Abstract
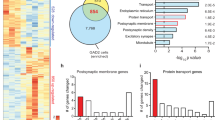
The lifetime prevalence (∼16%)1 and the economic burden ($100 billion annually)2,3 associated with major depressive disorder (MDD) make it one of the most common and debilitating neurobiological illnesses. To date, the exact cellular and molecular mechanisms underlying the pathophysiology of MDD have not been identified. Here we use whole-genome expression profiling of postmortem tissue and show significantly increased expression of mitogen-activated protein kinase (MAPK) phosphatase-1 (MKP-1, encoded by DUSP1, but hereafter called MKP-1) in the hippocampal subfields of subjects with MDD compared to matched controls. MKP-1, also known as dual-specificity phosphatase-1 (DUSP1), is a member of a family of proteins that dephosphorylate both threonine and tyrosine residues and thereby serves as a key negative regulator of the MAPK cascade4, a major signaling pathway involved in neuronal plasticity, function and survival5,6. We tested the role of altered MKP-1 expression in rat and mouse models of depression and found that increased hippocampal MKP-1 expression, as a result of stress or viral-mediated gene transfer, causes depressive behaviors. Conversely, chronic antidepressant treatment normalizes stress-induced MKP-1 expression and behavior, and mice lacking MKP-1 are resilient to stress. These postmortem and preclinical studies identify MKP-1 as a key factor in MDD pathophysiology and as a new target for therapeutic interventions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Kessler, R.C. et al. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). J. Am. Med. Assoc. 289, 3095–3105 (2003).
Greenberg, P.E. et al. The economic burden of depression in the United States: how did it change between 1990 and 2000? J. Clin. Psychiatry 64, 1465–1475 (2003).
Simon, G.E. Social and economic burden of mood disorders. Biol. Psychiatry 54, 208–215 (2003).
Jeffrey, K.L., Camps, M., Rommel, C. & Mackay, C.R. Targeting dual-specificity phosphatases: manipulating MAP kinase signalling and immune responses. Nat. Rev. Drug Discov. 6, 391–403 (2007).
Grewal, S.S., York, R.D. & Stork, P.J. Extracellular signal–regulated kinase signalling in neurons. Curr. Opin. Neurobiol. 9, 544–553 (1999).
Fukunaga, K. & Miyamoto, E. Role of MAP kinase in neurons. Mol. Neurobiol. 16, 79–95 (1998).
Sheline, Y.I., Wang, P.W., Gado, M.H., Csernansky, J.G. & Vannier, M.W. Hippocampal atrophy in recurrent major depression. Proc. Natl. Acad. Sci. USA 93, 3908–3913 (1996).
Sheline, Y.I., Gado, M.H. & Kraemer, H.C. Untreated depression and hippocampal volume loss. Am. J. Psychiatry 160, 1516–1518 (2003).
Neumeister, A. et al. Reduced hippocampal volume in unmedicated, remitted patients with major depression versus control subjects. Biol. Psychiatry 57, 935–937 (2005).
Stockmeier, C.A. et al. Cellular changes in the postmortem hippocampus in major depression. Biol. Psychiatry 56, 640–650 (2004).
Watanabe, Y., Gould, E. & McEwen, B.S. Stress induces atrophy of apical dendrites of hippocampal CA3 pyramidal neurons. Brain Res. 588, 341–345 (1992).
Magariños, A.M., Deslandes, A. & McEwen, B.S. Effects of antidepressants and benzodiazepine treatments on the dendritic structure of CA3 pyramidal neurons after chronic stress. Eur. J. Pharmacol. 371, 113–122 (1999).
Schmidt, H.D. & Duman, R.S. The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav. Pharmacol. 18, 391–418 (2007).
Drevets, W.C. Functional anatomical abnormalities in limbic and prefrontal cortical structures in major depression. Prog. Brain Res. 126, 413–431 (2000).
Drevets, W.C. Neuroimaging and neuropathological studies of depression: implications for the cognitive-emotional features of mood disorders. Curr. Opin. Neurobiol. 11, 240–249 (2001).
Verkhratsky, N.S. Limbic control of endocrine glands in aged rats. Exp. Gerontol. 30, 415–421 (1995).
