Abstract
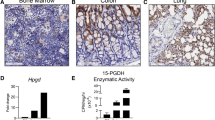
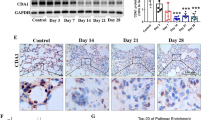
Idiopathic pulmonary fibrosis (IPF) is a progressive disease characterized by fibroblast proliferation and excess deposition of collagen and other extracellular matrix (ECM) proteins, which lead to distorted lung architecture and function1. Given that anti-inflammatory or immunosuppressive therapy currently used for IPF does not improve disease progression therapies targeted to blocking the mechanisms of fibrogenesis are needed1. Although transforming growth factor-β (TGF-β) functions are crucial in fibrosis2,3, antagonizing this pathway in bleomycin-induced pulmonary fibrosis, an animal model of IPF, does not prevent fibrosis completely4,5,6,7, indicating an additional pathway also has a key role in fibrogenesis. Given that the loss of cytosolic phospholipase A2 (cPLA2) suppresses bleomycin-induced pulmonary fibrosis8, we examined the roles of prostaglandins using mice lacking each prostoaglandin receptor9,10,11,12,13,14,15. Here we show that loss of prostaglandin F (PGF) receptor (FP) selectively attenuates pulmonary fibrosis while maintaining similar levels of alveolar inflammation and TGF-β stimulation as compared to wild-type (WT) mice, and that FP deficiency and inhibition of TGF-β signaling additively decrease fibrosis. Furthermore, PGF2α is abundant in bronchoalveolar lavage fluid (BALF) of subjects with IPF and stimulates proliferation and collagen production of lung fibroblasts via FP, independently of TGF-β. These findings show that PGF2α-FP signaling facilitates pulmonary fibrosis independently of TGF-β and suggests this signaling pathway as a therapeutic target for IPF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Selman, M., King, T.E. & Pardo, A. Idiopathic pulmonary fibrosis: prevailing and evolving hypotheses about its pathogenesis and implications for therapy. Ann. Intern. Med. 134, 136–151 (2001).
Border, W.A. & Noble, N.A. Transforming growth factor β in tissue fibrosis. N. Engl. J. Med. 331, 1286–1292 (1994).
Munger, J.S. et al. The integrin αvβ6 binds and activates latent TGFβ1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96, 319–328 (1999).
Giri, S.N., Hyde, D.M. & Hollinger, M.A. Effect of antibody to transforming growth factor β on bleomycin induced accumulation of lung collagen in mice. Thorax 48, 959–966 (1993).
Wang, Q. et al. Reduction of bleomycin induced lung fibrosis by transforming growth factor β soluble receptor in hamsters. Thorax 54, 805–812 (1999).
Nakao, A. et al. Transient gene transfer and expression of Smad7 prevents bleomycin-induced lung fibrosis in mice. J. Clin. Invest. 104, 5–11 (1999).
Kapoun, A.M. et al. Transforming growth factor-β receptor type 1 (TGFβRI) kinase activity but not p38 activation is required for TGFβRI-induced myofibroblast differentiation and profibrotic gene expression. Mol. Pharmacol. 70, 518–531 (2006).
Nagase, T. et al. A pivotal role of cytosolic phospholipase A2 in bleomycin-induced pulmonary fibrosis. Nat. Med. 8, 480–484 (2002).
Hizaki, H. et al. Abortive expansion of the cumulus and impaired fertility in mice lacking the prostaglandin E receptor subtype EP2. Proc. Natl. Acad. Sci. USA 96, 10501–10506 (1999).
Ushikubi, F. et al. Impaired febrile response in mice lacking the prostaglandin E receptor subtype EP3. Nature 395, 281–284 (1998).
Segi, E. et al. Patent ductus arteriosus and neonatal death in prostaglandin receptor EP4-deficient mice. Biochem. Biophys. Res. Commun. 246, 7–12 (1998).
Matsuoka, T. et al. Prostaglandin D2 as a mediator of allergic asthma. Science 287, 2013–2017 (2000).
Sugimoto, Y. et al. Failure of parturition in mice lacking the prostaglandin F receptor. Science 277, 681–683 (1997).
Murata, T. et al. Altered pain perception and inflammatory response in mice lacking prostacyclin receptor. Nature 388, 678–682 (1997).
Kabashima, K. et al. Thromboxane A2 modulates interaction of dendritic cells and T cells and regulates acquired immunity. Nat. Immunol. 4, 694–701 (2003).
Narumiya, S. Physiology and pathophysiology of prostanoid receptors. Proc. Jpn. Acad. Ser. B. 83, 296–319 (2007).
Kabashima, K. et al. The prostaglandin receptor EP4 suppresses colitis, mucosal damage and CD4 cell activation in the gut. J. Clin. Invest. 109, 883–893 (2002).
Kita, Y. et al. Pathway-oriented profiling of lipid mediators in macrophages. Biochem. Biophys. Res. Commun. 330, 898–906 (2005).
Yoshikawa, K., Kita, Y., Kishimoto, K. & Shimizu, T. Profiling of eicosanoid production in the rat hippocampus during kainic acid–induced seizure: dual phase regulation and differential involvement of COX-1 and COX-2. J. Biol. Chem. 281, 14663–14669 (2006).
Aono, Y. et al. Imatinib as a novel antifibrotic agent in bleomycin-induced pulmonary fibrosis in mice. Am. J. Respir. Crit. Care Med. 171, 1279–1285 (2005).
DaCosta Byfield, S., Major, C., Laping, N.J. & Roberts, A.B. SB-505124 is a selective inhibitor of transforming growth factor-β type I receptors ALK4, ALK5 and ALK7. Mol. Pharmacol. 65, 744–752 (2003).
