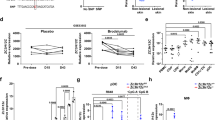
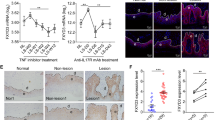
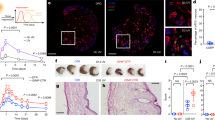
Abstract
Transforming growth factor-β1 (TGF-β1) regulates inflammation and can inhibit activation of the transcription factor NF-κB in certain cell types. Here we show that the TGF-β-induced signaling protein Smad7 bound to TAB2 and TAB3, which are adaptors that link the kinase TAK1 to 'upstream' regulators in the proinflammatory tumor necrosis factor (TNF) signaling pathway. Smad7 thereby promoted TGF-β-mediated anti-inflammatory effects. The formation of Smad7-TAB2 and Smad7-TAB3 complexes resulted in the suppression of TNF signaling through the adaptors TRAF2, TAB2 and/or TAB3, and TAK1. Furthermore, expression of a transgene encoding Smad7 in mouse skin suppressed inflammation and NF-κB nuclear translocation substantially and disrupted the formation of endogenous TRAF2-TAK1-TAB2 and TRAF2-TAK1-TAB3 complexes. Thus, Smad7 is a critical mediator of TGF-β signals that block proinflammatory TNF signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chen, G. & Goeddel, D.V. TNF-R1 signaling: a beautiful pathway. Science 296, 1634–1635 (2002).
Yeh, W.C. et al. Early lethality, functional NF-κB activation, and increased sensitivity to TNF-induced cell death in TRAF2-deficient mice. Immunity 7, 715–725 (1997).
Devin, A. et al. The distinct roles of TRAF2 and RIP in IKK activation by TNF-R1: TRAF2 recruits IKK to TNF-R1 while RIP mediates IKK activation. Immunity 12, 419–429 (2000).
Pikarsky, E. et al. NF-κB functions as a tumour promoter in inflammation-associated cancer. Nature 431, 461–466 (2004).
Luo, J.L., Maeda, S., Hsu, L.C., Yagita, H. & Karin, M. Inhibition of NF-κB in cancer cells converts inflammation-induced tumor growth mediated by TNFα to TRAIL-mediated tumor regression. Cancer Cell 6, 297–305 (2004).
Greten, F.R. et al. IKKβ links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118, 285–296 (2004).
Yamaguchi, K. et al. Identification of a member of the MAPKKK family as a potential mediator of TGF-β signal transduction. Science 270, 2008–2011 (1995).
Ninomiya-Tsuji, J. et al. The kinase TAK1 can activate the NIK-IκB as well as the MAP kinase cascade in the IL-1 signalling pathway. Nature 398, 252–256 (1999).
Takaesu, G. et al. TAB2, a novel adaptor protein, mediates activation of TAK1 MAPKKK by linking TAK1 to TRAF6 in the IL-1 signal transduction pathway. Mol. Cell 5, 649–658 (2000).
Ishitani, T. et al. Role of the TAB2-related protein TAB3 in IL-1 and TNF signaling. EMBO J. 22, 6277–6288 (2003).
Massague, J., Seoane, J. & Wotton, D. Smad transcription factors. Genes Dev. 19, 2783–2810 (2005).
Wrana, J.L. et al. TGF β signals through a heteromeric protein kinase receptor complex. Cell 71, 1003–1014 (1992).
Yoshimura, A., Mori, H., Ohishi, M., Aki, D. & Hanada, T. Negative regulation of cytokine signaling influences inflammation. Curr. Opin. Immunol. 15, 704–708 (2003).
Wang, W. et al. Signaling mechanism of TGF-β1 in prevention of renal inflammation: role of Smad7. J. Am. Soc. Nephrol. 16, 1371–1383 (2005).
Geiser, A.G. et al. Transforming growth factor β1 (TGF-β1) controls expression of major histocompatibility genes in the postnatal mouse: aberrant histocompatibility antigen expression in the pathogenesis of the TGF-β1 null mouse phenotype. Proc. Natl. Acad. Sci. USA 90, 9944–9948 (1993).
Hayashi, H. et al. The MAD-related protein Smad7 associates with the TGF-β receptor and functions as an antagonist of TGF-β signaling. Cell 89, 1165–1173 (1997).
Von Gersdorff, G. et al. Smad3 and Smad4 mediate transcriptional activation of the human Smad7 promoter by transforming growth factor β. J. Biol. Chem. 275, 11320–11326 (2000).
Ulloa, L., Doody, J. & Massague, J. Inhibition of transforming growth factor-β/SMAD signalling by the interferon-γ/STAT pathway. Nature 397, 710–713 (1999).
Benus, G.F. et al. Inhibition of the transforming growth factor β (TGF-β) pathway by interleukin-1β is mediated through TGF-β-activated kinase 1 phosphorylation of SMAD3. Mol. Biol. Cell 16, 3501–3510 (2005).
