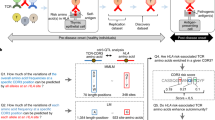
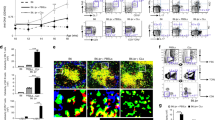
Abstract
The major known genetic risk factors in multiple sclerosis reside in the major histocompatibility complex (MHC) region. Although there is strong evidence implicating MHC class II alleles and CD4+ T cells in multiple sclerosis pathogenesis, possible contributions from MHC class I genes and CD8+ T cells are controversial. We have generated humanized mice expressing the multiple sclerosis–associated MHC class I alleles HLA-A*0301 (encoding human leukocyte antigen-A3 (HLA-A3)) and HLA-A*0201 (encoding HLA-A2) and a myelin-specific autoreactive T cell receptor (TCR) derived from a CD8+ T cell clone from an individual with multiple sclerosis to study mechanisms of disease susceptibility. We demonstrate roles for HLA-A3–restricted CD8+ T cells in induction of multiple sclerosis–like disease and for CD4+ T cells in its progression, and we also define a possible mechanism for HLA-A*0201–mediated protection. To our knowledge, these data provide the first direct evidence incriminating MHC class I genes and CD8+ T cells in the pathogenesis of human multiple sclerosis and reveal a network of MHC interactions that shape the risk of multiple sclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sospedra, M. & Martin, R. Immunology of multiple sclerosis. Annu. Rev. Immunol. 23, 683–747 (2005).
Hafler, D.A. et al. Risk alleles for multiple sclerosis identified by a genomewide study. N. Engl. J. Med. 357, 851–862 (2007).
Olerup, O. & Hillert, J. HLA class II–associated genetic susceptibility in multiple sclerosis: a critical evaluation. Tissue Antigens 38, 1–15 (1991).
van Oosten, B.W. et al. Treatment of multiple sclerosis with the monoclonal anti-CD4 antibody cM-T412: results of a randomized, double-blind, placebo-controlled, MR-monitored phase II trial. Neurology 49, 351–357 (1997).
Segal, B.M. et al. Repeated subcutaneous injections of IL12/23 p40 neutralising antibody, ustekinumab, in patients with relapsing-remitting multiple sclerosis: a phase II, double-blind, placebo-controlled, randomised, dose-ranging study. Lancet Neurol. 7, 796–804 (2008).
Coles, A.J. et al. Monoclonal antibody treatment exposes three mechanisms underlying the clinical course of multiple sclerosis. Ann. Neurol. 46, 296–304 (1999).
Polman, C.H. et al. A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N. Engl. J. Med. 354, 899–910 (2006).
Lassmann, H. & Ransohoff, R.M. The CD4-TH1 model for multiple sclerosis: a critical [correction of crucial] re-appraisal. Trends Immunol. 25, 132–137 (2004).
Goverman, J., Perchellet, A. & Huseby, E.S. The role of CD8+ T cells in multiple sclerosis and its animal models. Curr. Drug Targets Inflamm. Allergy 4, 239–245 (2005).
Friese, M.A. & Fugger, L. Autoreactive CD8+ T cells in multiple sclerosis: a new target for therapy? Brain 128, 1747–1763 (2005).
Hauser, S.L. et al. Immunohistochemical analysis of the cellular infiltrate in multiple sclerosis lesions. Ann. Neurol. 19, 578–587 (1986).
Booss, J., Esiri, M.M., Tourtellotte, W.W. & Mason, D.Y. Immunohistological analysis of T lymphocyte subsets in the central nervous system in chronic progressive multiple sclerosis. J. Neurol. Sci. 62, 219–232 (1983).
Babbe, H. et al. Clonal expansions of CD8+ T cells dominate the T cell infiltrate in active multiple sclerosis lesions as shown by micromanipulation and single cell polymerase chain reaction. J. Exp. Med. 192, 393–404 (2000).
Junker, A. et al. Multiple sclerosis: T cell receptor expression in distinct brain regions. Brain 130, 2789–2799 (2007).
Huseby, E.S. et al. A pathogenic role for myelin-specific CD8+ T cells in a model for multiple sclerosis. J. Exp. Med. 194, 669–676 (2001).
Sun, D. et al. Myelin antigen-specific CD8+ T cells are encephalitogenic and produce severe disease in C57BL/6 mice. J. Immunol. 166, 7579–7587 (2001).
Koh, D.R. et al. Less mortality but more relapses in experimental allergic encephalomyelitis in CD8−/− mice. Science 256, 1210–1213 (1992).
Jiang, H., Zhang, S.I. & Pernis, B. Role of CD8+ T cells in murine experimental allergic encephalomyelitis. Science 256, 1213–1215 (1992).
