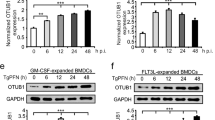
Abstract
Nucleotide-binding oligomerization domain–containing-2 (NOD2) acts as a bacterial sensor in dendritic cells (DCs), but it is not clear how bacterial recognition links with antigen presentation after NOD2 stimulation. NOD2 variants are associated with Crohn's disease, where breakdown in self-recognition of commensal bacteria leads to gastrointestinal inflammation. Here we show NOD2 triggering by muramyldipeptide induces autophagy in DCs. This effect requires receptor-interacting serine-threonine kinase-2 (RIPK-2), autophagy-related protein-5 (ATG5), ATG7 and ATG16L1 but not NLR family, pyrin domain containing-3 (NALP3).We show that NOD2-mediated autophagy is required for both bacterial handling and generation of major histocompatibility complex (MHC) class II antigen-specific CD4+ T cell responses in DCs. DCs from individuals with Crohn's disease expressing Crohn's disease—associated NOD2 or ATG16L1 risk variants are defective in autophagy induction, bacterial trafficking and antigen presentation. Our findings link two Crohn's disease–associated susceptibility genes in a single functional pathway and reveal defects in this pathway in Crohn's disease DCs that could lead to bacterial persistence via impaired lysosomal destruction and immune mediated clearance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 411, 603–606 (2001).
Hugot, J.P. et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature 411, 599–603 (2001).
Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Blander, J.M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Xu, Y. et al. Toll-like receptor 4 is a sensor for autophagy associated with innate immunity. Immunity 27, 135–144 (2007).
Delgado, M.A., Elmaoued, R.A., Davis, A.S., Kyei, G. & Deretic, V. Toll-like receptors control autophagy. EMBO J. 27, 1110–1121 (2008).
Levine, B. & Kroemer, G. Autophagy in the pathogenesis of disease. Cell 132, 27–42 (2008).
Schmid, D., Pypaert, M. & Münz, C. Antigen-loading compartments for major histocompatibility complex class II molecules continuously receive input from autophagosomes. Immunity 26, 79–92 (2007).
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nat. Genet. 39, 207–211 (2007).
Rioux, J.D. et al. Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nat. Genet. 39, 596–604 (2007).
Prescott, N.J. et al. A nonsynonymous SNP in ATG16L1 predisposes to ileal Crohn's disease and is independent of CARD15 and IBD5. Gastroenterology 132, 1665–1671 (2007).
Cummings, J.R. et al. Confirmation of the role of ATG16L1 as a Crohn's disease susceptibility gene. Inflamm. Bowel Dis. 13, 941–946 (2007).
Parkes, M. et al. Sequence variants in the autophagy gene IRGM and multiple other replicating loci contribute to Crohn's disease susceptibility. Nat. Genet. 39, 830–832 (2007).
Ichimura, Y. et al. A ubiquitin-like system mediates protein lipidation. Nature 408, 488–492 (2000).
Kabeya, Y. et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 19, 5720–5728 (2000).
Pétrilli, V., Dostert, C., Muruve, D.A. & Tschopp, J. The inflammasome: a danger sensing complex triggering innate immunity. Curr. Opin. Immunol. 19, 615–622 (2007).
Park, J.H. et al. RICK/RIP2 mediates innate immune responses induced through Nod1 and Nod2 but not TLRs. J. Immunol. 178, 2380–2386 (2007).
Yang, Y. et al. NOD2 pathway activation by MDP or Mycobacterium tuberculosis infection involves the stable polyubiquitination of Rip2. J. Biol. Chem. 282, 36223–36229 (2007).
Dengjel, J. et al. Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proc. Natl. Acad. Sci. USA 102, 7922–7927 (2005).
Nimmerjahn, F. et al. Major histocompatibility complex class II–restricted presentation of a cytosolic antigen by autophagy. Eur. J. Immunol. 33, 1250–1259 (2003).
