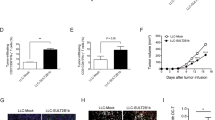
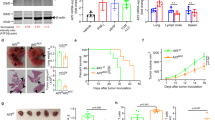
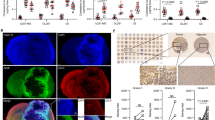
Abstract
Sterol metabolism has recently been linked to innate and adaptive immune responses through liver X receptor (LXR) signaling. Whether products of sterol metabolism interfere with antitumor responses is currently unknown. Dendritic cells (DCs) initiate immune responses, including antitumor activity after their CC chemokine receptor-7 (CCR7)-dependent migration to lymphoid organs. Here we report that human and mouse tumors produce LXR ligands that inhibit CCR7 expression on maturing DCs and, therefore, their migration to lymphoid organs. In agreement with this observation, we detected CD83+CCR7− DCs within human tumors. Mice injected with tumors expressing the LXR ligand–inactivating enzyme sulfotransferase 2B1b (SULT2B1b) successfully controlled tumor growth by regaining DC migration to tumor-draining lymph nodes and by developing overt inflammation within tumors. The control of tumor growth was also observed in chimeric mice transplanted with bone marrow from mice lacking the gene encoding LXR-α (Nr1h3−/− mice) Thus, we show a new mechanism of tumor immunoescape involving products of cholesterol metabolism. The manipulation of this pathway could restore antitumor immunity in individuals with cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rabinovich, G.A., Gabrilovich, D. & Sotomayor, E.M. Immunosuppressive strategies that are mediated by tumor cells. Annu. Rev. Immunol. 25, 267–296 (2007).
Munn, D.H. & Mellor, A.L. Indoleamine 2,3-dioxygenase and tumor-induced tolerance. J. Clin. Invest. 117, 1147–1154 (2007).
Bronte, V. & Zanovello, P. Regulation of immune responses by l-arginine metabolism. Nat. Rev. Immunol. 5, 641–654 (2005).
Peet, D.J., Janowski, B.A. & Mangelsdorf, D.J. The LXRs: a new class of oxysterol receptors. Curr. Opin. Genet. Dev. 8, 571–575 (1998).
Janowski, B.A. et al. Structural requirements of ligands for the oxysterol liver X receptors LXRα and LXRβ. Proc. Natl. Acad. Sci. USA 96, 266–271 (1999).
Yang, X., Lamia, K.A. & Evans, R.M. Nuclear receptors, metabolism and the circadian clock. Cold Spring Harb. Symp. Quant. Biol. 72, 387–394 (2007).
Repa, J.J. & Mangelsdorf, D.J. The role of orphan nuclear receptors in the regulation of cholesterol homeostasis. Annu. Rev. Cell Dev. Biol. 16, 459–481 (2000).
Bensinger, S.J. & Tontonoz, P. Integration of metabolism and inflammation by lipid-activated nuclear receptors. Nature 454, 470–477 (2008).
Banchereau, J. & Steinman, R.M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Jung, S. et al. In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002).
Randolph, G.J., Ochando, J. & Partida-Sanchez, S. Migration of dendritic cell subsets and their precursors. Annu. Rev. Immunol. 26, 293–316 (2008).
Martín-Fontecha, A. et al. Regulation of dendritic cell migration to the draining lymph node: impact on T lymphocyte traffic and priming. J. Exp. Med. 198, 615–621 (2003).
Ohl, L. et al. CCR7 governs skin dendritic cell migration under inflammatory and steady-state conditions. Immunity 21, 279–288 (2004).
Villablanca, E.J. et al. Selected natural and synthetic retinoids impair CCR7- and CXCR4-dependent cell migration in vitro and in vivo. J. Leukoc. Biol. 84, 871–879 (2008).
Allavena, P. et al. The chemokine receptor switch paradigm and dendritic cell migration: its significance in tumor tissues. Immunol. Rev. 177, 141–149 (2000).
Sallusto, F. & Lanzavecchia, A. Understanding dendritic cell and T-lymphocyte traffic through the analysis of chemokine receptor expression. Immunol. Rev. 177, 134–140 (2000).
Nencioni, A. et al. Dendritic cell immunogenicity is regulated by peroxisome proliferator-activated receptor γ. J. Immunol. 169, 1228–1235 (2002).
Geyeregger, R. et al. Liver X receptors regulate dendritic cell phenotype and function through blocked induction of the actin-bundling protein fascin. Blood 109, 4288–4295 (2007).
Bergstrom, J.D. et al. Zaragozic acids: a family of fungal metabolites that are picomolar competitive inhibitors of squalene synthase. Proc. Natl. Acad. Sci. USA 90, 80–84 (1993).
Shimizu, C., Fuda, H., Yanai, H. & Strott, C.A. Conservation of the hydroxysteroid sulfotransferase SULT2B1 gene structure in the mouse: pre- and postnatal expression, kinetic analysis of isoforms, and comparison with prototypical SULT2A1. Endocrinology 144, 1186–1193 (2003).