Pulaski, L., Landstrom, M., Heldin, C.H. & Souchelnytskyi, S. Phosphorylation of Smad7 at Ser-249 does not interfere with its inhibitory role in transforming growth factor-β-dependent signaling but affects Smad7-dependent transcriptional activation. J. Biol. Chem. 276, 14344–14349 (2001).
Mazars, A. et al. Evidence for a role of the JNK cascade in Smad7-mediated apoptosis. J. Biol. Chem. 276, 36797–36803 (2001).
Lallemand, F. et al. Smad7 inhibits the survival nuclear factor κB and potentiates apoptosis in epithelial cells. Oncogene 20, 879–884 (2001).
Karin, M. & Ben Neriah, Y. Phosphorylation meets ubiquitination: the control of NF-κB activity. Annu. Rev. Immunol. 18, 621–663 (2000).
Choi, K.-C. et al. Smad6 negatively regulates interleukin 1 receptor–Toll-like receptor signaling through direct interaction with the adaptor Pellino-1. Nat. Immunol. 7, 1057–1065 (2006).
Hanafusa, H. et al. Involvement of the p38 mitogen-activated protein kinase pathway in transforming growth factor-β-induced gene expression. J. Biol. Chem. 274, 27161–27167 (1999).
Baud, V. et al. Signaling by proinflammatory cytokines: oligomerization of TRAF2 and TRAF6 is sufficient for JNK and IKK activation and target gene induction via an amino-terminal effector domain. Genes Dev. 13, 1297–1308 (1999).
Habelhah, H. et al. Ubiquitination and translocation of TRAF2 is required for activation of JNK but not of p38 or NF-κB. EMBO J. 23, 322–332 (2004).
Xia, Z.P. & Chen, Z.J. TRAF2: a double-edged sword? Sci. STKE 2005, e7 (2005).
Li, H., Kobayashi, M., Blonska, M., You, Y. & Lin, X. Ubiquitination of RIP is required for tumor necrosis factor α-induced NF-κB activation. J. Biol. Chem. 281, 13636–13643 (2006).
He, W. et al. Overexpression of Smad7 results in severe pathological alterations in multiple epithelial tissues. EMBO J. 21, 2580–2590 (2002).
Schiffer, M. et al. Apoptosis in podocytes induced by TGF-β and Smad7. J. Clin. Invest. 108, 807–816 (2001).
Takaesu, G. et al. TAB2, a novel adaptor protein, mediates activation of TAK1 MAPKKK by linking TAK1 to TRAF6 in the IL-1 signal transduction pathway. Mol. Cell 5, 649–658 (2000).
Tada, K. et al. Critical roles of TRAF2 and TRAF5 in tumor necrosis factor-induced NF-κB activation and protection from cell death. J. Biol. Chem. 276, 36530–36534 (2001).
Kelliher, M.A. et al. The death domain kinase RIP mediates the TNF-induced NF-κB signal. Immunity 8, 297–303 (1998).
Vodovotz, Y. et al. Spontaneously increased production of nitric oxide and aberrant expression of the inducible nitric oxide synthase in vivo in the transforming growth factor β1 null mouse. J. Exp. Med. 183, 2337–2342 (1996).
McCartney-Francis, N.L. & Wahl, S.M. Dysregulation of IFN-γ signaling pathways in the absence of TGF-β1. J. Immunol. 169, 5941–5947 (2002).
Li, Q. & Verma, I.M. NF-κB regulation in the immune system. Nat. Rev. Immunol. 2, 725–734 (2002).
Jobin, C. & Sartor, R.B. The IκB/NF-κB system: a key determinant of mucosalinflammation and protection. Am. J. Physiol. Cell Physiol. 278, C451–C462 (2000).
Handel, M.L., McMorrow, L.B. & Gravallese, E.M. Nuclear factor-κB in rheumatoid synovium. Localization of p50 and p65. Arthritis Rheum. 38, 1762–1770 (1995).
Han, S.H., Yea, S.S., Jeon, Y.J., Yang, K.H. & Kaminski, N.E. Transforming growth factor-β1 (TGF-β1) promotes IL-2 mRNA expression through the up-regulation of NF-κB, AP-1 and NF-AT in EL4 cells. J. Pharmacol. Exp. Ther. 287, 1105–1112 (1998).
Sovak, M.A., Arsura, M., Zanieski, G., Kavanagh, K.T. & Sonenshein, G.E. The inhibitory effects of transforming growth factor β1 on breast cancer cell proliferation are mediated through regulation of aberrant nuclear factor-κB/Rel expression. Cell Growth Differ. 10, 537–544 (1999).
Arsura, M., Wu, M. & Sonenshein, G.E. TGF beta 1 inhibits NF-κB/Rel activity inducing apoptosis of B cells: transcriptional activation of IκBα. Immunity 5, 31–40 (1996).
