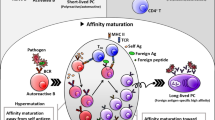
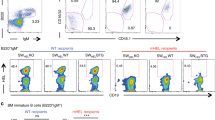
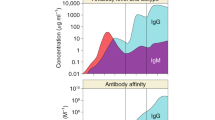
Abstract
Antibodies on the surface of B lymphocytes trigger adaptive immune responses and control a series of antigen-independent checkpoints during B cell development. These physiologic processes are regulated by a complex of membrane immunoglobulin and two signal transducing proteins known as Igα and Igβ. Here we focus on the role of antibodies in governing the maturation of B cells from early antigen-independent through the final antigen-dependent stages.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Landsteiner, K. The Specificity of Serological Reactions (Dover Publications, New York, 1936).
Burnet, F. M. The Clonal Selection Theory of Acquired Immunity (The University Press, Cambridge, 1959).
Talmage, D. W. Clonal selection theory. Science 129, 1643 –1648 (1959).
Hardy, R. R., Carmack, C. E., Shinton, S. A., Kemp, J. D. & Hayakawa, K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J. Exp. Med. 172, 1213–1225 (1991).
Allman, D., Li, J. & Hardy, R. R. Commitment to the B lymphoid lineage occurs before D H-JH recombination. J. Exp. Med. 189 , 735–740 (1999).
Schatz, D. G., Oettinger, M. A. & Baltimore, D. The V(D)J recombination activating gene, RAG-1. Cell 59, 1035–1048 ( 1989).
Oettinger, M. A., Schatz, D. G., Gorka, C. & Baltimore, D. RAG-1 and RAG-2, adjacent genes that synergistically activate V(D)J recombination. Science 248, 1517–1523 ( 1990).
Li, Y. -S., Hayakawa, K. & Hardy, R. R. The regulated expression of B lineage associated genes during B cell differentiation in bone marrow and fetal liver. J. Exp. Med. 178, 951–960 (1993).
O'Riordan, M. & Grosschedl, R. Transcriptional regulation of early B-lymphocyte differentiation. Immunol. Rev. 175 , 94–103 (2000).
Nutt, S. L., Heavey, B., Rolink, A. G. & Busslinger, M. Commitment to the B-lymphoid lineage depends on the transcription factor Pax5 . Nature 401, 556–562 (1999).
Rolink, A. G., Nutt, S. L., Melchers, F. & Busslinger, M. Long-term in vivo reconstitution of T-cell development by Pax5-deficient B-cell progenitors. Nature 401, 603– 606 (1999).
Hombach, J., Leclercq, L., Radbruch, A., Rajewsky, K. & Reth, M. A novel 34-kd protein co-isolated with the IgM molecule in surface IgM-expressing cells. EMBO J. 7, 3451–3456 (1988).
Hermanson, G. G., Eisenberg, D., Kincade, P. W. & Wall, R. B29: a member of the immunoglobulin gene superfamily exclusively expressed on B-lineage cells. Proc. Natl. Acad. Sci. USA 85, 6890–6894 (1988).
Nagata, K. et al. The Ig α/Igβ heterodimer on mu-negative proB cells is competent for transducing signals to induce early B cell differentiation . Immunity 7, 559–570 (1997).
Reth, M. Antigen receptor tail clue. Nature 338, 383–384 (1989).
Burkhardt, A. L., Brunswick, M., Bolen, J. B. & Mond, J. J. Anti-immunoglobulin stimulation of B lymphocytes activates src-related protein-tyrosine kinases. Proc. Natl Acad. Sci. USA 88, 7410 –7414 (1991).
Sanchez, M. et al. Signal transduction by immunoglobulin is mediated through IgA and IgB. J. Exp. Med. 178, 1049– 1056 (1993).
Kurosaki, T. et al. Syk activation by the Src-family tyrosine kinase in the B cell receptor. J. Exp. Med. 179, 1725– 1729 (1994).
Alt, F. W. et al. Ordered rearrangements of immunoglobulin heavy chain variable region segments. EMBO J. 3, 1209– 1219 (1984).
Ehlich, A., Martin, V., Muller, W. & Rajewsky, K. Analysis of the B cell progenitor compartment at the level of single cells. Curr. Biol. 4, 573–583 ( 1994).
ten Boekel, E., Melchers, F. & Rolink, A. The status of Ig loci rearrangements in single cells from different stages of B cell development. Int. Immunol. 7, 1013–1019 (1995).
Kaartinen, M. & Makela, O. Reading of D genes in variable frames as a source of antibody diversity. Immunol. Today 6 , 324–330 (1985).
Meek, K. Analysis of junctional diversity during B lymphocyte development. Science 250, 820–823 ( 1990).
Melchers, F. Fit for life in the immune system? Surrogate L chain tests H chains that test L chains. Proc. Natl Acad. Sci. USA 96, 2571–2573 (1999).