Fuda, H., Javitt, N.B., Mitamura, K., Ikegawa, S. & Strott, C.A. Oxysterols are substrates for cholesterol sulfotransferase. J. Lipid Res. 48, 1343–1352 (2007).
Chen, W., Chen, G., Head, D.L., Mangelsdorf, D.J. & Russell, D.W. Enzymatic reduction of oxysterols impairs LXR signaling in cultured cells and the livers of mice. Cell Metab. 5, 73–79 (2007).
Mavilio, F. et al. Peripheral blood lymphocytes as target cells of retroviral vector-mediated gene transfer. Blood 83, 1988–1997 (1994).
Hogquist, K.A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
He, D. & Falany, C.N. Characterization of proline-serine–rich carboxyl terminus in human sulfotransferase 2B1b: immunogenicity, subcellular localization, kinetic properties and phosphorylation. Drug Metab. Dispos. 34, 1749–1755 (2006).
Sutmuller, R.P. et al. Synergism of cytotoxic T lymphocyte–associated antigen 4 blockade and depletion of CD25+ regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J. Exp. Med. 194, 823–832 (2001).
Degl'Innocenti, E. et al. Peripheral T-cell tolerance associated with prostate cancer is independent from CD4+CD25+ regulatory T cells. Cancer Res. 68, 292–300 (2008).
Alberti, S. et al. Hepatic cholesterol metabolism and resistance to dietary cholesterol in LXRβ-deficient mice. J. Clin. Invest. 107, 565–573 (2001).
Joseph, S.B. et al. LXR-dependent gene expression is important for macrophage survival and the innate immune response. Cell 119, 299–309 (2004).
Gabrilovich, D. Mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat. Rev. Immunol. 4, 941–952 (2004).
Huang, Q. et al. The plasticity of dendritic cell responses to pathogens and their components. Science 294, 870–875 (2001).
Le Naour, F. et al. Profiling changes in gene expression during differentiation and maturation of monocyte-derived dendritic cells using both oligonucleotide microarrays and proteomics. J. Biol. Chem. 276, 17920–17931 (2001).
Randolph, G.J. Emigration of monocyte-derived cells to lymph nodes during resolution of inflammation and its failure in atherosclerosis. Curr. Opin. Lipidol. 19, 462–468 (2008).
Bensinger, S.J. et al. LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 134, 97–111 (2008).
Greenwood, J., Steinman, L. & Zamvil, S.S. Statin therapy and autoimmune disease: from protein prenylation to immunomodulation. Nat. Rev. Immunol. 6, 358–370 (2006).
Acknowledgements
We thank M.E. Bianchi and G. Parmiani for helpful discussions and for the critical review of this manuscript. Additionally, we thank D. Mangelsdorf, Howard Hughes Medical Institute, University of Texas Southwestern Medical Center, for providing us with GAL4-LXRs plasmids; M. Lidén, Ludwig Institute for Cancer Research, Stockholm, for GAL4-RXR and TK-MHC100-luc plasmids; F. Nystrom, Linköping University, for GAL4–PPAR-γ plasmid; C. Strott, National Institutes of Health, for the mouse SULT2B1b–encoding plasmid; L. Naldini, Scientific Institute S.Raffaele, for the lentiviral vector hPGK.GFP.wPRE.mhCMV.ΔNGFr.SV40PA; and M. Rocchi and C. Lanterna for technical help. This work was supported by Fondazione Cariplo, Italian Association for Cancer Research, European Community Project 'Cancer Immunotherapy' LSHC-CT-2006-518334 and the Italian Ministry of Health.
Author information
Authors and Affiliations
Contributions
E.J.V., L.R., D.Z., R.F., D.M., A.N., B.V., A.P. and V.R. performed in vitro and in vivo experiments. F.S., M.P. and C.D. analyzed and interpreted the immunohistochemistry and histology experiments. K.R.S. and J.-A.G. provided knockout mice and gave scientific advice. S.S., M.B. and C.B. provided a key contribution for the development of the project by discussing some aspects of the experimental strategies. E.J.V., C.T. and V.R. prepared the figures and drafted the manuscript. C.T. and V.R. contributed equally to the design of the experiments and to the writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.-A.G. is a shareholder and consultant for KaroBio AB.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Table 1 and Supplementary Methods (PDF 1551 kb)
Rights and permissions
About this article
Cite this article
Villablanca, E., Raccosta, L., Zhou, D. et al. Tumor-mediated liver X receptor-α activation inhibits CC chemokine receptor-7 expression on dendritic cells and dampens antitumor responses. Nat Med 16, 98–105 (2010). https://doi.org/10.1038/nm.2074
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2074
This article is cited by
-
Implications of metabolism-driven myeloid dysfunctions in cancer therapy
Cellular & Molecular Immunology (2021)
-
Liver X receptors regulate natural killer T cell population and antitumor activity in the liver of mice
Scientific Reports (2021)
-
Liver X receptor regulates Th17 and RORγt+ Treg cells by distinct mechanisms
Mucosal Immunology (2021)
-
Targeting the alternative bile acid synthetic pathway for metabolic diseases
Protein & Cell (2021)
-
Dendritic cells in cancer immunology and immunotherapy
Nature Reviews Immunology (2020)