Abstract
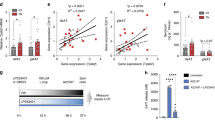
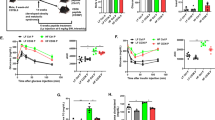
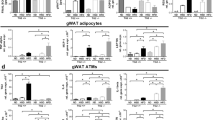
Although mast cell functions have classically been related to allergic responses1,2,3, recent studies indicate that these cells contribute to other common diseases such as multiple sclerosis, rheumatoid arthritis, atherosclerosis, aortic aneurysm and cancer4,5,6,7,8. This study presents evidence that mast cells also contribute to diet-induced obesity and diabetes. For example, white adipose tissue (WAT) from obese humans and mice contain more mast cells than WAT from their lean counterparts. Furthermore, in the context of mice on a Western diet, genetically induced deficiency of mast cells, or their pharmacological stabilization, reduces body weight gain and levels of inflammatory cytokines, chemokines and proteases in serum and WAT, in concert with improved glucose homeostasis and energy expenditure. Mechanistic studies reveal that mast cells contribute to WAT and muscle angiogenesis and associated cell apoptosis and cathepsin activity. Adoptive transfer experiments of cytokine-deficient mast cells show that these cells, by producing interleukin-6 (IL-6) and interferon-γ (IFN-γ), contribute to mouse adipose tissue cysteine protease cathepsin expression, apoptosis and angiogenesis, thereby promoting diet-induced obesity and glucose intolerance. Our results showing reduced obesity and diabetes in mice treated with clinically available mast cell-stabilizing agents suggest the potential of developing new therapies for these common human metabolic disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Galli, S.J., Nakae, S. & Tsai, M. Mast cells in the development of adaptive immune responses. Nat. Immunol. 6, 135–142 (2005).
Bingham, C.O. III & Austen, K.F. Mast-cell responses in the development of asthma. J. Allergy Clin. Immunol. 105, S527–S534 (2000).
Robbie-Ryan, M. & Brown, M. The role of mast cells in allergy and autoimmunity. Curr. Opin. Immunol. 14, 728–733 (2002).
Secor, V.H., Secor, W.E., Gutekunst, C.A. & Brown, M.A. Mast cells are essential for early onset and severe disease in a murine model of multiple sclerosis. J. Exp. Med. 191, 813–822 (2000).
Lee, D.M. et al. Mast cells: a cellular link between autoantibodies and inflammatory arthritis. Science 297, 1689–1692 (2002).
Sun, J. et al. Mast cells promote atherosclerosis by releasing proinflammatory cytokines. Nat. Med. 13, 719–724 (2007).
Sun, J. et al. Mast cells modulate the pathogenesis of elastase-induced abdominal aortic aneurysms in mice. J. Clin. Invest. 117, 3359–3368 (2007).
Coussens, L.M. et al. Inflammatory mast cells up-regulate angiogenesis during squamous epithelial carcinogenesis. Genes Dev. 13, 1382–1397 (1999).
Weisberg, S.P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Rocha, V.Z. et al. Interferon-γ, a TH1 cytokine, regulates fat inflammation: a role for adaptive immunity in obesity. Circ. Res. 103, 467–476 (2008).
Wu, H. et al. T-cell accumulation and regulated on activation, normal T cell expressed and secreted upregulation in adipose tissue in obesity. Circulation 115, 1029–1038 (2007).
Fantuzzi, G. Adipose tissue, adipokines, and inflammation. J. Allergy Clin. Immunol. 115, 911–919 (2005).
Duttlinger, R. et al. The Wsh and Ph mutations affect the c-kit expression profile: c-kit misexpression in embryogenesis impairs melanogenesis in Wsh and Ph mutant mice. Proc. Natl. Acad. Sci. USA 92, 3754–3758 (1995).