Reth, M. G. & Alt, F. W. Novel immunoglobulin heavy chains are produced from DJH gene segment rearrangements in lymphoid cells. Nature 312, 418–423 ( 1984).
Horne, M., Roth, P. E. & DeFranco, A. L. Assembly of the truncated immunoglobulin heavy chain dμ into antigen receptor-like complexes in pre-B cells but not in B cells . Immunity 4, 145– 158 (1996).
Gu, H., Kitamura, D. & Rajewsky, K. B cell development regulated by gene rearrangement: arrest of maturation by membrane-bound Dμ protein and selection of D H element reading frames. Cell 65, 47–54 (1991).
Tornberg, U. C., Bergqvist, I., Haury, M. & Holmberg, D. Regulation of B lymphocyte development by the truncated immunoglobulin heavy chain protein Dμ. J. Exp. Med. 187, 703– 709 (1998).
Reth, M. G., Jackson, S. & Alt, F. W. VHDJH formation and DJH replacement during pre-B differentiation: non-random usage of gene segments . EMBO J. 5, 2131–2138 (1986).
Gong, S., Sanchez, M. & Nussenzweig, M.C. Counterselection against Dμ is mediated through immunoglobulin (Ig)α-Igβ. J. Exp. Med. 184 , 2079–2084 (1996).
Tonegawa, S. Somatic generation of antibody diversity. Nature 302 , 575 (1983).
Nutt, S. L., Urbanek, P., Rolink, A. & Busslinger, M. Essential functions of Pax5 (BSAP) in pro-B cell development: difference between fetal and adult B lymphopoiesis and reduced V-to-DJ recombination at the IgH locus. Genes Dev. 11, 476–491 ( 1997).
Corcoran, A. E., Riddell, A., Krooshoop, D. & Venkitaraman, A. R. Impaired immunoglobulin gene rearrangement in mice lacking the IL-7 receptor . Nature 391, 904–907 (1998).
Gong, S. & Nussenzweig, M. C. Regulation of an early developmental checkpoint in the B cell pathway by Igβ. Science 272, 411–414 (1996).
Yancopoulos, G. D. & Alt, F. W. Developmentally controlled and tissue-specific expression of unrearranged VH gene segments. Cell 40, 271– 281 (1985).
Alt, F. W., Blackwell, T. K. & Yancopoulos, G. D. Development of the primary antibody repertoire. Science 238, 1079–1087 ( 1987).
Jenuwein, T. et al. Extension of chromatin accessibility by nuclear matrix attachment regions. Nature 385, 269– 272 (1997).
Pillai, S. & Baltimore, D. Formation of disulfide-linked m 2w 2 tetramers in pre-B cells by the 18k w-immunoglobulin light chain. Nature 329, 172–174 ( 1987).
Kerr, W. G., Cooper, M. D., Feng, L., Burrows, P. D. & Hendershot, L. M. μ heavy chains can associate with a pseudo-light chain complex (yL) in human pre-B cell lines. Int. Immunol. 1, 355–361 (1989).
Tsubata, T. & Reth, M. The products of pre-B cell-specific genes (λ5 and VpreB) and the immunoglobulin μ chain form a complex that is transported onto the cell surface. J. Exp. Med. 172, 973–976 (1990).
Karasuyama, H., Kudo, A. & Melchers, F. The proteins encoded by the VpreB and λ5 pre-B cell-specific genes can associate with each other and with μ heavy chain . J. Exp. Med. 172, 969– 972 (1990).
Nossal, G. J. V. & Lederberg, J. Antibody production by single cells. Nature 181, 1419– 1420 (1958).
Pernis, B. G., Chiappino, G., Kelus, A. S. & Gell, P. G. H. Cellular localization of immunoglobulins with different allotypes specificities in rabbit lymphoid tissue. J. Exp. Med. 122, 853–875 (1965).
Coleclough, C., Perry, R. P., Karjalainen, K. & Weigert, M. Aberrant rearrangements contribute significantly to the allelic exclusion of immunoglobulin gene expression. Nature 290, 372–378 (1981).
Weaver, D., Costantini, F., Imanishi, K. T. & Baltimore, D. A transgenic immunoglobulin μ gene prevents rearrangement of endogenous genes. Cell 42, 117–127 (1985).
Rusconi, S. & Kohler, G. Transmission and expression of a specific pair of rearranged immunoglobulin μ and κ genes in a transgenic mouse line. Nature 314, 330– 334 (1985).
Nussenzweig, M. C. et al. Allelic exclusion in transgenic mice that express the membrane form of immunoglobulin M. Science 236, 816 –819 (1987).
Manz, J., Denis, K., Witte, O., Brinster, R. & Storb, U. Feedback inhibition of immunoglobulin gene rearrangement by membrane μ, but not by secreted μ heavy chains. J. Exp. Med. 168, 1363–1381 ( 1988). [Erratum in J. Exp. Med. 169, 2269 (1989)].
