Abstract
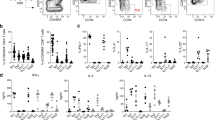
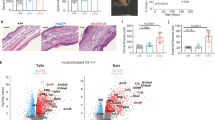
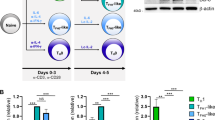
TH-17 cells are a distinct lineage of proinflammatory T helper cells that are essential for autoimmune disease. In mice, commitment to the TH-17 lineage is dependent on transforming growth factor-β and interleukin 6 (IL-6). Here we demonstrate that IL-23 and IL-1β induced the development of human TH-17 cells expressing IL-17A, IL-17F, IL-22, IL-26, interferon-γ, the chemokine CCL20 and transcription factor RORγt. In situ, TH-17 cells were identified by expression of the IL-23 receptor and the memory T cell marker CD45RO. Psoriatic skin lesions contained IL-23-producing dendritic cells and were enriched in the cytokines produced by human TH-17 cells that promote the production of antimicrobial peptides in human keratinocytes. Our data collectively indicate that human and mouse TH-17 cells require distinct factors during differentiation and that human TH-17 cells may regulate innate immunity in epithelial cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mosmann, T.R. & Coffman, R.L. TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. Annu. Rev. Immunol. 7, 145–173 (1989).
Abbas, A.K., Murphy, K.M. & Sher, A. Functional diversity of helper T lymphocytes. Nature 383, 787–793 (1996).
Aggarwal, S., Ghilardi, N., Xie, M.H., de Sauvage, F.J. & Gurney, A.L. Interleukin-23 promotes a distinct CD4 T cell activation state characterized by the production of interleukin-17. J. Biol. Chem. 278, 1910–1914 (2003).
Harrington, L.E. et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 6, 1123–1132 (2005).
Steinman, L. A brief history of TH17, the first major revision in the TH1/TH2 hypothesis of T cell–mediated tissue damage. Nat. Med. 13, 139–145 (2007).
Bettelli, E., Oukka, M. & Kuchroo, V.K. TH-17 cells in the circle of immunity and autoimmunity. Nat. Immunol. 8, 345–350 (2007).
Zheng, Y. et al. Interleukin-22, a TH17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature 445, 648–651 (2007).
Liang, S.C. et al. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J. Exp. Med. 203, 2271–2279 (2006).
Langrish, C.L. et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005).
Cua, D.J. et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421, 744–748 (2003).
Murphy, C.A. et al. Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J. Exp. Med. 198, 1951–1957 (2003).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Yen, D. et al. IL-23 is essential for T cell-mediated colitis and promotes inflammation via IL-17 and IL-6. J. Clin. Invest. 116, 1310–1316 (2006).
Oppmann, B. et al. Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12. Immunity 13, 715–725 (2000).
Mangan, P.R. et al. Transforming growth factor-β induces development of the TH17 lineage. Nature 441, 231–234 (2006).
Veldhoen, M., Hocking, R.J., Atkins, C.J., Locksley, R.M. & Stockinger, B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24, 179–189 (2006).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Sutton, C., Brereton, C., Keogh, B., Mills, K.H. & Lavelle, E.C. A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J. Exp. Med. 203, 1685–1691 (2006).
Batten, M. et al. Interleukin 27 limits autoimmune encephalomyelitis by suppressing the development of interleukin 17–producing T cells. Nat. Immunol. 7, 929–936 (2006).
Stumhofer, J.S. et al. Interleukin 27 negatively regulates the development of interleukin 17–producing T helper cells during chronic inflammation of the central nervous system. Nat. Immunol. 7, 937–945 (2006).
Laurence, A. et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 26, 371–381 (2007).
Chan, J.R. et al. IL-23 stimulates epidermal hyperplasia via TNF and IL-20R2-dependent mechanisms with implications for psoriasis pathogenesis. J. Exp. Med. 203, 2577–2587 (2006).
Schon, M.P. & Boehncke, W.H. Psoriasis. N. Engl. J. Med. 352, 1899–1912 (2005).
Harder, J. & Schroder, J.M. Psoriatic scales: a promising source for the isolation of human skin-derived antimicrobial proteins. J. Leukoc. Biol. 77, 476–486 (2005).
Selsted, M.E. & Ouellette, A.J. Mammalian defensins in the antimicrobial immune response. Nat. Immunol. 6, 551–557 (2005).
Lee, E. et al. Increased expression of interleukin 23 p19 and p40 in lesional skin of patients with psoriasis vulgaris. J. Exp. Med. 199, 125–130 (2004).
Wolk, K. et al. IL-22 regulates the expression of genes responsible for antimicrobial defense, cellular differentiation, and mobility in keratinocytes: a potential role in psoriasis. Eur. J. Immunol. 36, 1309–1323 (2006).
Teunissen, M.B., Koomen, C.W., de Waal Malefyt, R., Wierenga, E.A. & Bos, J.D. Interleukin-17 and interferon-γ synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J. Invest. Dermatol. 111, 645–649 (1998).
