Abstract
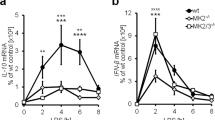
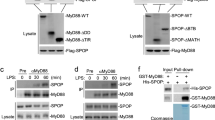
Interleukin 1 receptor (IL-1R) and Toll-like receptors (TLRs) induce inflammatory genes through the complex of MyD88, IL-1R-associated protein kinase (IRAK) and tumor necrosis factor receptor–associated factor 6 (TRAF6), which is believed to function 'upstream' of the cascades of IκB kinase (IKK) and nuclear factor-κB (NF-κB); extracellular signal-regulated protein kinase (ERK); c-Jun N-terminal kinase (JNK); and p38 mitogen-activated protein kinase (MAPK). Here we show that MAPK–ERK kinase kinase (MEKK3) is an essential signal transducer of the MyD88–IRAK–TRAF6 complex in IL-1R–TLR4 signaling. MEKK3 forms a complex with TRAF6 in response to IL-1 and lipopolysaccharide (LPS) but not CpG, and is required for IL-1R- and TLR4-induced IL-6 production. Furthermore, MEKK3 is crucial for IL-1- and LPS-induced activation of NF-κB and JNK-p38 but not ERK, indicating that MAPKs are differentially activated during IL-1R–TLR4 signaling. These data demonstrate that MEKK3 is crucial for IL-1R and TLR4 signaling through the IKK–NF-κB and JNK–p38 MAPK pathways.
*Note: In the version of this article originally published online, the third author's name was incorrect. The correct author name should be Yong Lin. This error has been corrected for the HTML and print versions of this article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
15 December 2003
appended aop PDF with corrigendum (will be corrected for print issue), and placed footnote in SGML at abstract
References
Dunne, A. & O'Neill, L.A. The interleukin-1 receptor/Toll-like receptor superfamily: signal transduction during inflammation and host defense. Sci. STKE 2003, re3 (2003).
Takeda, K., Kaisho, T. & Akira, S. Toll-like receptors. Annu. Rev. Immunol. 21, 335–376 (2003).
Akira, S. & Hemmi, H. Recognition of pathogen-associated molecular patterns by TLR family. Immunol. Lett. 85, 85–95 (2003).
Medzhitov, R. Toll-like receptors and innate immunity. Nat. Rev. Immunol. 1, 135–145 (2001).
Medzhitov, R. & Janeway, C. Jr. Innate immune recognition: mechanisms and pathways. Immunol. Rev. 173, 89–97 (2000).
Takeuchi, O. & Akira, S. Toll-like receptors; their physiological role and signal transduction system. Int. Immunopharmacol. 1, 625–635 (2001).
Takeuchi, O. & Akira, S. MyD88 as a bottle neck in Toll/IL-1 signaling. Curr. Top Microbiol. Immunol. 270, 155–167 (2002).
Janeway, C.A. Jr. & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
Yamamoto, M. et al. Cutting edge: a novel Toll/IL-1 receptor domain-containing adapter that preferentially activates the IFN-b promoter in the Toll-like receptor signaling. J. Immunol. 169, 6668–6672 (2002).
Yamamoto, M. et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 301, 640–643 (2003).
Oshiumi, H., Matsumoto, M., Funami, K., Akazawa, T. & Seya, T. TICAM-1, an adaptor molecule that participates in Toll-like receptor 3-mediated interferon-b induction. Nat. Immunol. 4, 161–167 (2003).
Hoebe, K. et al. Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature 424, 743–748 (2003).
Ninomiya-Tsuji, J. et al. The kinase TAK1 can activate the NIK-IkB as well as the MAP kinase cascade in the IL-1 signalling pathway. Nature 398, 252–256 (1999).
Wang, C. et al. TAK1 is a ubiquitin-dependent kinase of MKK and IKK. Nature 412, 346–351 (2001).
Kopp, E. et al. ECSIT is an evolutionarily conserved intermediate in the Toll/IL-1 signal transduction pathway. Genes Dev. 13, 2059–2071 (1999).
Lee, F.S., Hagler, J., Chen, Z.J. & Maniatis, T. Activation of the IkBa kinase complex by MEKK1, a kinase of the JNK pathway. Cell 88, 213–222 (1997).
Baud, V. et al. Signaling by proinflammatory cytokines: oligomerization of TRAF2 and TRAF6 is sufficient for JNK and IKK activation and target gene induction via an amino-terminal effector domain. Genes Dev. 13, 1297–1308 (1999).
Zhao, Q. & Lee, F.S. Mitogen-activated protein kinase/ERK kinase kinases 2 and 3 activate nuclear factor-kB through IkB kinase-a and IkB kinase-b. J. Biol. Chem. 274, 8355–8358 (1999).
Yang, J. et al. The essential role of MEKK3 in TNF-induced NF-kB activation. Nat. Immunol. 2, 620–624 (2001).
Malinin, N.L., Boldin, M.P., Kovalenko, A.V. & Wallach, D. MAP3K-related kinase involved in NF-kB induction by TNF, CD95 and IL-1. Nature 385, 540–544 (1997).