Najafian, N. et al. Regulatory functions of CD8+CD28− T cells in an autoimmune disease model. J. Clin. Invest. 112, 1037–1048 (2003).
Sawcer, S. et al. A high-density screen for linkage in multiple sclerosis. Am. J. Hum. Genet. 77, 454–467 (2005).
Haines, J.L. et al. Linkage of the MHC to familial multiple sclerosis suggests genetic heterogeneity. The Multiple Sclerosis Genetics Group. Hum. Mol. Genet. 7, 1229–1234 (1998).
Jersild, C. et al. Histocompatibility determinants in multiple sclerosis, with special reference to clinical course. Lancet 2, 1221–1225 (1973).
Marrosu, M.G. et al. Dissection of the HLA association with multiple sclerosis in the founder isolated population of Sardinia. Hum. Mol. Genet. 10, 2907–2916 (2001).
Rubio, J.P. et al. Genetic dissection of the human leukocyte antigen region by use of haplotypes of Tasmanians with multiple sclerosis. Am. J. Hum. Genet. 70, 1125–1137 (2002).
Burfoot, R.K. et al. SNP mapping and candidate gene sequencing in the class I region of the HLA complex: searching for multiple sclerosis susceptibility genes in Tasmanians. Tissue Antigens 71, 42–50 (2008).
Fogdell-Hahn, A., Ligers, A., Gronning, M., Hillert, J. & Olerup, O. Multiple sclerosis: a modifying influence of HLA class I genes in an HLA class II associated autoimmune disease. Tissue Antigens 55, 140–148 (2000).
Harbo, H.F. et al. Genes in the HLA class I region may contribute to the HLA class II-associated genetic susceptibility to multiple sclerosis. Tissue Antigens 63, 237–247 (2004).
Brynedal, B. et al. HLA-A confers an HLA-DRB1 independent influence on the risk of multiple sclerosis. PLoS ONE 2, e664 (2007).
Lincoln, M.R. et al. A predominant role for the HLA class II region in the association of the MHC region with multiple sclerosis. Nat. Genet. 37, 1108–1112 (2005).
Chao, M.J. et al. Transmission of class I/II multi-locus MHC haplotypes and multiple sclerosis susceptibility: accounting for linkage disequilibrium. Hum. Mol. Genet. 16, 1951–1958 (2007).
Risch, N. & Merikangas, K. The future of genetic studies of complex human diseases. Science 273, 1516–1517 (1996).
Koeleman, B.P., Dudbridge, F., Cordell, H.J. & Todd, J.A. Adaptation of the extended transmission/disequilibrium test to distinguish disease associations of multiple loci: the Conditional Extended Transmission/Disequilibrium Test. Ann. Hum. Genet. 64, 207–213 (2000).
Gregersen, J.W. et al. Functional epistasis on a common MHC haplotype associated with multiple sclerosis. Nature 443, 574–577 (2006).
Honma, K. et al. Identification of an epitope derived from human proteolipid protein that can induce autoreactive CD8+ cytotoxic T lymphocytes restricted by HLA-A3: evidence for cross-reactivity with an environmental microorganism. J. Neuroimmunol. 73, 7–14 (1997).
Klein, L., Klugmann, M., Nave, K.A., Tuohy, V.K. & Kyewski, B. Shaping of the autoreactive T cell repertoire by a splice variant of self protein expressed in thymic epithelial cells. Nat. Med. 6, 56–61 (2000).
Serafini, B. et al. Dysregulated Epstein-Barr virus infection in the multiple sclerosis brain. J. Exp. Med. 204, 2899–2912 (2007).
Alam, S.M. & Gascoigne, N.R. Posttranslational regulation of TCR Vα allelic exclusion during T cell differentiation. J. Immunol. 160, 3883–3890 (1998).
McGargill, M.A., Derbinski, J.M. & Hogquist, K.A. Receptor editing in developing T cells. Nat. Immunol. 1, 336–341 (2000).
Sim, B.C. et al. Thymic skewing of the CD4/CD8 ratio maps with the T cell receptor α-chain locus. Curr. Biol. 8, 701–704 (1998).
Wallstrom, E. et al. Increased reactivity to myelin oligodendrocyte glycoprotein peptides and epitope mapping in HLA DR2(15)+ multiple sclerosis. Eur. J. Immunol. 28, 3329–3335 (1998).
Vandenbark, A.A. et al. Recombinant TCR ligand induces tolerance to myelin oligodendrocyte glycoprotein 35–55 peptide and reverses clinical and histological signs of chronic experimental autoimmune encephalomyelitis in HLA-DR2 transgenic mice. J. Immunol. 171, 127–133 (2003).