Costa, T. E. F., Suh, H. & Nussenzweig, M. C. Chromosomal position of rearranging gene segments influences allelic exclusion in transgenic mice. Proc. Natl Acad. Sci. USA 89, 2205–2208 ( 1992).
Kitamura, D., Roes, J., Kuhn, R. & Rajewsky, K. A B cell deficient mouse by targeted disruption of the membrane exons of the immunoglobulin μ chain gene. Nature 350, 423– 426 (1991).
Nussenzweig, M. C. et al. A human immunoglobulin gene reduces the incidence of lymphomas in c-Myc-bearing transgenic mice. Nature 336, 446–450 (1988).
Yel, L. et al. Mutations in the mu heavy-chain gene in patients with agammaglobulinemia . N. Engl. J. Med. 335, 1486– 1493 (1996).
Schiff, C., Lemmers, B., Deville, A., Fougereau, M. & Meffre, E. Autosomal primary immunodeficiencies affecting human bone marrow B cell differentiation. Immunol. Rev. (in the press, 2000).
Minegishi, Y. et al. Mutations in IgA (CD79a) result in a complete block in B-cell development. J. Clin Invest 104, 1115– 1121 (1999).
Rolink, A., Kudo, A., Karasuyama, H., Kikuchi, Y. & Melchers, F. Long-term proliferating early pre B cell lines and clones with the potential to develop to surface Ig-positive, mitogen reactive B cells in vitro and in vivo. EMBO J. 10, 327–336 (1991).
Kitamura, D. et al. A critical role of l5 protein in B cell development. Cell 69, 823–831 ( 1992).
Rolink, A. et al. B cell development in mice with a defective λ5 gene . Eur J. Immunol. 23, 1284– 1288 (1993).
Löffert, D., Ehlich, A., Muller, W. & Rajewsky, K. Surrogate light chain expression is required to establish immunoglobulin heavy chain allelic exclusion during early B cell development. Immunity 4, 133–144 (1996).
ten Boekel, E., Melchers, F. & Rolink, A.G. Precursor B cells showing H chain allelic inclusion display allelic exclusion at the level of pre-B cell receptor surface expression . Immunity 8, 199–207 (1998).
Ehlich, A. et al. Immunoglobulin heavy and light chain genes rearrange independently at early stages of B cell development. Cell 72, 695–704 (1993).
Chen, J. et al. Immunoglobulin gene rearrangement in B cell deficient mice generated by targeted deletion of the JH locus. Int. Immunol. 5, 647–656 ( 1993).
Ghia, P. et al. Ordering of human bone marrow B lymphocyte precursors by single-cell polymerase chain reaction analyses of the rearrangement status of the immunoglobulin H and L chain loci. J. Exp. Med. 184, 2217 –2229 (1996).
Novobrantseva, T. I. et al. Rearrangement and expression of immunoglobulin light chain genes can precede heavy chain expression during normal B cell development in mice. J. Exp. Med. 189, 75– 88 (1999).
Papavasiliou, F., Jankovic, M. & Nussenzweig, M. C. Surrogate or conventional light chains are required for membrane immunoglobulin μ to activate the precursor B cell transition . J. Exp. Med. 184, 2025– 2029 (1996).
Pelanda, R., Schaal, S., Torres, R. M. & Rajewsky, K. A prematurely expressed Ig(κ) transgene, but not V(κ)J(κ) gene segment targeted into the Ig(κ) locus, can rescue B cell development in λ5-deficient mice. Immunity 5, 229–239 (1996).
Corcos, D. et al. Pre-B-cell development in the absence of lambda 5 in transgenic mice expressing a heavy-chain disease protein. Curr. Biol. 5, 1140–1148 (1995).
Shaffer, A. L. & Schlissel, M. S. A truncated heavy chain protein relieves the requirement for surrogate light chains in early B cell development. J. Immunol. 159, 1265–1275 (1997).
Papavasiliou, F., Misulovin, Z., Suh, H. & Nussenzweig, M. C. The role of Igb in precursor B cell transition and allelic exclusion. Science 268, 408–411 ( 1995).
Hibbs, M. L. et al. Multiple defects in the immune system of Lyn-deficient mice, culminating in autoimmune disease. Cell 83, 301–311 (1995).
Nishizumi, H. et al. Impaired proliferation of peripheral B cells and indication of autoimmune disease in lyn-deficient mice. Immunity 3, 549–560 (1995).
Appleby, M. W. et al. Involvement of p59fynT in interleukin-5 receptor signaling . J. Exp. Med. 182, 811– 820 (1995).
Texido, G. et al. The B-cell-specific Src-family kinase Blk is dispensable for B-cell development and activation. Mol. Cell. Biol. 20, 1227–1233 (2000).
Turner, M. et al. Perinatal lethality and blocked B-cell development in mice lacking the tyrosine kinase Syk. Nature 378, 298–302 (1995).
Cheng, A. M. et al. Syk tyrosine kinase required for mouse viability and B-cell development. Nature 378, 303– 306 (1995).