Boniface, K. et al. IL-22 inhibits epidermal differentiation and induces proinflammatory gene expression and migration of human keratinocytes. J. Immunol. 174, 3695–3702 (2005).
Hor, S. et al. The T-cell lymphokine interleukin-26 targets epithelial cells through the interleukin-20 receptor 1 and interleukin-10 receptor 2 chains. J. Biol. Chem. 279, 33343–33351 (2004).
Wiekowski, M.T. et al. Ubiquitous transgenic expression of the IL-23 subunit p19 induces multiorgan inflammation, runting, infertility, and premature death. J. Immunol. 166, 7563–7570 (2001).
Cargill, M. et al. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am. J. Hum. Genet. 80, 273–290 (2007).
Krueger, G.G. et al. A human interleukin-12/23 monoclonal antibody for the treatment of psoriasis. N. Engl. J. Med. 356, 580–592 (2007).
Nagalakshmi, M.L., Murphy, E., McClanahan, T. & de Waal Malefyt, R. Expression patterns of IL-10 ligand and receptor gene families provide leads for biological characterization. Int. Immunopharmacol. 4, 577–592 (2004).
Brown, K.L. & Hancock, R.E. Cationic host defense (antimicrobial) peptides. Curr. Opin. Immunol. 18, 24–30 (2006).
Gurney, A.L. IL-22, a Th1 cytokine that targets the pancreas and select other peripheral tissues. Int. Immunopharmacol. 4, 669–677 (2004).
Fossiez, F. et al. T cell interleukin-17 induces stromal cells to produce proinflammatory and hematopoietic cytokines. J. Exp. Med. 183, 2593–2603 (1996).
Sheikh, F. et al. Cutting edge: IL-26 signals through a novel receptor complex composed of IL-20 receptor 1 and IL-10 receptor 2. J. Immunol. 172, 2006–2010 (2004).
Hoover, D.M. et al. The structure of human macrophage inflammatory protein-3α/CCL20. Linking antimicrobial and CC chemokine receptor-6-binding activities with human β-defensins. J. Biol. Chem. 277, 37647–37654 (2002).
Yang, D. et al. Many chemokines including CCL20/MIP-3α display antimicrobial activity. J. Leukoc. Biol. 74, 448–455 (2003).
Iwasaki, A. & Kelsall, B.L. Localization of distinct Peyer's patch dendritic cell subsets and their recruitment by chemokines macrophage inflammatory protein (MIP)-3α, MIP-3β, and secondary lymphoid organ chemokine. J. Exp. Med. 191, 1381–1394 (2000).
Vanbervliet, B. et al. Sequential involvement of CCR2 and CCR6 ligands for immature dendritic cell recruitment: possible role at inflamed epithelial surfaces. Eur. J. Immunol. 32, 231–242 (2002).
Liao, F. et al. CC-chemokine receptor 6 is expressed on diverse memory subsets of T cells and determines responsiveness to macrophage inflammatory protein 3 α. J. Immunol. 162, 186–194 (1999).
Yang, D. et al. Beta-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6. Science 286, 525–528 (1999).
Stark, M.A. et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 22, 285–294 (2005).
Happel, K.I. et al. Divergent roles of IL-23 and IL-12 in host defense against Klebsiella pneumoniae. J. Exp. Med. 202, 761–769 (2005).
Ye, P. et al. Requirement of interleukin 17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. J. Exp. Med. 194, 519–527 (2001).
Kleinschek, M.A. et al. IL-23 enhances the inflammatory cell response in Cryptococcus neoformans infection and induces a cytokine pattern distinct from IL-12. J. Immunol. 176, 1098–1106 (2006).
Yssel, H.S. & Spits, H. in Current Protocols in Immunology 7–19 (Green and Wiley, New York, 2001).
Acknowledgements
Supported by the National Health and Medical Research Council of Australia (N.J.W.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
N.J.W., K.B., J.R.C., B.S.M., W.M.B., J.D.M., B.B., K.S., T.C., R.A.K., D.J.C., T.K.M., E.P.B. & R.d.W.M. are employed by Schering-Plough Biopharma, which is supported by Schering-Plough Corporation.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–2 and Methods (PDF 1304 kb)
Rights and permissions
About this article
Cite this article
Wilson, N., Boniface, K., Chan, J. et al. Development, cytokine profile and function of human interleukin 17–producing helper T cells. Nat Immunol 8, 950–957 (2007). https://doi.org/10.1038/ni1497
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1497
This article is cited by
-
Differentiation of IL-26+ TH17 intermediates into IL-17A producers via epithelial crosstalk in psoriasis
Nature Communications (2023)
-
Innate and Adaptive Immune Responses Induced by Aspergillus fumigatus Conidia and Hyphae
Current Microbiology (2023)
-
Five-year Maintenance of Clinical Response and Consistent Safety Profile for Guselkumab in Asian patients with Psoriasis from VOYAGE 1 and VOYAGE 2
Dermatology and Therapy (2023)
-
The role of Th17 cells in the pathogenesis and treatment of breast cancer
Cancer Cell International (2022)
-
CD147 mediates epidermal malignant transformation through the RSK2/AP-1 pathway
Journal of Experimental & Clinical Cancer Research (2022)