McMahon, E.J., Bailey, S.L., Castenada, C.V., Waldner, H. & Miller, S.D. Epitope spreading initiates in the CNS in two mouse models of multiple sclerosis. Nat. Med. 11, 335–339 (2005).
Janssen, E.M. et al. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421, 852–856 (2003).
Shedlock, D.J. & Shen, H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300, 337–339 (2003).
Sun, J.C. & Bevan, M.J. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300, 339–342 (2003).
Hogquist, K.A., Baldwin, T.A. & Jameson, S.C. Central tolerance: learning self-control in the thymus. Nat. Rev. Immunol. 5, 772–782 (2005).
Liston, A., Lesage, S., Wilson, J., Peltonen, L. & Goodnow, C.C. Aire regulates negative selection of organ-specific T cells. Nat. Immunol. 4, 350–354 (2003).
Pascolo, S. et al. HLA-A2.1-restricted education and cytolytic activity of CD8+ T lymphocytes from β2 microglobulin (β2m) HLA-A2.1 monochain transgenic H-2Db β2m double knockout mice. J. Exp. Med. 185, 2043–2051 (1997).
Vitiello, A., Marchesini, D., Furze, J., Sherman, L.A. & Chesnut, R.W. Analysis of the HLA-restricted influenza-specific cytotoxic T lymphocyte response in transgenic mice carrying a chimeric human-mouse class I major histocompatibility complex. J. Exp. Med. 173, 1007–1015 (1991).
Madsen, L. et al. Mice lacking all conventional MHC class II genes. Proc. Natl. Acad. Sci. USA 96, 10338–10343 (1999).
Acknowledgements
We thank H. von Boehmer (Dana-Farber Cancer Institute) for the BW58TCR-αβ− T cell hybridoma, A. McMichael (University of Oxford) for GAP.A3, W.E. Biddison (US National Institutes of Health) for providing DNA and RNA from the 2D1 T cell clone and N. Willcox and A. Vincent for critical reading of the manuscript. Work in the authors' laboratories is supported by the Danish (to L. Fugger) and UK (to L. Fugger and E.Y.J.) Medical Research Councils, the Karen Elise Jensen Foundation, the Lundbeck Foundation, the Danish Multiple Sclerosis Society, the European Union (European Commission Descartes Prize and FP6 Neuropromise, Mugen and ARDIS) and GB Holding (Viby) Aps (L. Fugger). M.A.F. was supported by the Deutsche Forschungsgemeinschaft (DFG FR1720/1-1) and a Medical Research Council UK Career Development Fellowship and is supported by the Deutsche Forschungsgemeinschaft Emmy Noether Programme (FR1720/3-1). R.E. is supported by the Schweizerische MS Gesellschaft and a Berrow Scholarship. E.Y.J. is a Cancer Research UK Principal Research Fellow.
Author information
Authors and Affiliations
Contributions
M.A.F. and L. Fugger formulated the hypothesis and initiated and organized the study. M.A.F. constructed the transgenic vectors, generated the transgenic mice, performed the main experimental work and analyzed the data. K.B.J. and M.J.C. conducted the histological and immunohistochemical analyses. L.Friis, R.E., R.M.M., L.T.J. and V.H. helped with some experimental procedures. L. Fugger oversaw the experiments, analyzed the data and provided the funding for the research. M.A.F. drafted the manuscript, and E.Y.J, J.I.B. and L. Fugger helped with writing the final manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–7 and Supplementary Methods (PDF 4514 kb)
Rights and permissions
About this article
Cite this article
Friese, M., Jakobsen, K., Friis, L. et al. Opposing effects of HLA class I molecules in tuning autoreactive CD8+ T cells in multiple sclerosis. Nat Med 14, 1227–1235 (2008). https://doi.org/10.1038/nm.1881
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1881
This article is cited by
-
CD8+ T cells specific for cryptic apoptosis-associated epitopes exacerbate experimental autoimmune encephalomyelitis
Cell Death & Disease (2021)
-
HLA-A and HLA-DRB1 may play a unique role in ovarian teratoma-associated anti-N-methyl-D-aspartate receptor encephalitis
Reproductive Biology and Endocrinology (2020)
-
Field Synopsis and Re-analysis of Systematic Meta-analyses of Genetic Association Studies in Multiple Sclerosis: a Bayesian Approach
Molecular Neurobiology (2018)
-
Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis
Nature Reviews Neurology (2017)
-
Multiple sclerosis: experimental models and reality
Acta Neuropathologica (2017)