10, 545–552 (2004).
Huyn, S.T. et al. A potent, imaging adenoviral vector driven by the cancer-selective mucin-1 promoter that targets breast cancer metastasis. Clin. Cancer Res. 15, 3126–3134 (2009).
Jacobs, A. et al. Positron-emission tomography of vector-mediated gene expression in gene therapy for gliomas. Lancet 358, 727–729 (2001).
Immonen, A. et al. AdvHSV-tk gene therapy with intravenous ganciclovir improves survival in human malignant glioma: a randomised, controlled study. Mol. Ther. 10, 967–972 (2004).
Klatzmann, D. et al. A phase I/II study of herpes simplex virus type 1 thymidine kinase “suicide” gene therapy for recurrent glioblastoma. Study Group on Gene Therapy for Glioblastoma. Hum. Gene Ther. 9, 2595–2604 (1998).
Trask, T.W. et al. Phase I study of adenoviral delivery of the HSV-tk gene and ganciclovir administration in patients with current malignant brain tumors. Mol. Ther. 1, 195–203 (2000).
Bonnet, M.E., Erbacher, P. & Bolcato-Bellemin, A.L. Systemic delivery of DNA or siRNA mediated by linear polyethylenimine (L-PEI) does not induce an inflammatory response. Pharm. Res. 25, 2972–2982 (2008).
Coelho-Castelo, A.A. et al. Tissue distribution of a plasmid DNA encoding Hsp65 gene is dependent on the dose administered through intramuscular delivery. Genet. Vaccines Ther. 4, 1 (2006).
Kang, K.K. et al. Safety evaluation of GX-12, a new HIV therapeutic vaccine: investigation of integration into the host genome and expression in the reproductive organs. Intervirology 46, 270–276 (2003).
Manam, S. et al. Plasmid DNA vaccines: tissue distribution and effects of DNA sequence, adjuvants and delivery method on integration into host DNA. Intervirology 43, 273–281 (2000).
Ramirez, K. et al. Preclinical safety and biodistribution of Sindbis virus measles DNA vaccines administered as a single dose or followed by live attenuated measles vaccine in a heterologous prime-boost regimen. Hum. Gene Ther. 19, 522–531 (2008).
Dwyer, R.M., Bergert, E.R., O'Connor, M.K., Gendler, S.J. & Morris, J.C. In vivo radioiodide imaging and treatment of breast cancer xenografts after MUC1-driven expression of the sodium iodide symporter. Clin. Cancer Res. 11, 1483–1489 (2005).
Tsuruta, Y. et al. A fiber-modified mesothelin promoter-based conditionally replicating adenovirus for treatment of ovarian cancer. Clin. Cancer Res. 14, 3582–3588 (2008).
Su, Z.Z. et al. Potential molecular mechanism for rodent tumorigenesis: mutational generation of Progression Elevated Gene-3 (PEG-3). Oncogene 24, 2247–2255 (2005).
Loening, A.M. & Gambhir, S.S. AMIDE: a free software tool for multimodality medical image analysis. Mol. Imaging 2, 131–137 (2003).
Acknowledgements
We appreciate the technical support provided by S. Dhara, S. Nimmagadda, M. Goggins and M. Griffith. We are grateful to J. Fox and G. Green for providing [125I]FIAU and assisting in the SPECT-CT and PET-CT imaging studies. We also thank C. Endres, B. Tsui, J. Yu and J. Fox for help with SPECT and PET data processing. The MDA-MB-231 cell line and pCMV-Tri construct were generous gifts from Z. Bhujwalla (Johns Hopkins University) and S. Gambhir (Stanford University), respectively. Funding was provided by US National Institutes of Health grant U24 CA92871 (to M.G.P.), by the Predoctoral Molecular Imaging Scholar Program from the Society of Nuclear Medicine and the Korea Science and Engineering Foundation Fellowship Program (to H.C.B.) and by the US National Institutes of Health grant P01 CA104177 and the US National Foundation for Cancer Research (to P.B.F.). P.B.F. holds the Thelma Newmeyer Corman Chair in Cancer Research at the Virginia Commonwealth University Massey Cancer Center.
Author information
Authors and Affiliations
Contributions
H.-e.C.B. designed and performed the experiments, analyzed data and prepared the manuscript. K.L.G. provided technical support in histopathology. J.L., K.L.G. and P.B.F. gave conceptual advice and edited the manuscript. M.G.P. and P.B.F. conceived of the project. M.G.P. supervised the project and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Figures 1–8 (PDF 1603 kb)
Supplementary Video 1
Detection of metastatic melanoma after systemic administration of pPEG-HSV1tk/PEI polyplex using SPECT-CT. Movie of a representative melanoma metastasis model, Mel-2 from Figure 4b and Figure 5a,b. The image was acquired at 24 h post injection of [125I]FIAU (1.4 mCi), which was 70 h after the pPEG-HSV1tk delivery. Multiple metastatic sites are predicted in the lung and upper dorsal area of the animal by [125I]FIAU SPECT. Melanoma masses were confirmed under the brown adipose tissue in the corresponding area (Fig. 5b) as well as in its lung by the gross pathological analysis. (MOV 3009 kb)
Supplementary Video 2
Movie of a representative control animal, Ctrl-3 from Figure 4a. This whole body SPECT-CT image was acquired at 24 h post injection of 1.4 mCi of [125I]FIAU, which was 70 h after the IV injection of pPEG-HSV1tk/PEI polyplex. Accumulated radioactivity was only detected in the urinary bladder and intestines of the animal. The same pseudo color scale used for Mel-2 in Supplementary Video 1 was applied. (MOV 2835 kb)
Rights and permissions
About this article
Cite this article
Bhang, He., Gabrielson, K., Laterra, J. et al. Tumor-specific imaging through progression elevated gene-3 promoter-driven gene expression. Nat Med 17, 123–129 (2011). https://doi.org/10.1038/nm.2269
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2269
This article is cited by
-
MRI detection of the malignant transformation of stem cells through reporter gene expression driven by a tumor-specific promoter
Stem Cell Research & Therapy (2021)
-
Synthetic biomarkers: a twenty-first century path to early cancer detection
Nature Reviews Cancer (2021)
-
Nanoparticle-mediated tumor cell expression of mIL-12 via systemic gene delivery treats syngeneic models of murine lung cancers
Scientific Reports (2021)
-
Molecular Bio-Imaging Probe for Non-Invasive Differentiation Between Human Leiomyoma Versus Leiomyosarcoma
Reproductive Sciences (2020)
-
A novel approach for assessment of prostate cancer aggressiveness using survivin-driven tumour-activatable minicircles
Gene Therapy (2019)