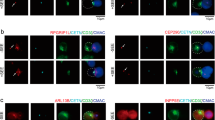
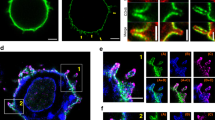
Abstract
Activation of phosphoinositide 3-kinase (PI3K) at the immunological synapse between a T cell and an antigen-presenting cell (APC) has not been demonstrated. Using fluorescent-specific probes, we show here that the formation of an immunological synapse led to sustained production of 3′-phosphoinositides in the T cell, whereby phosphatidylinositol-3,4,5-trisphosphate (PIP3) but not phosphatidylinositol-3,4-bisphosphate was localized to the cell membrane. The accumulation of PIP3 after T cell activation preceded the increase in intracellular calcium. Neither the formation of conjugates between T cells and APCs nor signaling events such as phosphotyrosine accumulation and calcium increase changed substantially when PI3K was inhibited, and only a limited reduction in synthesis of interleukin 2 occurred. In T cell–APC conjugates, PIP3 accumulated at the T cell–APC synapse as well as in the rest of the T cell plasma membrane, which indicated unusual regulation of PI3K activity during antigen presentation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Paul, W.E. & Seder, R.A. Lymphocyte responses and cytokines. Cell 76, 241–251 (1994).
Shaw, A.S. & Dustin, M.L. Making the T cell receptor go the distance: a topological view of T cell activation. Immunity 6, 361–369 (1997).
Grakoui, A. et al. The immunological synapse: a molecular machine controlling T cell activation. Science 285, 221–227 (1999).
Monks, C.R.F., Freiberg, B.A., Kupfer, H., Sciaky, N. & Kupfer, A. Three-dimensional segregation of supramolacular activation clusters in T cells. Nature 395, 82–86 (1999).
Krummel, M.F., Sjaastad, M.D., Wulfing, C. & Davis, M.M. Differential clustering of CD4 and CD3ζ during T cell recognition. Science 289, 1349–1352 (2000).
Leupin, O., Zaru, R., Laroche, T., Müller, S. & Valitutti, S. Exclusion of CD45 from the T-cell receptor signaling area in antigen-stimulated T lymphocytes. Curr. Biol. 10, 277–280 (2000).
Tuosto, L. & Acuto, O. CD28 affects the earliest signaling events generated by TCR engagement. Eur. J. Immunol. 28, 2131–2142 (1998).
Bromley, S.K. et al. The immunological synapse and CD28-CD80 interactions. Nature Immunol. 2, 1159–1166 (2001).
Morgan, M.M. et al. Superantigen-induced T cell:B cell conjugation is mediated by LFA-1 and requires signaling through Lck, but not ZAP-70. J. Immunol. 167, 5708–5718 (2001).
Wulfing, C. & Davis, M.M. A receptor/cytoskeletal movement triggered by costimulation during T cell activation. Science 282, 2266–2269 (1998).
Delon, J., Bercovici, N., Liblau, R. & Trautmann, A. Imaging antigen recognition by naive CD4+ T cells: compulsory cytoskeletal alterations for the triggering of an intracellular calcium response. Eur. J. Immunol. 28, 716–729 (1998).
Cantrell, D. T cell antigen receptor signal transduction pathways. Annu. Rev. Immunol. 14, 259–274 (1996).
Mustelin, T., Coggeshall, K.M., Isakov, N. & Altman, A. T cell antigen receptor-mediated activation of phospholipase C requires tyrosine phosphorylation. Science 247, 1584–1587 (1990).
Divecha, N. & Irvine, R.F. Phospholipid signaling. Cell 80, 269–278 (1995).
Lemmon, M.A., Ferguson, K.M. & Abrams, C.S. Pleckstrin homology domains and the cytoskeleton. FEBS Lett. 513, 71–76 (2002).
Rebecchi, M.J. & Scarlata, S. Pleckstrin homology domains: a common fold with diverse functions. Annu. Rev. Biophys. Biomol. Struct. 27, 503–528 (1998).
