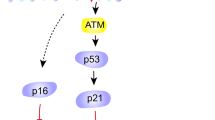
Abstract
Various stimuli, such as telomere dysfunction and oxidative stress, can induce irreversible cell growth arrest, which is termed 'cellular senescence'1,2. This response is controlled by tumor suppressor proteins such as p53 and pRb. There is also evidence that senescent cells promote changes related to aging or age-related diseases3,4,5,6. Here we show that p53 expression in adipose tissue is crucially involved in the development of insulin resistance, which underlies age-related cardiovascular and metabolic disorders. We found that excessive calorie intake led to the accumulation of oxidative stress in the adipose tissue of mice with type 2 diabetes–like disease and promoted senescence-like changes, such as increased activity of senescence-associated β-galactosidase, increased expression of p53 and increased production of proinflammatory cytokines. Inhibition of p53 activity in adipose tissue markedly ameliorated these senescence-like changes, decreased the expression of proinflammatory cytokines and improved insulin resistance in mice with type 2 diabetes–like disease. Conversely, upregulation of p53 in adipose tissue caused an inflammatory response that led to insulin resistance. Adipose tissue from individuals with diabetes also showed senescence-like features. Our results show a previously unappreciated role of adipose tissue p53 expression in the regulation of insulin resistance and suggest that cellular aging signals in adipose tissue could be a new target for the treatment of diabetes (pages 996–967).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stewart, S.A. & Weinberg, R.A. Telomeres: cancer to human aging. Annu. Rev. Cell Dev. Biol. 22, 531–557 (2006).
Serrano, M. & Blasco, M.A. Putting the stress on senescence. Curr. Opin. Cell Biol. 13, 748–753 (2001).
Campisi, J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120, 513–522 (2005).
Shay, J.W. & Wright, W.E. Senescence and immortalization: role of telomeres and telomerase. Carcinogenesis 26, 867–874 (2005).
Minamino, T. & Komuro, I. Vascular cell senescence: contribution to atherosclerosis. Circ. Res. 100, 15–26 (2007).
Minamino, T. & Komuro, I. Vascular aging: insights from studies on cellular senescence, stem cell aging, and progeroid syndromes. Nat. Clin. Pract. Cardiovasc. Med. 5, 637–648 (2008).
Greider, C.W. Telomere length regulation. Annu. Rev. Biochem. 65, 337–365 (1996).
Herbig, U., Ferreira, M., Condel, L., Carey, D. & Sedivy, J.M. Cellular senescence in aging primates. Science 311, 1257 (2006).
Dimri, G.P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. USA 92, 9363–9367 (1995).
Furukawa, S. et al. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest. 114, 1752–1761 (2004).
Hotamisligil, G.S., Shargill, N.S. & Spiegelman, B.M. Adipose expression of tumor necrosis factor-α: direct role in obesity-linked insulin resistance. Science 259, 87–91 (1993).
Weisberg, S.P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Kamei, N. et al. Overexpression of monocyte chemoattractant protein-1 in adipose tissues causes macrophage recruitment and insulin resistance. J. Biol. Chem. 281, 26602–26614 (2006).
Makowski, L. et al. Lack of macrophage fatty-acid–binding protein aP2 protects mice deficient in apolipoprotein E against atherosclerosis. Nat. Med. 7, 699–705 (2001).
Makowski, L., Brittingham, K.C., Reynolds, J.M., Suttles, J. & Hotamisligil, G.S. The fatty acid–binding protein, aP2, coordinates macrophage cholesterol trafficking and inflammatory activity. Macrophage expression of aP2 impacts peroxisome proliferator–activated receptor γ and IκB kinase activities. J. Biol. Chem. 280, 12888–12895 (2005).
Gardner, J.P. et al. Rise in insulin resistance is associated with escalated telomere attrition. Circulation 111, 2171–2177 (2005).
Chin, L. et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 97, 527–538 (1999).
Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. & de Lange, T. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283, 1321–1325 (1999).
Blasco, M.A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
Lee, H.W. et al. Essential role of mouse telomerase in highly proliferative organs. Nature 392, 569–574 (1998).