Pappu, R. et al. Requirement for B cell linker protein (BLNK) in B cell development . Science 286, 1949–1954 (1999).
Minegishi, Y. et al. An essential role for BLNK in human B cell development. Science 286, 1954–197 ( 1999).
Jumaa, H. et al. Abnormal development and function of B lymphocytes in mice deficient for the signaling adaptor protein SLP-65. Immunity 11, 547–554 (1999).
Fruman, D. A. et al. Impaired B cell development and proliferation in absence of phosphoinositide 3-kinase p85α. Science 283, 393–397 (1999).
Grawunder, U. et al. Down-regulation of RAG1 and RAG2 gene expression in preB cells after functional immunoglobulin heavy chain rearrangement. Immunity 3, 601–608 ( 1995).
Stanhope-Baker, P., Hudson, K. M., Shaffer, A. L., Constantinescu, A. & Schissel, M. S. Cell type-specific chromatin structure determines to targeting of V(D)J recombinase activity in vitro. Cell 85, 1– 20 (1996).
Constantinescu, A. & Schlissel, M. S. Changes in locus-specific V(D)J recombinase activity induced by immunoglobulin gene products during B cell development. J. Exp. Med. 185, 609–620 (1997).
Perlmutter, R. M., Kearney, J. F., Chang, S. P. & Hood, L. E. Developmentally controlled expression of immunoglobulin VH genes . Science 227, 1597–1601 (1985).
Marshall, A. J., Wu, G. E. & Paige, G. J. Frequency of VH81x usage during B cell development: initial decline in usage is independent of Ig heavy chain cell surface expression . J. Immunol. 156, 2077– 2084 (1996).
Yancopoulos, G. D. et al. Preferential utilization of the most JH-proximal VH gene segments in pre-B-cell lines. Nature 311, 727–733 (1984).
Wu, G. E. & Paige, C. J. VH gene family utilization in colonies derived from B and pre-B cells detected by the RNA colony blot assay. EMBO J. 5, 3475– 3481 (1986).
Jenuwein, T., Forrester, W. C., Qiu, R. G. & Grosschedl, R. The immunoglobulin μ enhancer core establishes local factor access in nuclear chromatin independent of transcriptional stimulation. Genes Dev. 7, 2016–2032 ( 1993).
Keyna, U., Beck-Engeser, G. B., Jongstra, J., Applequist, S. E. & Jack, H. M. Surrogate light chain-dependent selection of Ig heavy chain V regions. J. Immunol. 155, 5536–5542 (1995).
ten Boekel, E., Melchers, F. & Rolink, A. G. Changes in the V(H) gene repertoire of developing precursor B lymphocytes in mouse bone marrow mediated by the pre-B cell receptor . Immunity 7, 357–368 (1997).
Shiokawa, S. et al. IgM heavy chain complementarity-determining region 3 diversity is constrained by genetic and somatic mechanisms until two months after birth . J. Immunol. 162, 6060– 6070 (1999).
Wasserman, R. et al. A novel mechanism for B cell repertoire maturation based on response by B cell precursors to pre-B receptor assembly. J. Exp. Med. 187, 259–264 ( 1998).
Reth, M., Petrac, E., Wiese, P., Lobel, L. & Alt, F. W. Activation of Vκ gene rearrangement in pre-B cells follows the expression of membrane-bound immunoglobulin heavy chains. EMBO J. 6, 3299–3305 ( 1987).
Schlissel, M. S. & Baltimore, D. Activation of immunoglobulin κ gene rearrangement correlates with induction of germline κ gene transcription. Cell 58, 1001– 1007 (1989).
Maki, K., Nagata, K., Kitamura, F., Takemori, T. & Karasuyama, H. Immunoglobulin β signaling regulates locus accessibility for ordered immunoglobulin gene rearrangements. J. Exp. Med. 191, 1333–1340 (2000).
Tsukamoto, A., Weissman, I. L. & Hunt, S. V. Allelic exclusion in rat κ immunoglobulin chains: extent of Jk rearrangement in normal B lymphocytes. EMBO J. 3, 975–981 (1984).
Ritchie, K. A., Brinster, R. L. & Storb, U. Allelic exclusion and control of endogenous immunoglobulin gene rearrangement in κ transgenic mice. Nature 312, 517–520 (1984).
Rusconi, S. & Kohler, G. Transmission and expression of a specific pair of rearranged immunoglobulin μ and κ genes in a transgenic mouse line. Nature 314, 330– 334 (1985).
Giachino, C., Padovan, E. & Lanzavecchia, A. κ+λ+ dual receptor B cells are present in the human peripheral repertoire. J. Exp. Med. 181, 1245–1250 (1995).
Bernard, O., Gough, N. M. & Adams, J. M. Plasmacytomas with more than one immunoglobulin κ mRNA: implications for allelic exclusion. Proc. Natl Acad. Sci. USA 78, 5812–5816 ( 1981).
