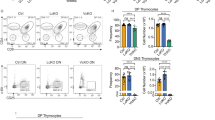
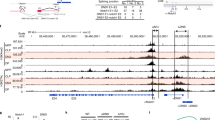
Abstract
Commitment of hematopoietic progenitors to the T cell lineage requires the integration of multiple signaling pathways. Evidence has suggested involvement of hedgehog (Hh) signaling in T cell differentiation through its signal transducer smoothened (Smo). However, the precise function of the Hh pathway remains controversial, mainly because T cell–specific in vivo genetic models have not been used. Using pre–T cell–specific, mature T cell–specific and poly(I)·poly(C)-inducible deletions of Smo and antagonists of Smo signaling, we report here that Hh is an essential positive regulator of T cell progenitor differentiation. Furthermore, we localize Hh function to a stage preceding pre–T cell receptor signaling, connect Smo signaling to the activity of the Gli1 and Gli2 transcription factors and demonstrate that Hh affects regulators of thymocyte survival and proliferation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Schwarz, B.A. & Bhandoola, A. Circulating hematopoietic progenitors with T lineage potential. Nat. Immunol. 5, 953–960 (2004).
von Boehmer, H., Aifantis, I., Azogui, O., Saint-Ruf, C. & Grassi, F. The impact of pre-T-cell receptor signals on gene expression in developing T cells. Cold Spring Harb. Symp. Quant. Biol. 64, 283–289 (1999).
Aifantis, I., Buer, J., von Boehmer, H. & Azogui, O. Essential role of the pre-T cell receptor in allelic exclusion of the T cell receptor β locus. Immunity 7, 601–607 (1997).
Aifantis, I. et al. On the role of the pre-T cell receptor in αβ versus γδ T lineage commitment. Immunity 9, 649–655 (1998).
Radtke, F. et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity 10, 547–558 (1999).
Pui, J.C. et al. Notch1 expression in early lymphopoiesis influences B versus T lineage determination. Immunity 11, 299–308 (1999).
Maillard, I., Adler, S.H. & Pear, W.S. Notch and the immune system. Immunity 19, 781–791 (2003).
Okamura, R.M. et al. Redundant regulation of T cell differentiation and TCRα gene expression by the transcription factors LEF-1 and TCF-1. Immunity 8, 11–20 (1998).
Staal, F.J. & Clevers, H.C. Wnt signaling in the thymus. Curr. Opin. Immunol. 15, 204–208 (2003).
Ioannidis, V., Beermann, F., Clevers, H. & Held, W. The β-catenin–TCF-1 pathway ensures CD4+CD8+ thymocyte survival. Nat. Immunol. 2, 691–697 (2001).
Gounari, F. et al. Somatic activation of β-catenin bypasses pre-TCR signaling and TCR selection in thymocyte development. Nat. Immunol. 2, 863–869 (2001).
St Johnston, D. & Nusslein-Volhard, C. The origin of pattern and polarity in the Drosophila embryo. Cell 68, 201–219 (1992).
Voas, M.G. & Rebay, I. Signal integration during development: insights from the Drosophila eye. Dev. Dyn. 229, 162–175 (2004).
Nusslein-Volhard, C. & Wieschaus, E. Mutations affecting segment number and polarity in Drosophila. Nature 287, 795–801 (1980).
Hammerschmidt, M., Brook, A. & McMahon, A.P. The world according to hedgehog. Trends Genet. 13, 14–21 (1997).
McMahon, A.P. More surprises in the Hedgehog signaling pathway. Cell 100, 185–188 (2000).
Outram, S.V., Varas, A., Pepicelli, C.V. & Crompton, T. Hedgehog signaling regulates differentiation from double-negative to double-positive thymocyte. Immunity 13, 187–197 (2000).
Shah, D.K. et al. Reduced thymocyte development in sonic hedgehog knockout embryos. J. Immunol. 172, 2296–2306 (2004).
Gutierrez-Frias, C. et al. Sonic hedgehog regulates early human thymocyte differentiation by counteracting the IL-7-induced development of CD34+ precursor cells. J. Immunol. 173, 5046–5053 (2004).
Sacedon, R. et al. Expression of hedgehog proteins in the human thymus. J. Histochem. Cytochem. 51, 1557–1566 (2003).
Zhang, X.M., Ramalho-Santos, M. & McMahon, A.P. Smoothened mutants reveal redundant roles for Shh and Ihh signaling including regulation of L/R asymmetry by the mouse node. Cell 105, 781–792 (2001).
Long, F., Zhang, X.M., Karp, S., Yang, Y. & McMahon, A.P. Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation. Development 128, 5099–5108 (2001).
Mao, X., Fujiwara, Y., Chapdelaine, A., Yang, H. & Orkin, S.H. Activation of EGFP expression by Cre-mediated excision in a new ROSA26 reporter mouse strain. Blood 97, 324–326 (2001).
Zuniga-Pflucker, J.C. T-cell development made simple. Nat. Rev. Immunol. 4, 67–72 (2004).
