Abstract
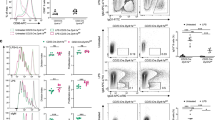
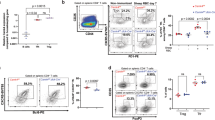
Mice lacking activity of the kinase MEKK1 ('Map3k1ΔKD' mice) have defective activation of the kinase Jnk and increased production of T helper type 2 cytokines after T cell receptor ligation. Here we show that Map3k1ΔKD mice had defective germinal center formation and diminished production of antibodies recognizing thymus-dependent antigens. Those defects were B cell intrinsic, as MEKK1 was necessary for CD40-mediated activation of the kinases Jnk and p38 and transcription factor c-Jun, as well as for expression of cyclin D2 and activation-induced deaminase. MEKK1 was recruited to CD40 and adaptor molecule TRAF2 after CD40 ligation, and Map3k1ΔKD B cells were hypoproliferative after CD40 stimulation. Our data emphasize that MEKK1 is an essential component of signaling cascades needed for thymus-dependent antigen-induced B cell proliferation and antibody production.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chang, L. & Karin, M. Mammalian MAP kinase signalling cascades. Nature 410, 37–40 (2001).
Dong, C., Davis, R.J. & Flavell, R.A. MAP kinases in the immune response. Annu. Rev. Immunol. 20, 55–72 (2002).
Schreck, R. & Rapp, U.R. Raf kinases: Oncogenesis and drug discovery. Int. J. Cancer 119, 2261–2271 (2006).
Minden, A. et al. Differential activation of ERK and JNK mitogen-activated protein kinases by Raf-1 and MEKK. Science 266, 1719–1723 (1994).
Zhang, L. et al. A role for MEK kinase 1 in TGF-b/activin-induced epithelium movement and embryonic eyelid closure. EMBO J. 22, 4443–4454 (2003).
Baud, V. et al. Signaling by proinflammatory cytokines: oligomerization of TRAF2 and TRAF6 is sufficient for JNK and IKK activation and target gene induction via an amino-terminal effector domain. Genes Dev. 13, 1297–1308 (1999).
Xia, Y. et al. MEK kinase 1 is critically required for c-Jun N-terminal kinase activation by proinflammatory stimuli and growth factor-induced cell migration. Proc. Natl. Acad. Sci. USA 97, 5243–5248 (2000).
Gao, M. et al. Jun turnover is controlled through JNK-dependent phosphorylation of the E3 ligase Itch. Science 306, 271–275 (2004).
Fang, D. et al. Dysregulation of T lymphocyte function in itchy mice: a role for Itch in TH2 differentiation. Nat. Immunol. 3, 281–287 (2002).
Hartenstein, B. et al. Th2 cell-specific cytokine expression and allergen-induced airway inflammation depend on JunB. EMBO J. 21, 6321–6329 (2002).
Rothe, M., Sarma, V., Dixit, V.M. & Goeddel, D.V. TRAF-2-mediated activation of NF-κB by TNF receptor 2 and CD40. Science 269, 1424–1427 (1995).
Yuasa, T., Ohno, S., Kehrl, J.H. & Kyriakis, J.M. Tumor necrosis factor signaling to stress-activated protein kinase (SAPK)/Jun NH2-terminal kinase (JNK) and p38. J. Biol. Chem. 273, 22681–22692 (1998).
Habelhah, H. et al. Ubiquitination and translocation of TRAF2 is required for activation of JNK but not of p38 or NF-κB. EMBO J. 23, 322–332 (2004).
Witowsky, J.A. & Johnson, G.L. Ubiquitylation of MEKK1 inhibits its phosphorylation of MKK1 and MKK4 and activation of the ERK1/2 and JNK pathways. J. Biol. Chem. 278, 1403–1406 (2003).