Kwan, S. P., Max, E. E., Seidman, J. G., Leder, P. & Scharff, M. D. Two κ immunoglobulin genes are expressed in the myeloma S107. Cell 26, 57–66 (1981).
Yamagami, T., Boekel, E., Andersson, J., Rolink, A. & Melchers, F. Frequencies of multiple Ig L chain gene rearrangements in single normal or κ L chain-deficient B lineage cells. Immunity 11, 317– 327 (1999).
Berinstein, N., Levy, S. & Levy, R. Activation of an excluded immunoglobulin allele in a human B lymphoma cell line. Science 244, 337– 339 (1989).
Rassenti, L. Z., Pratt, L. F., Chen, P. P., Carson, D. A. & Kipps, T. J. Autoantibody-encoding κ L chain genes frequently rearranged in λ L chain-expressing chronic lymphocytic leukemia. J. Immunol. 147, 1060– 1066 (1991).
Mostoslavsky, R. et al. κ chain monoallelic demethylation and the establishment of allelic exclusion. Genes Dev. 12, 1801 –1811 (1998).
Hsieh, C. L. & Lieber, M. R. CpG methylated minichromosomes become inaccessible for V(D)J recombination after undergoing replication. EMBO J. 11, 315–325 ( 1992).
Cherry, S. R. & Baltimore, D. Chromatin remodeling directly activates V(D)J recombination. Proc. Natl Acad. Sci. USA 96, 10788–10793 ( 1999).
Yu, W. et al. Continued RAG expression in late stages of B cell development and no apparent reinduction after immunization. Nature 400, 682–687 (1999).
Monroe, R. et al. RAG2: GFP knockin mice reveal novel aspects of RAG2 expression in primary and peripheral lymphoid tissues. Immunity 11, 201–212 (1999).
Tiegs, S. L., Russell, D. M. & Nemazee, D. Receptor editing in self-reactive bone marrow B cells . J. Exp. Med. 177, 1009– 1020 (1993).
Yamagami, T. et al. Four of five RAG-expressing JCκ−/− small pre-BII cells have no L chain gene rearrangements: detection by high-efficiency single cell PCR. Immunity 11, 309–316 (1999).
Torres, R. M., Flaswinkel, H., Reth, M. & Rajewsky, K. Aberrant B Cell Development and immune response in mice with a compromised BCR complex. Science 272, 1804–1808 ( 1996).
Feddersen, R. M. & Van Ness, B. G. Double recombination of a single immunoglobulin κ-chain allele: implications for the mechanism of rearrangement. Proc. Natl Acad. Sci. USA 82, 4793–4797 (1985).
Kleinfield, R. et al. Recombination between an expressed immunoglobulin heavy-chain gene and a germline variable gene segment in a Ly 1+ B-cell lymphoma. Nature 322, 843– 846 (1986).
Levy, S., Campbell, M. J. & Levy, R. Functional immunoglobulin light chain genes are replaced by ongoing rearrangements of germline Vκ genes to downstream Jκ segment in a murine B cell line. J. Exp. Med. 170, 1–13 (1989).
Feddersen, R. M., Martin, D. J. & Van Ness, B. G. Novel recombinations of the IG κ-locus that result in allelic exclusion. J. Immunol. 145, 745–750 (1990).
Harada, K. & Yamagishi, H. Lack of feedback inhibition of Vκ gene rearrangement by productively rearranged alleles. J. Exp. Med. 173, 409–415 (1991).
Reth, M., Gehrmann, P., Petrac, E. & Wiese, P. A novel VH to VHDJH joining mechanism in heavy-chain-negative (null) pre-B cells results in heavy-chain production. Nature 322, 840–842 (1986).
Gay, D., Saunders, T., Camper, S. & Weigert, M. Receptor editing: an approach by autoreactive B cells to escape tolerance. J. Exp. Med. 177, 999–1008 ( 1993).
Radic, M. Z., Erickson, J., Litwin, S. & Weigert, M. B lymphocytes may escape tolerance by revising their antigen receptors. J. Exp. Med. 177, 1165–1173 ( 1993).
Hertz, M. & Nemazee, D. BCR ligation induces receptor editing in IgM+IgD− bone marrow B cells in vitro. Immunity 6, 429– 436 (1997).
Melamed, D. & Nemazee, D. Self-antigen does not accelerate immature B cell apoptosis, but stimulates receptor editing as a consequence of developmental arrest. Proc. Natl Acad. Sci. USA 94, 9267–72 (1997).
Melamed, D., Benschop, R. J., Cambier, J. C. & Nemazee, D. Developmental regulation of B lymphocyte immune tolerance compartmentalizes clonal selection from receptor selection. Cell 92, 173–182 (1998).
Nemazee, D. A. & Bürki, K. Clonal deletion of B lymphocytes in a transgenic mouse bearing anti-MHC class I antibody genes . Nature 337, 562–566 (1989).