Kavran, J.M. et al. Specificity and promiscuity in phosphoinositide binding by pleckstrin homology domains. J. Biol. Chem. 273, 30497–30508 (1998).
Rameh, L.E. et al. A comparative analysis of the phosphoinositide binding specificity of pleckstrin homology domains. J. Biol. Chem. 272, 22059–22066 (1997).
Burgering, B.M. & Coffer, P.J. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature 376, 599–602 (1995).
Datta, K., Bellacosa, A., Chan, T.O. & Tsichlis, P.N. Akt is a direct target of the phosphatidylinositol 3-kinase. Activation by growth factors, v-src and v-Ha-ras, in Sf9 and mammalian cells. J. Biol. Chem. 271, 30835–30839 (1996).
Gray, A., Van Der Kaay, J. & Downes, C.P. The pleckstrin homology domains of protein kinase B and GRP1 (general receptor for phosphoinositides-1) are sensitive and selective probes for the cellular detection of phosphatidylinositol 3,4-bisphosphate and/or phosphatidylinositol 3,4,5-trisphosphate in vivo. Biochem. J. 344, 929–936 (1999).
Astoul, E., Watton, S. & Cantrell, D. The dynamics of protein kinase B regulation during B cell antigen receptor engagement. J. Cell Biol. 145, 1511–1520 (1999).
Astoul, E., Cantrell, D.A., Edmunds, C. & Ward, S.G. PI 3-K and T-cell activation: limitations of T-leukemic cell lines as signaling models. Trends Immunol. 22, 490–496 (2001).
Genot, E. & Cantrell, D.A. Ras regulation and function in lymphocytes. Curr. Opin. Immunol. 12, 289–294 (2000).
Alessi, D.R. et al. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J. 15, 6541–6551 (1996).
Anderson, K.E., Coadwell, J., Stephens, L.R. & Hawkins, P.T. Translocation of PDK-1 to the plasma membrane is important in allowing PDK-1 to activate protein kinase B. Curr. Biol. 8, 684–691 (1998).
Weiner, O.D. et al. Spatial control of actin polymerization during neutrophil chemotaxis. Nature Cell Biol. 1, 75–81 (1999).
Sanchez-Madrid, F. & del Pozo, M.A. Leukocyte polarization in cell migration and immune interactions. EMBO J. 18, 501–511 (1999).
Weiner, O.D. Regulation of cell polarity during eukaryotic chemotaxis: the chemotactic compass. Curr. Opin. Cell Biol. 14, 196–202 (2002).
Meili, R. et al. Chemoattractant-mediated transient activation and membrane localization of Akt/PKB is required for efficient chemotaxis to cAMP in Dictyostelium. EMBO J. 18, 2092–2105 (1999).
Haugh, J.M., Codazzi, F., Teruel, M. & Meyer, T. Spatial sensing in fibroblasts mediated by 3′ phosphoinositides. J. Cell Biol. 151, 1269–1280 (2000).
Servant, G. et al. Polarization of chemoattractant receptor signaling during neutrophil chemotaxis. Science 287, 1037–1040 (2000).
Ridley, A.J. Rho proteins, PI 3-kinases, and monocyte/macrophage motility. FEBS Lett. 498, 168–171 (2001).
Rickert, P., Weiner, O.D., Wang, F., Bourne, H.R. & Servant, G. Leukocytes navigate by compass: roles of PI3Kg and its lipid products. Trends Cell Biol. 10, 466–473 (2000).
Van der Kaay, J., Beck, M., Gray, A. & Downes, C.P. Distinct phosphatidylinositol 3-kinase lipid products accumulate upon oxidative and osmotic stress and lead to different cellular responses. J. Biol. Chem. 274, 35963–35968 (1999).
Banfic, H. et al. A novel integrin-activated pathway forms PKB/Akt-stimulatory phosphatidylinositol 3,4-bisphosphate via phosphatidylinositol 3-phosphate in platelets. J. Biol. Chem. 273, 13–16 (1998).