Taipale, J. et al. Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine. Nature 406, 1005–1009 (2000).
Gu, H., Marth, J.D., Orban, P.C., Mossmann, H. & Rajewsky, K. Deletion of a DNA polymerase β gene segment in T cells using cell type-specific gene targeting. Science 265, 103–106 (1994).
Wolfer, A. et al. Inactivation of Notch 1 in immature thymocytes does not perturb CD4 or CD8 T cell development. Nat. Immunol. 2, 235–241 (2001).
Corcoran, R.B. & Scott, M.P. A mouse model for medulloblastoma and basal cell nevus syndrome. J. Neurooncol. 53, 307–318 (2001).
Bhardwaj, G. et al. Sonic hedgehog induces the proliferation of primitive human hematopoietic cells via BMP regulation. Nat. Immunol. 2, 172–180 (2001).
Ruiz i Altaba, A., Sanchez, P. & Dahmane, N. Gli and hedgehog in cancer: tumours, embryos and stem cells. Nat. Rev. Cancer 2, 361–372 (2002).
Zhang, Y. et al. A new function of BMP4: dual role for BMP4 in regulation of Sonic hedgehog expression in the mouse tooth germ. Development 127, 1431–1443 (2000).
Winnier, G., Blessing, M., Labosky, P.A. & Hogan, B.L. Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse. Genes Dev. 9, 2105–2116 (1995).
Bleul, C.C. & Boehm, T. BMP signaling is required for normal thymus development. J. Immunol. 175, 5213–5221 (2005).
Gering, M. & Patient, R. Hedgehog signaling is required for adult blood stem cell formation in zebrafish embryos. Dev. Cell 8, 389–400 (2005).
Lowrey, J.A. et al. Sonic hedgehog promotes cell cycle progression in activated peripheral CD4+ T lymphocytes. J. Immunol. 169, 1869–1875 (2002).
Stewart, G.A. et al. Sonic hedgehog signaling modulates activation of and cytokine production by human peripheral CD4+ T cells. J. Immunol. 169, 5451–5457 (2002).
Sacedon, R. et al. Sonic hedgehog is produced by follicular dendritic cells and protects germinal center B cells from apoptosis. J. Immunol. 174, 1456–1461 (2005).
Bale, A.E. Hedgehog signaling and human disease. Annu. Rev. Genomics Hum. Genet. 3, 47–65 (2002).
Mombaerts, P. et al. RAG-1-deficient mice have no mature B and T lymphocytes. Cell 68, 869–877 (1992).
Aifantis, I., Gounari, F., Scorrano, L., Borowski, C. & von Boehmer, H. Constitutive pre-TCR signaling promotes differentiation through Ca2+ mobilization and activation of NF-κB and NFAT. Nat. Immunol. 2, 403–409 (2001).
Aifantis, I. et al. A critical role for the cytoplasmic tail of pTα in T lymphocyte development. Nat. Immunol. 3, 483–488 (2002).
Acknowledgements
We thank A. McMahon for Smoflox mice; J. McMahon for technical support; H. von Boehmer for support; H. Petrie, H. Nakase, W. Du, J. Pogoriler, J.-C. Zuniga-Pflucker, R. Wechsler-Reya, M. Allegre, F. Gounari, N. Liu and P. Ashton-Rickard for mice and materials; and B. Kee, J. Crispino and A. Wilson for discussions and critical review of the project. Supported by the Cancer Research Institute, the 'V' Foundation for Cancer Research, the Sidney Kimmel Foundation for Cancer Research (I.A.), the University of Chicago Medical Scientist Training Program (S.G.), the University of Chicago Committee on Cancer Biology (M.M.) and the Leukemia Research Foundation (M.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Shh expression in the adult thymus. (PDF 2569 kb)
Supplementary Fig. 2
Pre-T cell specific Smo deletion. (PDF 213 kb)
Supplementary Fig. 3
Effect of Smo deletion on thymic B and TCRγδ cell populations. (PDF 199 kb)
Supplementary Fig. 4
In vitro inhibition of Smo signaling affects development of (DN2) pro-T cells. (PDF 457 kb)
Rights and permissions
About this article
Cite this article
Andaloussi, A., Graves, S., Meng, F. et al. Hedgehog signaling controls thymocyte progenitor homeostasis and differentiation in the thymus. Nat Immunol 7, 418–426 (2006). https://doi.org/10.1038/ni1313
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1313
This article is cited by
-
Activation of the Hedgehog signaling pathway leads to fibrosis in aortic valves
Cell & Bioscience (2023)
-
Effect of the spatial–temporal specific theca cell Cyp17 overexpression on the reproductive phenotype of the novel TC17 mouse
Journal of Translational Medicine (2021)
-
Hedgehog pathway mutations drive oncogenic transformation in high-risk T-cell acute lymphoblastic leukemia
Leukemia (2018)
-
Evidence that glioma-associated oncogene homolog 1 is not a universal risk gene for inflammatory bowel disease in Caucasians
Genes & Immunity (2010)
-
Launching the T-cell-lineage developmental programme
Nature Reviews Immunology (2008)