Dumitru, C.D. et al. TNF-a induction by LPS is regulated posttranscriptionally via a Tpl2/ERK-dependent pathway. Cell 103, 1071–1083 (2000).
Karin, M. & Ben-Neriah, Y. Phosphorylation meets ubiquitination: the control of NF-kB activity. Annu. Rev. Immunol. 18, 621–663 (2000).
Karin, M. & Delhase, M. JNK or IKK, AP-1 or NF-kB, which are the targets for MEK kinase 1 action? Proc. Natl. Acad. Sci. USA 95, 9067–9069 (1998).
Ghosh, S. & Karin, M. Missing pieces in the NF-kB puzzle. Cell 109, S81–96 (2002).
Xia, Y. et al. MEK kinase 1 is critically required for c-Jun N-terminal kinase activation by proinflammatory stimuli and growth factor-induced cell migration. Proc. Natl. Acad. Sci. USA 97, 5243–5248 (2000).
Shinkura, R. et al. Alymphoplasia is caused by a point mutation in the mouse gene encoding NF-kb-inducing kinase. Nat. Genet. 22, 74–77 (1999).
Yin, L. et al. Defective lymphotoxin-b receptor-induced NF-kB transcriptional activity in NIK-deficient mice. Science 291, 2162–2165 (2001).
Sanjo, H. et al. TAB2 is essential for prevention of apoptosis in fetal liver but not for interleukin-1 signaling. Mol. Cell Biol. 23, 1231–1238 (2003).
Blank, J.L., Gerwins, P., Elliott, E.M., Sather, S. & Johnson, G.L. Molecular cloning of mitogen-activated protein/ERK kinase kinases (MEKK) 2 and 3. Regulation of sequential phosphorylation pathways involving mitogen-activated protein kinase and c-Jun kinase. J. Biol. Chem. 271, 5361–5368 (1996).
Ellinger-Ziegelbauer, H., Brown, K., Kelly, K. & Siebenlist, U. Direct activation of the stress-activated protein kinase (SAPK) and extracellular signal-regulated protein kinase (ERK) pathways by an inducible mitogen-activated protein kinase/ERK kinase kinase 3 (MEKK) derivative. J. Biol. Chem. 272, 2668–2674 (1997).
Su, B., Cheng, J., Yang, J. & Guo, Z. MEKK2 is required for T-cell receptor signals in JNK activation and interleukin-2 gene expression. J. Biol. Chem. 276, 14784–14790 (2001).
Yang, J. et al. Mekk3 is essential for early embryonic cardiovascular development. Nat. Genet. 24, 309–313 (2000).
Guo, Z. et al. Disruption of Mekk2 in mice reveals an unexpected role for MEKK2 in modulating T-cell receptor signal transduction. Mol. Cell Biol. 22, 5761–5768 (2002).
Takaesu, G. et al. TAB2, a novel adaptor protein, mediates activation of TAK1 MAPKKK by linking TAK1 to TRAF6 in the IL-1 signal transduction pathway. Mol. Cell. 5, 649–658 (2000).
Naviaux, R.K., Costanzi, E., Haas, M. & Verma, I.M. The pCL vector system: rapid production of helper-free, high-titer, recombinant retroviruses. J. Virol. 70, 5701–5705 (1996).
Acknowledgements
We thank R. Medzhitov, Y. Liu and Y.J. Liu for suggestions and critically reading the manuscript; M. Goode for editing the manuscript; X. Li for sharing plasmids; and B. Wang for technical assistance. Supported partially by the National Institutes of Health (AI44016 and HL070225) and Texas High Education (ARP; both to B.S.), by the Department of Defense (DAMD17-02-0449; Q.H.) and by a Cancer Center Core grant (CA16672; M.D. Anderson Cancer Center).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Huang, Q., Yang, J., Lin, Y. et al. Differential regulation of interleukin 1 receptor and Toll-like receptor signaling by MEKK3. Nat Immunol 5, 98–103 (2004). https://doi.org/10.1038/ni1014
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1014
This article is cited by
-
Elevated proportion of TLR2- and TLR4-expressing Th17-like cells and activated memory B cells was associated with clinical activity of cerebral cavernous malformations
Journal of Neuroinflammation (2022)
-
The Toll-like receptor ligand, CpG oligodeoxynucleotides, regulate proliferation and osteogenic differentiation of osteoblast
Journal of Orthopaedic Surgery and Research (2020)
-
Dexamethasone sodium phosphate attenuates lipopolysaccharide-induced neuroinflammation in microglia BV2 cells
Naunyn-Schmiedeberg's Archives of Pharmacology (2020)
-
Overexpression of MAP3K3 promotes tumour growth through activation of the NF-κB signalling pathway in ovarian carcinoma
Scientific Reports (2019)
-
MicroRNA-124 regulates the expression of MEKK3 in the inflammatory pathogenesis of Parkinson’s disease
Journal of Neuroinflammation (2018)