Lu, Z., Xu, S., Joazeiro, C., Cobb, M.H. & Hunter, T. The PHD domain of MEKK1 acts as an E3 ubiquitin ligase and mediates ubiquitination and degradation of ERK1/2. Mol. Cell 9, 945–956 (2002).
Shaulian, E. & Karin, M. AP-1 as a regulator of cell life and death. Nat. Cell Biol. 4, E131–E136 (2002).
Solvason, N. et al. Cyclin D2 is essential for BCR-mediated proliferation and CD5 B cell development. Int. Immunol. 12, 631–638 (2000).
Lam, E.W. et al. Cyclin D3 compensates for loss of cyclin D2 in mouse B-lymphocytes activated via the antigen receptor and CD40. J. Biol. Chem. 275, 3479–3484 (2000).
Bleul, C.C. et al. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin and blocks HIV-1 entry. Nature 382, 829–833 (1996).
Nagata, S. & Golstein, P. The FAS death factor. Science 267, 1449–1456 (1995).
Berberich, I. et al. Cross-linking CD40 of B cells preferentially induces stress-activated protein kinases rather than mitogen-activated protein kinases. EMBO J. 15, 92–101 (1996).
Jabara, H. et al. The binding site for TRAF2 and TRAF3 but not for TRAF6 is essential for CD40-mediated immunoglobulin class switching. Immunity 17, 265–276 (2002).
Ahonen, C. et al. The CD40-TRAF6 axis controls affinity maturation and the generation of long-lived plasma cells. Nat. Immunol. 3, 451–456 (2002).
Yasui, T. et al. Dissection of B cell differentiation during primary immune responses in mice with altered CD40 signals. Int. Immunol. 14, 319–329 (2002).
Hostager, B.S., Haxhinasto, S.A., Rowland, S.L. & Bishop, G.A. Tumor necrosis factor receptor-associated factor 2 (TRAF2)-deficient B lymphocytes reveal novel roles for TRAF2 in CD40 signaling. J. Biol. Chem. 278, 45382–45390 (2003).
Morrison, M.D., Reiley, W., Zhang, M. & Sun, S.C. An atypical tumor necrosis factor (TNF) receptor-associated factor-binding motif of B cell-activating factor belonging to the TNF family (BAFF) receptor mediates induction of the noncanonical NF-κB signaling pathway. J. Biol. Chem. 280, 10018–10024 (2005).
Kawabe, T. et al. The immune responses in CD40-deficient mice: impaired immunoglobulin class switching and germinal center formation. Immunity 1, 167–178 (1994).
Lee, H.H., Dadgostar, H., Cheng, Q., Shu, J. & Cheng, G. NF-κB-mediated up-regulation of Bcl-x and Bfl-1/A1 is required for CD40 survival signaling in B lymphocytes. Proc. Natl. Acad. Sci. USA 96, 9136–9141 (1999).
Li, Z.W., Rickert, R.C. & Karin, M. Genetic dissection of antigen receptor induced-NF-κB activation. Mol. Immunol. 41, 701–714 (2004).
Bonizzi, G. & Karin, M. The two NF-κB activation pathways and their role in innate and adaptive immunity. Trends Immunol. 25, 280–288 (2004).
Shim, J.H. et al. TAK1, but not TAB1 or TAB2, plays an essential role in multiple signaling pathways in vivo. Genes Dev. 19, 2668–2681 (2005).
Sato, S. et al. Essential function for the kinase TAK1 in innate and adaptive immune responses. Nat. Immunol. 6, 1087–1095 (2005).
Gao, M. & Karin, M. Regulating the regulators: control of protein ubiquitination and ubiquitin-like modifications by extracellular stimuli. Mol. Cell 19, 581–593 (2005).
Gallagher, E.D., Xu, S., Moomaw, C., Slaughter, C.A. & Cobb, M.H. Binding of JNK/SAPK to MEKK1 is regulated by phosphorylation. J. Biol. Chem. 277, 45785–45792 (2002).