Eigen, H. et al. Evaluation of the addition of cromolyn sodium to bronchodilator maintenance therapy in the long-term management of asthma. J. Allergy Clin. Immunol. 80, 612–621 (1987).
Paterson, W.G. Role of mast cell–derived mediators in acid-induced shortening of the esophagus. Am. J. Physiol. 274, G385–G388 (1998).
Shin, H.Y., Kim, J.S., An, N.H., Park, R.K. & Kim, H.M. Effect of disodium cromoglycate on mast cell–mediated immediate-type allergic reactions. Life Sci. 74, 2877–2887 (2004).
Grimbaldeston, M.A. et al. Mast cell–deficient W-sash c-kit mutant KitW-sh/W-sh mice as a model for investigating mast cell biology in vivo. Am. J. Pathol. 167, 835–848 (2005).
Cannon, B. & Nedergaard, J. Brown adipose tissue: function and physiological significance. Physiol. Rev. 84, 277–359 (2004).
Koban, M. & Swinson, K.L. Chronic REM-sleep deprivation of rats elevates metabolic rate and increases UCP1 gene expression in brown adipose tissue. Am. J. Physiol. Endocrinol. Metab. 289, E68–E74 (2005).
Santone, D.J., Shahani, R., Rubin, B.B., Romaschin, A.D. & Lindsay, T.F. Mast cell stabilization improves cardiac contractile function following hemorrhagic shock and resuscitation. Am. J. Physiol. Heart Circ. Physiol. 294, H2456–H2464 (2008).
Serna, H., Porras, M. & Vergara, P. Mast cell stabilizer ketotifen [4-(1-methyl-4-piperidylidene)-4h-benzo[4,5]cyclohepta[1,2-b]thiophen-10(9H)-one fumarate] prevents mucosal mast cell hyperplasia and intestinal dysmotility in experimental Trichinella spiralis inflammation in the rat. J. Pharmacol. Exp. Ther. 319, 1104–1111 (2006).
Galli, S.J. & Kitamura, Y. Genetically mast-cell–deficient W/Wv and Sl/Sld mice. Their value for the analysis of the roles of mast cells in biologic responses in vivo. Am. J. Pathol. 127, 191–198 (1987).
Crandall, D.L., Hausman, G.J. & Kral, J.G. A review of the microcirculation of adipose tissue: anatomic, metabolic and angiogenic perspectives. Microcirculation 4, 211–232 (1997).
Rupnick, M.A. et al. Adipose tissue mass can be regulated through the vasculature. Proc. Natl. Acad. Sci. USA 99, 10730–10735 (2002).
Pang, C. et al. Macrophage infiltration into adipose tissue may promote angiogenesis for adipose tissue remodeling in obesity. Am. J. Physiol. Endocrinol. Metab. 295, E313–E322 (2008).
Kintscher, U. et al. T-lymphocyte infiltration in visceral adipose tissue: a primary event in adipose tissue inflammation and the development of obesity-mediated insulin resistance. Arterioscler. Thromb. Vasc. Biol. 28, 1304–1310 (2008).
Sato, K. et al. Autophagy is activated in colorectal cancer cells and contributes to the tolerance to nutrient deprivation. Cancer Res. 67, 9677–9684 (2007).
Skoura, A. et al. Essential role of sphingosine 1-phosphate receptor 2 in pathological angiogenesis of the mouse retina. J. Clin. Invest. 117, 2506–2516 (2007).
Xu, J. et al. Proteolytic exposure of a cryptic site within collagen type IV is required for angiogenesis and tumor growth in vivo. J. Cell Biol. 154, 1069–1079 (2001).
Wang, B. et al. Cathepsin S controls angiogenesis and tumor growth via matrix-derived angiogenic factors. J. Biol. Chem. 281, 6020–6029 (2006).
Shi, G.P., Munger, J.S., Meara, J.P., Rich, D.H. & Chapman, H.A. Molecular cloning and expression of human alveolar macrophage cathepsin S, an elastinolytic cysteine protease. J. Biol. Chem. 267, 7258–7262 (1992).