Holgado-Madruga, M., Emlet, D.R., Moscatello, D.K., Godwin, A.K. & Wong, A.J. A Grb2-associated docking protein in EGF- and insulin-receptor signalling. Nature 379, 560–564 (1996).
Ferguson, K.M. et al. Structural basis for discrimination of 3-phosphoinositides by pleckstrin homology domains. Molecular Cell 6, 373–384 (2000).
Dowler, S. et al. Identification of pleckstrin-homology-domain-containing proteins with novel phosphoinositide-binding specificities. Biochem. J. 351, 19–31 (2000).
Kimber, W.A. et al. Evidence that the tandem-pleckstrin-homology-domain-containing protein TAPP1 interacts with Ptd(3,4)P2 and the multi-PDZ-domain-containing protein MUPP1 in vivo. Biochem. J. 361, 525–536 (2002).
Marshall, A.J., Krahn, A.K., Ma, K., Duronio, V. & Hou, S. TAPP1 and TAPP2 are targets of phosphatidylinositol 3-kinase signaling in B cells: Sustained plasma membrane recruitment triggered by the B-cell antigen receptor. Mol. Cell. Biol. 22, 5479–5491 (2002).
Donnadieu, E. et al. Differential roles of Lck and Itk in T cell response to antigen recognition revealed by calcium imaging and electron microscopy. J. Immunol. 166, 5540–5549 (2001).
Chung, C.Y., Funamoto, S. & Firtel, R.A. Signaling pathways controlling cell polarity and chemotaxis. Trends Biochem. Sci. 26, 557–566 (2001).
Krummel, M.F. & Davis, M.M. Dynamics of the immunological synapse: finding, establishing and solidifying a connection. Curr. Opin. Immunol. 14, 66–74 (2002).
Haefner, B., Baxter, R., Fincham, V.J., Downes, C.P. & Frame, M.C. Cooperation of Src homology domains in the regulated binding of phosphatidylinositol 3-kinase. A role for the Src homology 2 domain. J. Biol. Chem. 270, 7937–7943 (1995).
Pages, F. et al. Two distinct intracytoplasmic regions of the T-cell adhesion molecule CD28 participate in phosphatidylinositol 3-kinase association. J. Biol. Chem. 271, 9403–9409 (1996).
Bruyns, E. et al. T cell receptor (TCR) interacting molecule (TRIM), a novel disulfide-linked dimer associated with the TCR-CD3-ζ complex, recruits intracellular signaling proteins to the plasma membrane. J. Exp. Med. 188, 561–575 (1998).
Zhang, W., Sloan-Lancaster, J., Kitchen, J., Trible, R.P. & Samelson, L.E. LAT: the ZAP-70 tyrosine kinase substrate that links T cell receptor to cellular activation. Cell 92, 83–92 (1998).
Truitt, K.E. et al. CD28 delivers costimulatory signals independently of its association with phosphatidylinositol 3-kinase. J. Immunol. 155, 4702–4710 (1995).
Ni, H.T., Deeths, M.J. & Mescher, M.F. Phosphatidylinositol 3 kinase activity is not essential for B7-1-mediated costimulation of proliferation or development of cytotoxicity in murine T cells. J. Immunol. 157, 2243–2246 (1996).
Shi, J., Cinek, T., Truitt, K.E. & Imboden, J.B. Wortmannin, a phosphatidylinositol 3-kinase inhibitor, blocks antigen-mediated, but not CD3 monoclonal antibody-induced, activation of murine CD4+ T cells. J. Immunol. 158, 4688–4695 (1997).
Weiner, O.D. et al. A PtdInsP(3)- and Rho GTPase-mediated positive feedback loop regulates neutrophil polarity. Nature Cell Biol. 24, 509–512 (2002).
Wang, F. et al. Lipid products of PI(3)Ks maintain persistent cell polarity and directed motility in neutrophils. Nature Cell Biol. 24, 513–518 (2002).
Iijima, M. & Devreotes, P. Tumor suppressor PTEN mediates sensing of chemoattractant gradients. Cell 109, 599–610 (2002).