Hartley, S. B. et al. Elimination of self-reactive B lymphocytes proceeds in two stages: arrested development and cell death. Cell 72 , 325–335 (1993).
Chen, C. et al. The site and stage of anti-DNA B-cell deletion. Nature 373, 252–255 ( 1995).
Pelanda, R. et al. Receptor editing in a transgenic mouse model: site, efficiency, and role in B cell tolerance and antibody diversification. Immunity 7, 765–775 ( 1997).
Retter, M.W. & Nemazee, D. Receptor editing occurs frequently during normal B cell development. J. Exp. Med. 188, 1231–1238 (1998).
Osmond, D. G. The turnover of B-cell populations. Immunol. Today 14, 34–37 (1993).
Sandel, P. C. & Monroe, J. G. Negative selection of immature B cells by receptor editing or deletion is determined by site of antigen encounter. Immunity 10, 289– 299 (1999).
Lederberg, J. Genes and antibodies. Science 129, 1649– 1653 (1959).
Lawton, A. R., Asofsky, R., Hylton, M. B. & Cooper, M. D. Suppression of immunoglobulin class synthesis in mice. I. Effects of treatment with antibody to μ chain. J. Exp. Med. 135, 277–297 (1972).
Hartley, S. B. et al. Elimination from peripheral lymphoid tissues of self-reactive B lymphocytes recognizing membrane-bound antigens. Nature 353, 765–769 (1991).
Okamoto, M. et al. A transgenic model of autoimmune hemolytic anemia. J. Exp. Med. 175, 71–79 (1992).
Chen, C. et al. The site and stage of anti-DNA B-cell deletion. Nature 373, 252–255 ( 1995).
Pike, B. L., Boyd, A.W. & Nossal, G. J. Clonal anergy: the universally anergic B lymphocyte . Proc. Natl Acad. Sci. USA 79, 2013– 2017 (1982).
Goodnow, C. C. et al. Altered immunoglobulin expression and functional silencing of self-reactive B lymphocytes in transgenic mice. Nature 334, 676–682 (1988).
Erikson, J. et al. Expression of anti-DNA immunoglobulin transgenes in non-autoimmune mice. Nature 349, 331–334 (1991).
Tsao, B. P. et al. B cells are anergic in transgenic mice that express IgM anti-DNA antibodies. Eur J. Immunol. 23, 2332– 2339 (1993).
Fulcher, D. A. & Basten, A. Reduced life span of anergic self-reactive B cells in a double- transgenic model. J. Exp. Med. 179, 125–134 (1994).
Cyster, J. G., Hartley, S. B. & Goodnow, C. C. Competition for follicular niches excludes self-reactive cells from the recirculating B-cell repertoire. Nature 371, 389–395 (1994).
Bell, S. E. & Goodnow, C. C. A selective defect in IgM antigen receptor synthesis and transport causes loss of cell surface IgM expression on tolerant B lymphocytes. EMBO J. 13, 816 –826 (1994).
Prodeus, A. P. et al. A critical role for complement in maintenance of self-tolerance . Immunity 9, 721–731 (1998).
Rathmell, J. C., Townsend, S. E., Xu, J. C., Flavell, R. A. & Goodnow, C. C. Expansion or elimination of B cells in vivo: dual roles for CD40- and Fas (CD95)-ligands modulated by the B cell antigen receptor. Cell 87, 319–329 (1996).
Cyster, J. G. & Goodnow, C. C. Protein tyrosine phosphatase 1C negatively regulates antigen receptor signaling in B lymphocytes and determines thresholds for negative selection. Immunity 2, 13–24 (1995).
Cyster, J. G. et al. Regulation of B-lymphocyte negative and positive selection by tyrosine phosphatase CD45. Nature 381, 325–328 (1996).
Healy, J. I. et al. Different nuclear signals are activated by the B cell receptor during positive versus negative signaling. Immunity 6, 419–428 (1997).
Inaoki, M., Sato, S., Weintraub, B. C., Goodnow, C. C. & Tedder, T. F. CD19-regulated signaling thresholds control peripheral tolerance and autoantibody production in B lymphocytes . J. Exp. Med. 186, 1923– 1931 (1997).
Bolland, S. & Ravetch, J. V. Inhibitory pathways triggered by ITIM-containing receptors. Adv Immunol. 72, 149–177 (1999).
Hardy, R. R., Hayakawa, K., Parks, D. R. & Herzenberg, L. A. Demonstration of B-cell maturation in X-linked immunodeficient mice by simultaneous three-colour immunofluorescence. Nature 306, 270–272 (1983).
Khan, W. N. et al. Defective B cell development and function in Btk-deficient mice. Immunity 3, 283–299 (1995).
Engel, P. et al. Abnormal B lymphocyte development, activation, and differentiation in mice that lack or overexpress the CD19 signal transduction molecule. Immunity 3, 39–50 ( 1995).