Magalhaes, J.G. et al. Nod2-dependent TH2 polarization of antigen-specific immunity. J. Immunol. 181, 7925–7935 (2008).
Birmingham, C.L., Smith, A.C., Bakowski, M.A., Yoshimori, T. & Brumell, J.H. Autophagy controls Salmonella infection in response to damage to the Salmonella-containing vacuole. J. Biol. Chem. 281, 11374–11383 (2006).
Darfeuille-Michaud, A. Adherent-invasive Escherichia coli: a putative new E. coli pathotype associated with Crohn's disease. Int. J. Med. Microbiol. 292, 185–193 (2002).
Darfeuille-Michaud, A. et al. High prevalence of adherent-invasive Escherichia coli associated with ileal mucosa in Crohn's disease. Gastroenterology 127, 412–421 (2004).
Suzuki, T. et al. Differential regulation of caspase-1 activation, pyroptosis and autophagy via Ipaf and ASC in Shigella-infected macrophages. PLoS Pathog. 3, e111 (2007).
Acknowledgements
We would like to acknowledge the previous researchers in our department involved in genotyping of the Oxford Crohn's disease cohort and thank A.-K. Simon and A. McMichael for helpful discussions. Most particularly, we would like to thank all of our donors who generously gave blood samples for this study. We are supported by grants from the UK Medical Research Council (J.B., T.P. and A.S.), the UK Higher Education Funding Council (A.S.), Oxford National Institute for Health Research Biomedical Research Centre (A.S.) and the National Institute of Health Research Specialist Biomedical Research Centre in Microbial Disease (01CD1) (B.J.C.), Action Medical Research (R.C.), Digestive Disorders Foundation and National Association for Crohn's and Colitis and The Barbour Trust in Memory of Simon Ash (O.B.), Fondation Philippe Weiner Maurice Anspach (B.D.) and the Wellcome Trust (P.A.). The GFP-LC3 construct was a kind gift from H. Mellor (University of Oxford) GFP–S. enterica typhimurium was a kind gift from D. Holden (Imperial College, University of London) and SL5338 pTECH1 was a kind gift from K. Turner (Sanger Centre).
Author information
Authors and Affiliations
Contributions
A.S. conceived the idea; A.S., J.B. and R.C. designed the experiments and prepared the manuscript; R.C., J.B., O.B., P.A. and A.S. did RNAi, flow cytometry, immunoblot and confocal experiments. J.B., R.C., O.B. and P.A. were involved with gene sequencing of NOD2 and ATG16L1. T.P. and B.D. provided technical assistance and participated in flow cytometry experiments; D.J.P.F. did the electron microscopy analysis; B.J.C. provided GFP–E. coli, and B.J.C. and D.J. provided additional intellectual input.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2 and Supplementary Methods (PDF 771 kb)
Rights and permissions
About this article
Cite this article
Cooney, R., Baker, J., Brain, O. et al. NOD2 stimulation induces autophagy in dendritic cells influencing bacterial handling and antigen presentation. Nat Med 16, 90–97 (2010). https://doi.org/10.1038/nm.2069
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2069
This article is cited by
-
A probiotic bi-functional peptidoglycan hydrolase sheds NOD2 ligands to regulate gut homeostasis in female mice
Nature Communications (2023)
-
Advancing intestinal organoid technology to decipher nano-intestine interactions and treat intestinal disease
Nano Research (2023)
-
The role of diosgenin in crohn’s disease
Clinical Phytoscience (2022)
-
Hermansky-Pudlak syndrome type 1 causes impaired anti-microbial immunity and inflammation due to dysregulated immunometabolism
Mucosal Immunology (2022)
-
Hepatic NOD2 promotes hepatocarcinogenesis via a RIP2-mediated proinflammatory response and a novel nuclear autophagy-mediated DNA damage mechanism
Journal of Hematology & Oncology (2021)