Funamoto, S., Meili, R., Lee, S., Parry, L. & Firtel, R.A. Spatial and temporal regulation of 3-phosphoinositides by PI 3-kinase and PTEN mediates chemotaxis. Cell 109, 611–623 (2002).
Blank, U., Boitel, B., Mege, D., Ermonval, M. & Acuto, O. Analysis of tetanus toxin peptide/DR recognition by human T cell receptors reconstituted into a murine T cell hybridoma. Eur. J. Immunol. 23, 3057–3065 (1993).
Ingham, R.J. et al. The Gab1 docking protein links the B cell antigen receptor to the phosphatidylinositol 3-kinase/Akt signaling pathway and to the SHP2 tyrosine phosphatase. J. Biol. Chem. 276, 12257–12265 (2001).
Takebe, Y. et al. SRα: an efficient and versatile mammalian cDNA expression system composed of the simian virus 40 early promoter and the R-U5 segment of human T-cell leukemia virus type 1 long terminal repeat. Mol. Cell. Biol. 8, 466–472 (1988).
Acknowledgements
We thank E. Donnadieu, C. Randriamampita and A. Trautmann for their comments on the manuscript; C. Arrieumerlou for discussions; and M.O. Lhuillier for the movies. Supported by the Fondation pour la Recherche Médicale (J.H.) and grants from Institut National de la Santé et de la Recherche Médicale, Centre National de la Recherche Scientifique and the Ligue Nationale contre le Cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Web Movie 1.
GFP-Akt-PH translocation in T cell-APC conjugates. L625.7 cells were plated on glass coverslips mounted on Petri dishes and incubated overnight at 37 °C with antigen [1 μg/ml tt(830-843) peptide] before the addition of GFP-Akt-PH-expressing T8.1 cells previously loaded with Fura-2/AM. Transmitted light, fluorescence and intracellular calcium were measured every 10 s for 20 min. Transmitted light images are superimposed on fluorescence images showing Akt distribution. The black arrow indicates the conjugate shown in Fig. 1. Insert, left side, calcium response in the same conjugates. (AVI 4605 kb)
Web Movie 2.
Inhibition of Akt membrane relocalization by wortmannin. T8.1 cells were incubated with Fura-2/AM and 100 nM wortmannin at 37 °C for 20 min before being added to antigen-pulsed L625.7 cells. Transmitted light images are superimposed on calcium (left) and fluorescence images (right). Images were acquired every 10 sec for 18 min. (AVI 1683 kb)
Web Movie 3.
Delocalization of Akt in a T cell-APC conjugate after PI3K inhibition. T8.1 cells were incubated with Fura-2/AM and added to L625.7 cells pulsed with antigen. Then, 7 min after the beginning of acquisition, 1 ml of buffer containing 100 nM wortmannin was added (+WTN). Transmitted light images are superimposed on calcium images (left) and fluorescence images (right). Images were acquired every 10 sec for 16 min. (AVI 1547 kb)
Web Movie 4.
GFP-Akt-PH translocation in human T cells interacting with DCs. DCs were plated on polylysine-coated glass coverslips in the presence of 0.1 μg/ml SEE. Human primary T cells, transiently transfected with GFP-Akt-PH, were added. Left, transmitted light images; right, distribution of Akt. Images were acquired every 10 s for 17 min. (AVI 1806 kb)
Rights and permissions
About this article
Cite this article
Harriague, J., Bismuth, G. Imaging antigen-induced PI3K activation in T cells. Nat Immunol 3, 1090–1096 (2002). https://doi.org/10.1038/ni847
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni847
This article is cited by
-
CAR T-cell behavior and function revealed by real-time imaging
Seminars in Immunopathology (2023)
-
Regulation of T cell signalling by membrane lipids
Nature Reviews Immunology (2016)
-
Molecular mechanisms and functional implications of polarized actin remodeling at the T cell immunological synapse
Cellular and Molecular Life Sciences (2015)
-
Serine-threonine kinases in TCR signaling
Nature Immunology (2014)
-
Nucleofection induces transient eIF2α phosphorylation by GCN2 and PERK
Gene Therapy (2013)