Wolters, P.J. et al. Tissue-selective mast cell reconstitution and differential lung gene expression in mast cell-deficient KitW-sh/KitW-sh sash mice. Clin. Exp. Allergy 35, 82–88 (2005).
Yang, M. et al. Cathepsin L activity controls adipogenesis and glucose tolerance. Nat. Cell Biol. 9, 970–977 (2007).
Hadigan, C. et al. Fasting hyperinsulinemia and changes in regional body composition in human immunodeficiency virus–infected women. J. Clin. Endocrinol. Metab. 84, 1932–1937 (1999).
Wild, R., Ramakrishnan, S., Sedgewick, J. & Griffioen, A.W. Quantitative assessment of angiogenesis and tumor vessel architecture by computer-assisted digital image analysis: effects of VEGF-toxin conjugate on tumor microvessel density. Microvasc. Res. 59, 368–376 (2000).
Acknowledgements
We thank S. Rizkalla and C. Poitou, who contributed to the clinical investigation program. We also thank B. Spiegelman and R.K. Gupta for their help with the 3T3-L1 culture. This study was supported partially by the Established Investigator Award from the American Heart Association (0840118N) (to G.-P.S.) and by US National Institutes of Health grants HL60942, HL67283, HL81090, HL88547 (G.-P.S.), HL34636 (P.L.), DK57521, DK56116 (B.B.K.) and DK52539 and DK64360 (G.S.H.). The clinical work was supported by the Programme Hospitalier de Recherche Clinique, Assistance Publique des Hôpitaux de Paris (AOR 02076), a grant from French National Agency of Research (RIOMA program N°ANR05-PCOD-030-02), and by the Commission of the European Communities (ADAPT project) (to K.C.).
Author information
Authors and Affiliations
Contributions
J.L. induced mouse obesity and diabetes, monitored body weight, performed insulin tolerance and glucose tolerance assays and completed all immunoblot analyses. A. Divoux performed human adipose tissue immunohistology. J.S. cultured mouse mast cells and performed mast cell reconstitution. J.Z. performed human serum tryptase ELISA. K.C. collected and provided all human serum samples and assisted with manuscript writing. J.N.G. helped with the mouse gastrointestinal tract tissue histology analysis. G.K.S. performed mouse tissue immunohistology. P.J.W. helped with data explanation and manuscript writing. J.D. helped with mouse tissue immunohistology. C.Z.G. performed mouse body tissue composition analysis. A. Doria performed human serum tryptase statistic analysis. P.L. assisted with the data interpretation and manuscript writing. R.S.B. helped with the gastrointestinal tract tissue histology analysis and assisted with the manuscript writing. B.B.K. assisted with the data interpretation and manuscript writing. G.S.H. helped with the body tissue composition data analysis. G.-P.S. designed the experiments, participated in all data analyses and prepared the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Fig. 1 and Supplementary Tables 1–4 (PDF 2617 kb)
Rights and permissions
About this article
Cite this article
Liu, J., Divoux, A., Sun, J. et al. Genetic deficiency and pharmacological stabilization of mast cells reduce diet-induced obesity and diabetes in mice. Nat Med 15, 940–945 (2009). https://doi.org/10.1038/nm.1994
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1994
This article is cited by
-
Polarized Th2 cells attenuate high-fat-diet induced obesity through the suppression of lipogenesis
BMC Immunology (2024)
-
Central regulation of stress-evoked peripheral immune responses
Nature Reviews Neuroscience (2023)
-
Immune cells in adipose tissue microenvironment under physiological and obese conditions
Endocrine (2023)
-
The evolving view of thermogenic fat and its implications in cancer and metabolic diseases
Signal Transduction and Targeted Therapy (2022)
-
Immune and non-immune functions of adipose tissue leukocytes
Nature Reviews Immunology (2022)