Sapolsky, R.M. Glucocorticoids and hippocampal atrophy in neuropsychiatric disorders. Arch. Gen. Psychiatry 57, 925–935 (2000).
McEwen, B.S. Stress and hippocampal plasticity. Annu. Rev. Neurosci. 22, 105–122 (1999).
Dwivedi, Y. et al. Reduced activation and expression of ERK1/2 MAP kinase in the post-mortem brain of depressed suicide subjects. J. Neurochem. 77, 916–928 (2001).
Dwivedi, Y., Rizavi, H.S., Conley, R.R. & Pandey, G.N. ERK MAP kinase signaling in post-mortem brain of suicide subjects: differential regulation of upstream Raf kinases Raf-1 and B-Raf. Mol. Psychiatry 11, 86–98 (2006).
Hsiung, S.C. et al. Attenuated 5-HT1A receptor signaling in brains of suicide victims: involvement of adenylyl cyclase, phosphatidylinositol 3-kinase, Akt and mitogen-activated protein kinase. J. Neurochem. 87, 182–194 (2003).
Dwivedi, Y. et al. Altered gene expression of brain-derived neurotrophic factor and receptor tyrosine kinase B in postmortem brain of suicide subjects. Arch. Gen. Psychiatry 60, 804–815 (2003).
Dwivedi, Y., Mondal, A.C., Rizavi, H.S. & Conley, R.R. Suicide brain is associated with decreased expression of neurotrophins. Biol. Psychiatry 58, 315–324 (2005).
Chen, B., Dowlatshahi, D., MacQueen, G.M., Wang, J.F. & Young, L.T. Increased hippocampal BDNF immunoreactivity in subjects treated with antidepressant medication. Biol. Psychiatry 50, 260–265 (2001).
Karege, F., Vaudan, G., Schwald, M., Perroud, N. & La Harpe, R. Neurotrophin levels in postmortem brains of suicide victims and the effects of antemortem diagnosis and psychotropic drugs. Brain Res. Mol. Brain Res. 136, 29–37 (2005).
Willner, P. Chronic mild stress (CMS) revisited: consistency and behavioural-neurobiological concordance in the effects of CMS. Neuropsychobiology 52, 90–110 (2005).
Banasr, M. et al. Glial pathology in an animal model of depression: reversal of stress-induced cellular, metabolic and behavioral deficits by the glutamate-modulating drug riluzole. Mol. Psychiatry 15, 501–511 (2008).
Banasr, M. & Duman, R.S. Glial loss in the prefrontal cortex is sufficient to induce depressive-like behaviors. Biol. Psychiatry 64, 863–870 (2008).
Swaab, D.F., Bao, A.M. & Lucassen, P.J. The stress system in the human brain in depression and neurodegeneration. Ageing Res. Rev. 4, 141–194 (2005).
Gold, P.W. & Chrousos, G.P. Organization of the stress system and its dysregulation in melancholic and atypical depression: high vs low CRH/NE states. Mol. Psychiatry 7, 254–275 (2002).
Keyse, S.M. & Emslie, E.A. Oxidative stress and heat shock induce a human gene encoding a protein-tyrosine phosphatase. Nature 359, 644–647 (1992).
Laderoute, K.R. et al. Mitogen-activated protein kinase phosphatase-1 (MKP-1) expression is induced by low oxygen conditions found in solid tumor microenvironments. A candidate MKP for the inactivation of hypoxia-inducible stress-activated protein kinase/c-Jun N-terminal protein kinase activity. J. Biol. Chem. 274, 12890–12897 (1999).
Seta, K.A., Kim, R., Kim, H.W., Millhorn, D.E. & Beitner-Johnson, D. Hypoxia-induced regulation of MAPK phosphatase-1 as identified by subtractive suppression hybridization and cDNA microarray analysis. J. Biol. Chem. 276, 44405–44412 (2001).
Dorfman, K. et al. Disruption of the erp/mkp-1 gene does not affect mouse development: normal MAP kinase activity in ERP/MKP-1–deficient fibroblasts. Oncogene 13, 925–931 (1996).
Wu, J.J. et al. Mice lacking MAP kinase phosphatase-1 have enhanced MAP kinase activity and resistance to diet-induced obesity. Cell Metab. 4, 61–73 (2006).