Monteleone, G. et al. Blocking Smad7 restores TGF-β1 signaling in chronic inflammatory bowel disease. J. Clin. Invest. 108, 601–609 (2001).
Dong, C. et al. Deficient Smad7 expression: a putative molecular defect in scleroderma. Proc. Natl. Acad. Sci. USA 99, 3908–3913 (2002).
Asano, Y., Ihn, H., Yamane, K., Kubo, M. & Tamaki, K. Impaired Smad7-Smurf-mediated negative regulation of TGF-β signaling in scleroderma fibroblasts. J. Clin. Invest. 113, 253–264 (2004).
Landstrom, M. et al. Smad7 mediates apoptosis induced by transforming growth factor β in prostatic carcinoma cells. Curr. Biol. 10, 535–538 (2000).
Romieu-Mourez, R., Landesman-Bollag, E., Seldin, D.C. & Sonenshein, G.E. Protein kinase CK2 promotes aberrant activation of nuclear factor-κB, transformed phenotype, and survival of breast cancer cells. Cancer Res. 62, 6770–6778 (2002).
Xavier, S. et al. Amelioration of radiation-induced fibrosis: inhibition of transforming growth factor-β signaling by halofuginone. J. Biol. Chem. 279, 15167–15176 (2004).
Gross, D.J. et al. Treatment with halofuginone results in marked growth inhibition of a von Hippel-Lindau pheochromocytoma in vivo. Clin. Cancer Res. 9, 3788–3793 (2003).
Lee, B.I. et al. MS-275, a histone deacetylase inhibitor, selectively induces transforming growth factor β type II receptor expression in human breast cancer cells. Cancer Res. 61, 931–942 (2001).
Acknowledgements
We thank A. Roberts, G. Merlino and A. Hobbie for the critical reading of the manuscript; J.L. Wrana (University of Toronto) for plasmids expressing Smad7; K. Miyazono (University of Tokyo) for adenovirus expressing Smad7; and C.J. Hastie (University of Dundee) for the TAB3-specific antibody S81B. Supported by the Intramural Research Program of the National Cancer Institute, the National Institutes of Health (X.-J.W.) and the Korea Research Foundation (funded by the Korean Government; KRF-2003-015-C00528 and R08-2003-000-10077-0 to S.H.P).
Author information
Authors and Affiliations
Contributions
S.H., S.L. and C.L. did experimental work and analyzed data; A.G.L. and X.-J.W. developed the K5.Smad7 mice and did the skin inflammation study; Y.S.L., E.-K.L. and S.H.P. did the primary peritoneal macrophage study; S.H.P. participated in study design; S.-J.K. designed and conceptualized the research and supervised the experimental work; and S.H. and S.-J.K. analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
C-terminal region of Smad7 binds to TAB2. (PDF 99 kb)
Supplementary Fig. 2
Smad7 inhibits the activation of NF-κB. (PDF 133 kb)
Supplementary Fig. 3
TGF-β1 inhibits TNF-induced gene expression through Smad7. (PDF 78 kb)
Supplementary Fig. 4
Inhibition of endogenous Smad7 reduces the suppressive effect of TGF-β1 on TNF-induced IKK activity. (PDF 110 kb)
Supplementary Fig. 5
Smad7 does not block the oligomerization of TRAF2 and TRAF5. (PDF 72 kb)
Supplementary Fig. 6
Smad7 does not modulate the ubiquitination of RIP1. (PDF 65 kb)
Supplementary Fig. 7
Smad7 blocks the interaction of TAK1 and TRAF5. (PDF 136 kb)
Supplementary Fig. 8
Smad7C-M3 failed to suppress TRAF2–TAB2- or TRAF2–TAB3-induced IKK activity (PDF 104 kb)
Supplementary Fig. 9
Schematic diagram of crosstalk between TGF-β1 and TNF signaling. (PDF 65 kb)
Supplementary Table
Primer sequences for quantitative RT-PCR. (PDF 45 kb)
Rights and permissions
About this article
Cite this article
Hong, S., Lim, S., Li, A. et al. Smad7 binds to the adaptors TAB2 and TAB3 to block recruitment of the kinase TAK1 to the adaptor TRAF2. Nat Immunol 8, 504–513 (2007). https://doi.org/10.1038/ni1451
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1451
This article is cited by
-
Inactivation of TGF-β signaling in CAR-T cells
Cellular & Molecular Immunology (2024)
-
SMAD7 expression in CAR-T cells improves persistence and safety for solid tumors
Cellular & Molecular Immunology (2024)
-
Adipose-specific deletion of the cation channel TRPM7 inhibits TAK1 kinase-dependent inflammation and obesity in male mice
Nature Communications (2023)
-
The impact of β-glucan on the therapeutic outcome of experimental Trichinella spiralis infection
Parasitology Research (2023)
-
Novel therapies emerging in oncology to target the TGF-β pathway
Journal of Hematology & Oncology (2021)