Abstract
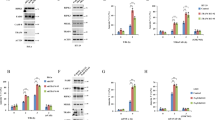
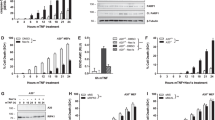
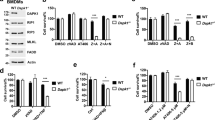
Cell death is achieved by two fundamentally different mechanisms: apoptosis and necrosis. Apoptosis is dependent on caspase activation, whereas the caspase-independent necrotic signaling pathway remains largely uncharacterized. We show here that Fas kills activated primary T cells efficiently in the absence of active caspases, which results in necrotic morphological changes and late mitochondrial damage but no cytochrome c release. This Fas ligand–induced caspase-independent death is absent in T cells that are deficient in either Fas-associated death domain (FADD) or receptor-interacting protein (RIP). RIP is also required for necrotic death induced by tumor necrosis factor (TNF) and TNF-related apoptosis-inducing ligand (TRAIL). In contrast to its role in nuclear factor κB activation, RIP requires its own kinase activity for death signaling. Thus, Fas, TRAIL and TNF receptors can initiate cell death by two alternative pathways, one relying on caspase-8 and the other dependent on the kinase RIP.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kerr, J., Wyllie, A. H. & Currie, A. R. Apoptosis a basic biological phenomenon with wide ranging implications in tissue kinetics. Br. J. Cancer 68, 239–257 (1972).
Fiers, W., Beyaert, R., Declercq, W. & Vandenabeele, P. More than one way to die: apoptosis, necrosis and reactive oxygen damage. Oncogene 18, 7719–7730 (1999).
Scaffidi, C., Kirchhoff, S., Krammer, P. H. & Peter, M. E. Apoptosis signaling in lymphocytes. Curr. Opin. Immunol. 11, 277–285 (1999).
Vercammen, D. et al. Dual signaling of the Fas receptor: initiation of both apoptotic and necrotic cell death pathways. J. Exp. Med. 188, 919–930 (1998).
Kawahara, A., Ohsawa, Y., Matsumura, H., Uchiyama, Y. & Nagata, S. Caspase-independent cell killing by Fas-associated protein with death domain. J. Cell Biol. 143, 1353–1360 (1998).
Schneider, P. et al. Conversion of membrane-bound Fas(CD95) ligand to its soluble form is associated with downregulation of its proapoptotic activity and loss of liver toxicity. J. Exp. Med. 187, 1205–1213 (1998).
Klas, C., Debatin, K. M., Jonker, R. R. & Krammer, P. H. Activation interferes with the APO-1 pathway in mature human T cells. Int. Immunol. 5, 625–630 (1993).
Irmler, M. et al. Inhibition of death receptor signals by cellular FLIP. Nature 388, 190–195 (1997).
Smith, K. G., Strasser, A. & Vaux, D. L. CrmA expression in T lymphocytes of transgenic mice inhibits CD95 (Fas/APO-1)-transduced apoptosis, but does not cause lymphadenopathy or autoimmune disease. EMBO J. 15, 5167–5176 (1996).
Juo, P., Kuo, C. J., Yuan, J. & Blenis, J. Essential requirement for caspase-8/FLICE in the initiation of the Fas- induced apoptotic cascade. Curr. Biol. 8, 1001–1008 (1998).
Juo, P. et al. FADD is required for multiple signaling events downstream of the receptor Fas. Cell Growth Differ. 10, 797–804 (1999).
Kataoka, T. et al. The caspase-8 inhibitor FLIP promotes activation of NF-κB and erk signaling pathways. Curr. Biol. 10, 640–648 (2000).
Stanger, B. Z., Leder, P., Lee, T. H., Kim, E. & Seed, B. RIP - a novel protein containing a death domain that interacts with Fas/Apo-1 (CD95) in yeast and causes cell death. Cell 81, 513–523 (1995).
Ting, A. T., Pimentel-Muinos, F. X. & Seed, B. RIP mediates tumor necrosis factor receptor 1 activation of NF-κB but not Fas/APO-1-initiated apoptosis. EMBO J. 15, 6189–6196 (1996).
Kelliher, M. A. et al. The death domain kinase RIP mediates the TNF-induced NF-κB signal. Immunity 8, 297–303 (1998).
Grimm, S., Stanger, B. Z. & Leder, P. RIP and FADD: two “death domain”-containing proteins can induce apoptosis by convergent, but dissociable, pathways. Proc. Natl Acad. Sci. USA 93, 10923–10927 (1996).
Hsu, H., Huang, J., Shu, H. B., Baichwal, V. & Goeddel, D. V. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity 4, 387–396 (1996).
Lewis, J. et al. Disruption of hsp90 function results in degradation of the death domain kinase, receptor-interacting protein (RIP), and blockage of tumor necrosis factor–induced nuclear factor-κB activation. J. Biol. Chem. 275, 10519–10526 (2000).