Duman, C.H., Schlesinger, L., Kodama, M., Russell, D.S. & Duman, R.S. A role for MAP kinase signaling in behavioral models of depression and antidepressant treatment. Biol. Psychiatry 61, 661–670 (2007).
Qi, X. et al. A role for the extracellular signal–regulated kinase signal pathway in depressive-like behavior. Behav. Brain Res. 199, 203–209 (2009).
Einat, H. et al. The role of the extracellular signal–regulated kinase signaling pathway in mood modulation. J. Neurosci. 23, 7311–7316 (2003).
Engel, S.R. et al. The extracellular signal–regulated kinase pathway contributes to the control of behavioral excitement. Mol. Psychiatry 14, 448–461 (2009).
Creson, T.K. et al. The anterior cingulate ERK pathway contributes to regulation of behavioral excitement and hedonic activity. Bipolar Disord. 11, 339–350 (2009).
Acknowledgements
This work is supported by US Public Health Service grants MH45481 (R.S.D.), 2 P01 MH25642 (R.S.D.), MH67996 (C.A.S.) and P20 RR17701 (C.A.S.) and the Connecticut Mental Health Center (R.S.D.). We acknowledge the invaluable contributions made by the families consenting to donate brain tissue and be interviewed. We also thank the Cuyahoga County Coroner and staff for their willing assistance. We are grateful for the contributions of J. Overholser, G. Jurjus, H. Meltzer, L. Konick, L. Dieter, N. Herbst, G. Mahajan, H. Kooiman and J. Cobb in the psychiatric assessment and human tissue dissection and preparation. We also thank R. Terwilliger and H.J. Kang for technical assistance with gene expression analysis, A. Bennett (Yale University School of Medicine) for Mkp-1−/− mice, X.Y. Li for assistance in breeding and genotyping Mkp-1−/− mice and R. DiLeone (Yale University School of Medicine) for generously donating AAV-CMV-GFP plasmid used to generate recombinant AAVs.
Author information
Authors and Affiliations
Contributions
V.D. prepared the original draft of the manuscript and was involved in all aspects of the experimental design and research, including execution of all microarray and molecular experiments, as well as behavioral tests in rats and mice. M.B. conducted the behavioral aspects of the rat CUS study, assisted with rat surgeries and was involved in analysis and interpretation of behavioral tests. P.L. was responsible for optimization, construction and preparation of recombinant AAVs. H.D.S. conducted baseline behavior tests in Mkp-1−/− mice. C.A.S. was responsible for human tissue generation and preparation of relevant human subjects' information tables and methodology. A.A.S. conducted statistical analysis of microarray experiments. S.S.N. assisted in the development and optimization of microarray experiments. R.S.D. was involved in all aspects of study design, data analysis, interpretation of results and preparation of the manuscript and figures. All authors discussed the results presented in the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Tables 1–6 and Supplementary Methods (PDF 1425 kb)
Supplementary Data 1
Dysregulated genes in dentate gyrus (XLS 1655 kb)
Supplementary Data 2
Dysregulated genes in CA1 (XLS 504 kb)
Rights and permissions
About this article
Cite this article
Duric, V., Banasr, M., Licznerski, P. et al. A negative regulator of MAP kinase causes depressive behavior. Nat Med 16, 1328–1332 (2010). https://doi.org/10.1038/nm.2219
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2219
This article is cited by
-
Differential expression of Dusp1 and immediate early response genes in the hippocampus of rats, subjected to forced swim test
Scientific Reports (2023)
-
LHPP, a risk factor for major depressive disorder, regulates stress-induced depression-like behaviors through its histidine phosphatase activity
Molecular Psychiatry (2023)
-
Identification of mitogen-activated protein kinase phosphatase-1 (MKP-1) protein partners using tandem affinity purification and mass spectrometry
Pharmacological Reports (2023)
-
DUSP1/MKP-1 represents another piece in the P2X7R intracellular signaling puzzle in cerebellar cells: our last journey with Mª Teresa along the purinergic pathways of Eden
Purinergic Signalling (2023)
-
RNA-seq based transcriptome analysis of ethanol extract of saffron protective effect against corticosterone-induced PC12 cell injury
BMC Complementary Medicine and Therapies (2022)