Rickert, R. C., Rajewsky, K. & Roes, J. Impairment of T-cell-dependent B-cell responses and B-1 cell development in CD19-deficient mice. Nature 376, 352–355 (1995).
Sato, S. et al. CD22 is both a positive and negative regulator of B lymphocyte antigen receptor signal transduction: altered signaling in CD22-deficient mice. Immunity 5, 551–562 (1996).
O'Keefe, T. L., Williams, G. T., Davies, S. L. & Neuberger, M. S. Hyperresponsive B cells in CD22-deficient mice. Science 274, 798–801 (1996).
Kishihara, K. et al. Normal B lymphocyte development but impaired T cell maturation in CD45-exon6 protein tyrosine phosphatase-deficient mice. Cell 74, 143–156 ( 1993).
Fulop, G., Gordon, J. & Osmond, D. G. Regulation of lymphocyte production in the bone marrow. I. Turnover of small lymphocytes in mice depleted of B lymphocytes by treatment with anti-IgM antibodies. J. Immunol. 130, 644–648 (1983).
Rolink, A. G., Andersson, J. & Melchers, F. Characterization of immature B cells by a novel monoclonal antibody, by turnover and by mitogen reactivity. Eur J. Immunol. 28, 3738–3748 ( 1998).
Meffre, E. et al. Circulating human B cells that express surrogate light chains and edited receptors. Nature Immunol. 1, 207–213 (2000).
Osmond, D. G. Proliferation kinetics and the lifespan of B cells in central and peripheral lymphoid organs. Curr Opin Immunol. 3, 179 –185 (1991).
Forster, I. & Rajewsky, K. The bulk of the peripheral B-cell pool in mice is stable and not rapidly renewed from the bone marrow. Proc. Natl Acad. Sci. USA 87, 4781– 4784 (1990).
Opstelten, D. & Osmond, D. G. Pre-B cells in mouse bone marrow: immunofluorescence stathmokinetic studies of the proliferation of cytoplasmic μ-chain-bearing cells in normal mice. J. Immunol. 131, 2635–2640 ( 1983).
Agenes, F. & Freitas, A. A. Transfer of small resting B cells into immunodeficient hosts results in the selection of a self-renewing activated B cell population. J. Exp. Med. 189, 319 –330 (1999).
Allman, D. M., Ferguson, S. E. & Cancro, M. P. Peripheral B cell maturation. I. Immature peripheral B cells in adults are heat-stable antigenhi and exhibit unique signaling characteristics. J. Immunol. 149, 2533–2540 (1992).
Nagaoka, H., Gonzalez-Aseguinolaza, G., Tsuji, M. & Nussenzweig, M.C. Immunization and infection change the number of recombination activating gene (RAG)-expressing B cells in the periphery by altering immature lymphocyte production. J. Exp. Med. 191, 2113– 2120 (2000).
Gu, H., Tarlinton, D., Muller, W., Rajewsky, K. & Forster, I. Most peripheral B cells in mice are ligand selected. J. Exp. Med. 173, 1357 –1371 (1991).
Levine, M. H. et al. A B-cell receptor-specific selection step governs immature to mature B cell differentiation. Proc. Natl Acad. Sci. USA 97, 2743–2748 (2000).
Lam, K. P., Kuhn, R. & Rajewsky, K. In vivo ablation of surface immunoglobulin on mature B cells by inducible gene targeting results in rapid cell death. Cell 90, 1073–1083 ( 1997).
Loder, F. et al. B cell development in the spleen takes place in discrete steps and is determined by the quality of B cell receptor-derived signals. J. Exp. Med. 190, 75–89 (1999).
Martin, F. & Kearney, J. F. Positive selection from newly formed to marginal zone B cells depends on the rate of clonal production CD19, and btk. Immunity 12, 39– 49 (2000).
Turner, M. et al. Syk tyrosine kinase is required for the positive selection of immature B cells into the recirculating B cell pool. J. Exp. Med. 186, 2013–2021 ( 1997).
Wang, J., Koizumi, T. & Watanabe, T. Altered antigen receptor signaling and impaired Fas-mediated apoptosis of B cells in Lyn-deficient mice. J. Exp. Med. 184, 831–838 (1996).
Chan, V. W., Meng, F., Soriano, P., DeFranco, A. L. & Lowell, C. A. Characterization of the B lymphocyte populations in Lyn-deficient mice and the role of Lyn in signal initiation and down-regulation . Immunity 7, 69–81 (1997).
Tarakhovsky, A. et al. Defective antigen receptor-mediated proliferation of B and T cells in the absence of Vav. Nature 374, 467–470 (1995).
Zhang, R., Alt, F. W., Davidson, L., Orkin, S. H. & Swat, W. Defective signalling through the T- and B-cell antigen receptors in lymphoid cells lacking the vav proto-oncogene. Nature 374, 470–473 ( 1995).
Hikida, M. et al. Reexpression of RAG-1 and RAG-2 genes in activated mature mouse B cells. Science 274, 2092– 2094 (1996).