Ryan, K.M., Ernst, M.K., Rice, N.R. & Vousden, K.H. Role of NF-κB in p53-mediated programmed cell death. Nature 404, 892–897 (2000).
Benoit, V. et al. Transcriptional activation of cyclooxygenase-2 by tumor suppressor p53 requires nuclear factor-κB. Oncogene 25, 5708–5718 (2006).
Kenyon, C. The plasticity of aging: insights from long-lived mutants. Cell 120, 449–460 (2005).
Hwangbo, D.S., Gershman, B., Tu, M.P., Palmer, M. & Tatar, M. Drosophila dFOXO controls lifespan and regulates insulin signalling in brain and fat body. Nature 429, 562–566 (2004).
Giannakou, M.E. et al. Long-lived Drosophila with overexpressed dFOXO in adult fat body. Science 305, 361 (2004).
Blüher, M., Kahn, B.B. & Kahn, C.R. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science 299, 572–574 (2003).
Blüher, M. et al. Adipose tissue selective insulin receptor knockout protects against obesity and obesity-related glucose intolerance. Dev. Cell 3, 25–38 (2002).
Libina, N., Berman, J.R. & Kenyon, C. Tissue-specific activities of C. elegans DAF-16 in the regulation of lifespan. Cell 115, 489–502 (2003).
Yuan, X. et al. Presence of telomeric G-strand tails in the telomerase catalytic subunit TERT knockout mice. Genes Cells 4, 563–572 (1999).
Maeda, N. et al. Diet-induced insulin resistance in mice lacking adiponectin/ACRP30. Nat. Med. 8, 731–737 (2002).
Marino, S., Vooijs, M., van Der Gulden, H., Jonkers, J. & Berns, A. Induction of medulloblastomas in p53-null mutant mice by somatic inactivation of Rb in the external granular layer cells of the cerebellum. Genes Dev. 14, 994–1004 (2000).
Acknowledgements
We thank H. Karagiri for discussion, W.C. Greene (University of California) and T. Fujita (The Tokyo Metropolitan Institute of Medical Science) for expression vector encoding constitutively active IκB and p55-A2-Luc (luciferase reporter vector containing the κB binding sites), respectively, A. Berns (The Netherlands Cancer Institute) for floxed Trp53 mice, and E. Fujita, Y. Ishiyama, R. Kobayashi and Y. Ishikawa for their excellent technical assistance. This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture and Health and Labor Sciences Research Grants (to I.K.); a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan; and grants from the Suzuken Memorial Foundation, the Japan Diabetes Foundation, the Ichiro Kanehara Foundation, the Tokyo Biochemical Research Foundation, the Takeda Science Foundation, the Cell Science Research Foundation and the Japan Foundation of Applied Enzymology (to T.M.).
Author information
Authors and Affiliations
Contributions
T.M. designed and conducted experiments and wrote the manuscript, M.O., I.S., T.K., M.Y., T.I., A. Nojima and Y.O. conducted experiments, A. Nabetani performed telomere analysis, H.M. performed the human studies, F.I. generated telomerase-deficient mice and I.K. evaluated the results, supervised this study and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Methods (PDF 1675 kb)
Rights and permissions
About this article
Cite this article
Minamino, T., Orimo, M., Shimizu, I. et al. A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nat Med 15, 1082–1087 (2009). https://doi.org/10.1038/nm.2014
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2014
This article is cited by
-
Fasting-mimicking diet causes hepatic and blood markers changes indicating reduced biological age and disease risk
Nature Communications (2024)
-
Adipocyte p53 coordinates the response to intermittent fasting by regulating adipose tissue immune cell landscape
Nature Communications (2024)
-
Targeting senescent cells in ageing-related endocrine diseases
Nature Reviews Endocrinology (2023)
-
Senescence of bone marrow mesenchymal stem cells in Wistar male rats receiving normal chow/high-calorie diets with/without vitamin D
Biogerontology (2023)
-
PBMC MicroRNAs: Promising Biomarkers for the Differential Diagnosis of COVID-19 Patients with Abnormal Coagulation Indices
Current Microbiology (2023)