Anzelon, A.N., Wu, H. & Rickert, R.C. Pten inactivation alters peripheral B lymphocyte fate and reconstitutes CD19 function. Nat. Immunol. 4, 287–294 (2003).
Haas, K.M. et al. Complement receptors CD21/35 link innate and protective immunity during Streptococcus pneumoniae infection by regulating IgG3 antibody responses. Immunity 17, 713–723 (2002).
Kracker, S. & Radbruch, A. in B Cell Protocols (eds. Gu, H. & Rajewsky, K.) 149–159 (Humana Press, Totowa, New Jersey, 2004).
Taguchi, T. et al. Detection of individual mouse splenic T cells producing IFN-γ and IL-5 using the enzyme-linked immunospot (ELISPOT) assay. J. Immunol. Methods 128, 65–73 (1990).
Bonizzi, G. et al. Activation of IKKalpha target genes depends on recognition of specific κB binding sites by RelB:p52 dimers. EMBO J. 23, 4202–4210 (2004).
Hardy, R.H. & Shinton, S.A. in B Cell Protocols (eds. Gu, H. & Rajewsky, K.) 1–24 (Humana Press, Totowa, New Jersey, 2004).
Acknowledgements
Supported by the National Institutes of Health (ES04151 and ES06376 to M.K.), the American Cancer Society (M.K.) and the Leukemia and Lymphoma Society of America (T.E.; SCOR grant 7332-06).
Author information
Authors and Affiliations
Contributions
M.K. designed research and wrote the paper, along with E.G. and T.E.; E.G., T.E. and A.M. did research and analyzed data; A.A. and D.O. helped with flow cytometry and histological analysis; R.H. and M.G. helped with research; and E.J. did the ELISPOT analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Histological analysis of CD35+ FDCs in spleens of KLH-immunized mice. (PDF 1854 kb)
Supplementary Fig. 2
CD40-induced proliferation is defective in Map3k1ΔKD B cells. (PDF 536 kb)
Supplementary Fig. 3
CD4+ T cells hyperproliferate in Map3k1ΔKD mice after KLH immunization. (PDF 749 kb)
Supplementary Fig. 4
Jnk2 (Mapk9) but not Jnk1 (Mapk8) is required for B cell proliferation. (PDF 459 kb)
Supplementary Fig. 5
Defective Jnk and p38 MAPK activation in Map3k1ΔKD B cells following stimulation by CD40L or BAFF. (PDF 517 kb)
Supplementary Fig. 6
Immunoprecipitation of endogenous MEKK1. (PDF 605 kb)
Supplementary Fig. 7
Recruitment of MEKK1 to CD40 and TRAF2 following stimulation with CD40L. (PDF 425 kb)
Supplementary Fig. 8
MEKK1 ubiquitination following CD40 engagement. (PDF 586 kb)
Supplementary Fig. 9
Interaction between MEKK1 and TRAFs. (PDF 588 kb)
Supplementary Table 1
Primer sequences used for real-time PCR. (PDF 1155 kb)
Rights and permissions
About this article
Cite this article
Gallagher, E., Enzler, T., Matsuzawa, A. et al. Kinase MEKK1 is required for CD40-dependent activation of the kinases Jnk and p38, germinal center formation, B cell proliferation and antibody production. Nat Immunol 8, 57–63 (2007). https://doi.org/10.1038/ni1421
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1421
This article is cited by
-
Functions of MAP3Ks in antiviral immunity
Immunologic Research (2023)
-
B-cell maturation antigen targeting strategies in multiple myeloma treatment, advantages and disadvantages
Journal of Translational Medicine (2022)
-
Mechanisms of CD40-dependent cDC1 licensing beyond costimulation
Nature Immunology (2022)
-
Transcriptional regulation of memory B cell differentiation
Nature Reviews Immunology (2021)
-
Signaling mechanisms regulating B-lymphocyte activation and tolerance
Journal of Molecular Medicine (2015)