Han, S., Zheng, B., Schatz, D. G., Spanopoulou, E. & Kelsoe, G. Neoteny in lymphocytes: Rag1 and Rag2 expression in germinal center B cells. Science 274, 2094 –2097 (1996).
Hikida, M., Mori, M., Kawabata, T., Takai, T. & Ohmori, H. Characterization of B cells expressing Recombination Activating Genes in germinal centers of immunized mouse lymph nodes. J. Immunol.. 158, 2509–2512 ( 1997).
Han, S. et al. V(D)J recombinase activity in a subset of germinal center B lymphocytes . Science 278, 301–305 (1997).
Papavasiliou, F. et al. V(D)J Recombination in mature B cells a new mechanism for diversification of antibody responses. Science 278, 298–301 (1997).
Hertz, M., Kouskoff, V., Nakamura, T. & Nemazee, D. V(D)J recombinase induction in splenic B lymphocytes is inhibited by antigen-receptor signalling. Nature 394, 292– 295 (1998).
Meffre, E. et al. Antigen receptor engagement turns off the V(D)J recombination machinery in human tonsil B cells. J. Exp. Med. 188 , 765–772 (1998).
Hayakawa, K., Hardy, R. R., Honda, M., Herzenberg, L. A. & Steinberg, A.D. Ly-1 B cells: functionally distinct lymphocytes that secrete IgM autoantibodies. Proc. Natl Acad. Sci. USA 81, 2494–2498 (1984).
Hardy, R. R., Hayakawa, K., Shimizu, M., Yamasaki, K. & Kishimoto, T. Rheumatoid factor secretion from human Leu-1+ B cells. Science 236, 81–83 (1987).
Mercolino, T. J., Arnold, L. W., Hawkins, L. A. & Haughton, G. Normal mouse peritoneum contains a large population of Ly-1+ (CD5) B cells that recognize phosphatidyl choline. Relationship to cells that secrete hemolytic antibody specific for autologous erythrocytes. J. Exp. Med. 168, 687–698 (1988).
Pennell, C. A. et al. Biased immunoglobulin variable region gene expression by Ly-1 B cells due to clonal selection. Eur J. Immunol. 19 , 1289–1295 (1989).
Arnold, L. W., Pennell, C. A., McCray, S. K. & Clarke, S. H. Development of B-1 cells: segregation of phosphatidyl choline-specific B cells to the B-1 population occurs after immunoglobulin gene expression. J. Exp. Med. 179, 1585–1595 (1994).
Watanabe, N. et al. Expression levels of B cell surface immunoglobulin regulate efficiency of allelic exclusion and size of autoreactive B-1 cell compartment . J. Exp. Med. 190, 461– 469 (1999).
Hayakawa, K. et al. Positive selection of natural autoreactive B cells. Science 285, 113–116 ( 1999).
Lam, K. P. & Rajewsky, K. B cell antigen receptor specificity and surface density together determine B-1 versus B-2 cell development . J. Exp. Med. 190, 471– 477 (1999).
Ahearn, J. M. et al. Disruption of the Cr2 locus results in a reduction in B-1a cells and in an impaired B cell response to T-dependent antigen. Immunity 4, 251–262 ( 1996).
Kerner, J. D. et al. Impaired expansion of mouse B cell progenitors lacking Btk . Immunity 3, 301–312 (1995).
Leitges, M. et al. Immunodeficiency in protein kinase cβ-deficient mice . Science 273, 788–791 (1996).
Wang, D. et al. Phospholipase Cγ2 is essential in the functions of B cell and several Fc receptors. Immunity 13, 25 –35 (2000).
Sidman, C. L., Shultz, L. D., Hardy, R. R., Hayakawa, K. & Herzenberg, L. A. Production of immunoglobulin isotypes by Ly-1+ B cells in viable motheaten and normal mice . Science 232, 1423–1425 (1986).
Shultz, L. D. et al. Mutations at the murine motheaten locus are within the hematopoietic cell protein-tyrosine phosphatase (Hcph) gene. Cell 73, 1445–1454 (1993).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meffre, E., Casellas, R. & Nussenzweig, M. Antibody regulation of B cell development. Nat Immunol 1, 379–385 (2000). https://doi.org/10.1038/80816
Issue Date:
DOI: https://doi.org/10.1038/80816
This article is cited by
-
STmut: a framework for visualizing somatic alterations in spatial transcriptomics data of cancer
Genome Biology (2023)
-
Deep thermal profiling for detection of functional proteoform groups
Nature Chemical Biology (2023)
-
Evolution of Anti-B Cell Therapeutics in Autoimmune Neurological Diseases
Neurotherapeutics (2022)
-
Function and dysfunction of plasma cells in intestine
Cell & Bioscience (2019)
-
Essential role of the initial activation signal in isotype selection upon deletion of a transcriptionally committed promoter
Scientific Reports (2019)