Griffin, B.W., Klimko, P., Crider, J.Y. & Sharif, N.A. AL-8810: a novel prostaglandin F2α analog with selective antagonist effects at the prostaglandin F2α (FP) receptor. J. Pharmacol. Exp. Ther. 290, 1278–1284 (1999).
Jinnin, M. et al. Potential regulatory elements of the constitutive up-regulated α2I collagen gene in scleroderma dermal fibroblasts. Biochem. Biophys. Res. Commun. 343, 904–909 (2006).
Ihn, H. et al. Transcriptional regulation of the human α2I collagen gene. Combined action of upstream stimulatory and inhibitory cis-acting elements. J. Biol. Chem. 271, 26717–26723 (1996).
Ishizaki, T. et al. Pharmacological properties of Y-27632, a specific inhibitor of rho-associated kinases. Mol. Pharmacol. 57, 976–983 (2000).
Jinnin, M., Ihn, H. & Tamaki, K. Characterization of SIS3, a novel specific inhibitor of Smad3, and its effect on transforming growth factor-β1–induced extracellular matrix expression. Mol. Pharmacol. 69, 597–607 (2006).
Ley, K. & Zarbock, A. From injury to fibrosis. Nat. Med. 14, 20–21 (2008).
Bhatt, N. et al. Promising pharmacologic innovations in treating pulmonary fibrosis. Curr. Opin. Pharmacol. 6, 284–292 (2006).
Border, W.A. & Noble, N.A. Targeting TGF-β for treatment of disease. Nat. Med. 1, 1000–1001 (1995).
Wyss-Coray, T. et al. TGF-β1 promotes microglial amyloid-β clearance and reduces plaque burden in transgenic mice. Nat. Med. 7, 612–618 (2001).
Moore, B.B. et al. Bleomycin-induced E prostanoid receptor changes alter fibroblast responses to prostaglandin E2 . J. Immunol. 174, 5644–5649 (2005).
Lovgren, A.K. et al. COX-2–derived prostacyclin protects against bleomycin-induced pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 291, L144–L156 (2006).
Peters-Golden, M. et al. Protection from pulmonary fibrosis in leukotriene-deficient mice. Am. J. Respir. Crit. Care Med. 165, 229–235 (2002).
Beller, T.C. et al. Targeted gene disruption reveals the role of the cysteinyl leukotriene 2 receptor in increased vascular permeability and in bleomycin-induced pulmonary fibrosis in mice. J. Biol. Chem. 279, 46129–46134 (2004).
Tager, A.M. et al. The lysophosphatidic acid receptor LPA1 links pulmonary fibrosis to lung injury by mediating fibroblast recruitment and vascular leak. Nat. Med. 14, 45–54 (2008).
Handa, T. et al. Polymorphisms of B7 (CD80 and CD86) genes do not affect disease susceptibility to sarcoidosis. Respiration 72, 243–248 (2005).
Acknowledgements
We thank M. Trojanowska (Medical University of South Carolina) and H. Ihn (Kumamoto University) for the −3500COL1A2/CAT construct, and Ono Pharmaceutical Company for ONO-AE3-208. We also thank Y. Kobashi for BALF analysis, T. Fujiwara for animal care and T. Arai for assistance. This work was supported in part by a Grant-in-Aid for Scientific Research (18002015) from the Ministry of Education, Culture, Sports, Science and Technology of Japan, a grant of the Program for Promotion of Fundamental Studies in Health Science from the National Institute of Biomedical Innovation of Japan and a grant from Ono Research Foundation.
Author information
Authors and Affiliations
Contributions
Experimental design and discussion: T.O., T.M. and S.N.; performance of experiments and data analysis and interpretation: T.O. (for bleomycin experiments, cell culture and microarray analysis), T.M. (for promoter assays), C.Y. (for RT-PCR), K.N. (for microarray analysis and western blotting), S.K. (for X-gal staining), D.S. (for cell proliferation assays and flow cytometry), Y.K. and T.S. (for liquid chromatography–tandem mass spectrometry analysis), K.T. and Y.T. (for BALF from human subjects), K.C. and M.M. (for analysis of lung function); manuscript preparation: T.O., T.M. and S.N.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Methods, Supplementary Tables 1–4 and Supplementary Figures 1–5 (PDF 3387 kb)
Rights and permissions
About this article
Cite this article
Oga, T., Matsuoka, T., Yao, C. et al. Prostaglandin F2α receptor signaling facilitates bleomycin-induced pulmonary fibrosis independently of transforming growth factor-β. Nat Med 15, 1426–1430 (2009). https://doi.org/10.1038/nm.2066
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2066
This article is cited by
-
The tobacco-specific carcinogen NNK induces pulmonary tumorigenesis via nAChR/Src/STAT3-mediated activation of the renin-angiotensin system and IGF-1R signaling
Experimental & Molecular Medicine (2023)
-
Lipid metabolism in idiopathic pulmonary fibrosis: From pathogenesis to therapy
Journal of Molecular Medicine (2023)
-
Immunophenotypic and molecular characterization of pancreatic neuroendocrine tumors producing serotonin
Modern Pathology (2022)
-
Addition of ROCK inhibitors to prostaglandin derivative (PG) synergistically affects adipogenesis of the 3D spheroids of human orbital fibroblasts (HOFs)
Human Cell (2022)
-
Prostaglandin F2α agonists induced enhancement in collagen1 expression is involved in the pathogenesis of the deepening of upper eyelid sulcus
Scientific Reports (2021)