Zhang, S. Q., Kovalenko, A., Cantarella, G. & Wallach, D. Recruitment of the IKK signalosome to the p55 TNF receptor: RIP and A20 bind to NEMO (IKKγ) upon receptor stimulation. Immunity 12, 301–311 (2000).
Kroemer, G. & Reed, J. C. Mitochondrial control of cell death. Nature Med. 6, 513–519 (2000).
Li, H., Zhu, H., Xu, C. J. & Yuan, J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94, 491–501 (1998).
Luo, X., Budihardjo, I., Zou, H., Slaughter, C. & Wang, X. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94, 481–490 (1998).
Vercammen, D. et al. Inhibition of caspases increases the sensitivity of L929 cells to necrosis mediated by tumor necrosis factor. J. Exp. Med. 187, 1477–1485 (1998).
Pimentel-Muinos, F. X. & Seed, B. Regulated commitment of TNF receptor signaling: a molecular switch for death or activation. Immunity 11, 783–793 (1999).
Bodmer, J. L. et al. TRAIL receptor-2 signals apoptosis through FADD and caspase-8. Nature Cell Biol. 2, 241–243 (2000).
Kischkel, F. C. et al. Apo2L/TRAIL-dependent recruitment of endogenous FADD and caspase-8 to death receptors 4 and 5. Immunity 12, 611–620 (2000).
Sprick, M. R. et al. FADD/MORT1 and caspase-8 are recruited to TRAIL receptors 1 and 2 and are essential for apoptosis mediated by TRAIL receptor 2. Immunity 12, 599–609 (2000).
Khwaja, A. & Tatton, L. Resistance to the cytotoxic effects of tumor necrosis factor alpha can be overcome by inhibition of a FADD/caspase-dependent signaling pathway. J. Biol. Chem. 274, 36817–36823 (1999).
Boone, E. et al. Structure/Function Analysis of p55 Tumor Necrosis Factor Receptor and Fas-Associated Death Domain: Effect on Necrosis in L929sA Cells. J. Biol. Chem. (in the press2000).
Luschen, S., Ussat, S., Scherer, G., Kabelitz, D. & Adam-Klages, S. Sensitization to death receptor cytotoxicity by inhibition of fas- associated death domain protein (FADD)/caspase signaling. Requirement of cell cycle progression. J. Biol. Chem. 275, 24670–24678 (2000).
Walsh, C. M. et al. A role for FADD in T cell activation and development. Immunity 8, 439–449 (1998).
Watanabe, F. R., Brannan, C. I., Copeland, N. G., Jenkins, N. A. & Nagata, S. Lymphoproliferation disorder in mice explained by defects in Fas antigen that mediates apoptosis. Nature 356, 314–317 (1992).
Gallucci, S., Lolkema, M. & Matzinger, P. Natural adjuvants: endogenous activators of dendritic cells. Nature Med. 5, 1249–1255 (1999).
Baker, B., Zambryski, P., Staskawicz, B. & Dinesh-Kumar, S. P. Signaling in plant-microbe interactions. Science 276, 726–733 (1997).
Berndt, C., Mopps, B., Angermuller, S., Gierschik, P. & Krammer, P. H. CXCR4 and CD4 mediate a rapid CD95-independent cell death in CD4(+) T cells. Proc. Natl Acad. Sci. USA 95, 12556–12561 (1998).
Drenou, B. et al. A caspase-independent pathway of MHC class II antigen-mediated apoptosis of human B lymphocytes. J. Immunol. 163, 4115–4124 (1999).
Kitanaka, C. & Kuchino, Y. Caspase-independent programmed cell death with necrotic morphology. Cell Death Differ. 6, 508–515 (1999).
Acknowledgements
We are grateful to A. Strasser for the gift of the CEM-FADD/DN and CEM-CrmA clones and J. Blenis for the FADD and caspase-8 deficient Jurkat cells. We thank K. Burns and L. French for helpful comments and C. Mattmann for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holler, N., Zaru, R., Micheau, O. et al. Fas triggers an alternative, caspase-8–independent cell death pathway using the kinase RIP as effector molecule. Nat Immunol 1, 489–495 (2000). https://doi.org/10.1038/82732
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/82732
This article is cited by
-
IKKε and TBK1 prevent RIPK1 dependent and independent inflammation
Nature Communications (2024)
-
Immunogenic cell death in cancer: targeting necroptosis to induce antitumour immunity
Nature Reviews Cancer (2024)
-
MLKL polymerization-induced lysosomal membrane permeabilization promotes necroptosis
Cell Death & Differentiation (2024)
-
Necroptosis does not drive disease pathogenesis in a mouse infective model of SARS-CoV-2 in vivo
Cell Death & Disease (2024)
-
RIPK1 polymorphisms and expression levels: impact on genetic susceptibility and clinical outcome of epithelial ovarian cancer
Cancer Cell International (2023)