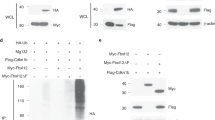
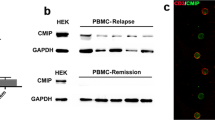
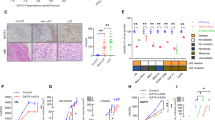
Abstract
The GTPase Rac-1 has the potential for pleiotropic functions due to its ability to interact with multiple effectors. Here, activation of Rac-1 is shown to potently regulate pre-T cell differentiation and proliferation at the point of T cell antigen receptor (TCR) β selection. An activated Rac-1 effector domain mutant that restricts signaling to particular actions on actin dynamics can drive pre-T cell differentiation. Rac-1 activation cannot fully substitute for the pre-TCR complex but can fully correct defects in pre-T cell development in mice lacking the adapter molecule Vav-1. The present study identifies the subset of Rac-1 responses that mediate Vav-1 action as critical regulators of TCRβ selection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Godfrey, D. I., Kennedy, J., Suda, T. & Zlotnik, A. A developmental pathway involving four phenotypically and functionally distinct subsets of CD3−CD4−CD8− triple-negative adult mouse thymocytes defined by CD44 and CD25 expression. J. Immunol. 150, 4244–4252 (1993).
Fehling, H. J. & von Boehmer, H. Early αβ T cell development in the thymus of normal and genetically altered mice. Curr. Opin. Immunol. 9, 263–275 (1997).
von Boehmer, H. et al. Pleiotropic changes controlled by the pre-T-cell receptor. Curr. Opin. Immunol. 11, 135–142 (1999).
Jameson, S. C. & Bevan, M. J. T-cell selection. Curr. Opin. Immunol. 10, 214–219 (1998).
Sebzda, E. et al. Selection of the T cell repertoire. Annu. Rev. Immunol. 17, 829–874 (1999).
Rodewald, H. R. et al. FcγRII/II and CD2 expression mark distinct subpopulations of immature CD4-CD8- murine thymocytes: in vivo developmental kinetics and T cell receptor β chain rearrangement status. J. Exp. Med. 177, 1072–1092 (1993).
Azzam, H. S. et al. CD5 expression is developmentally regulated by T cell receptor (TCR) signals and TCR avidity. J. Exp. Med. 188, 2301–2311 (1998).
Crespo, P., Schuebel, K. E., Ostrom, A. A., Gutkind, J. S. & Bustelo, X. R. Phosphotyrosine-dependent activation of Rac-1 GDP/GTP exchange by the vav proto-oncogene product. Nature 385, 169–172 (1997).
Han, J. et al. Lck regulates Vav activation of members of the Rho family of GTPases. Mol. Cell. Biol. 17, 1346–1353 (1997).
Fischer, K. D. et al. Defective T-cell receptor signaling and positive selection of Vav-deficient CD4+ CD8+ thymocytes. Nature 374, 474–477 (1995).
Tarakhovsky, A. et al. Defective antigen receptor-mediated proliferation of B and T cells in the absence of Vav. Nature 374, 467–470 (1995).
Zhang, R., Alt, F. W., Davidson, L., Orkin, S. H. & Swat, W. Defective signaling through the T- and B-cell antigen receptors in lymphoid cells lacking the vav proto-oncogene. Nature 374, 470–473 (1995).
Turner, M. et al. A requirement for the Rho-family GTP exchange factor Vav in positive and negative selection of thymocytes. Immunity 7, 451–460 (1997).
Acuto, O. & Cantrell, D. T cell activation and the cytoskeleton. Annu. Rev. Immunol. 18, 165–184 (2000).
Dustin, M. L. & Cooper, J. A. The immunological synapse and the actin cytoskeleton: molecular hardware for T cell signaling. Nature Immunol. 1, 23–29 (2000).
Fischer, K. D. et al. Vav is a regulator of cytoskeletal reorganization mediated by the T-cell receptor. Curr. Biol. 8, 554–562 (1998).
Holsinger, L. J. et al. Defects in actin-cap formation in Vav-deficient mice implicate an actin requirement for lymphocyte signal transduction. Curr. Biol. 8, 563–572 (1998).
Costello, P. S. et al. The Rho-family GTP exchange factor Vav is a critical transducer of T cell receptor signals to the calcium, ERK, and NF-kappaB pathways. Proc. Natl Acad. Sci. USA 96, 3035–3040 (1999).
Kuhne, M. R., Ku, G. & Weiss, A. A guanine nucleotide exchange factor-independent function of Vav1 in transcriptional activation. J. Biol. Chem. 275, 2185–2190 (2000).
Villalba, M., Hernandez, J., Deckert, M., Tanaka, Y. & Altman, M. Vav modulation of the Ras/MEK/ERK signaling pathway plays a role in NFAT activation and CD69 up-regulation. Eur. J. Immunol. 30, 1587–1596 (2000).
Genot, E., Cleverley, S., Henning, S. & Cantrell, D. A. Multiple p21ras effector pathways regulate nuclear factor of activated T cells. EMBO J. 15, 3923–3933 (1996).
Genot, E., Reif, K., Beach, S., Kramer, I. & Cantrell, D. p21ras initiates Rac-1 but not phosphatidyl inositol 3 kinase/PKB, mediated signaling pathways in T lymphocytes. Oncogene 17, 1731–1738 (1998).
Genot, E. M. et al. The T-cell receptor regulates Akt (protein kinase B) via a pathway involving Rac1 and phosphatidylinositol 3-kinase. Mol. Cell. Biol. 20, 5469–,5478 (2000).
Burbelo, P. D., Drechsel, D. & Hall, A. A conserved binding motif defines numerous candidate target proteins for both Cdc42 and Rac GTPases. J. Biol. Chem. 270, 29071–29074 (1995).
Bagrodia, S. & Cerione, R. A. Pak to the future. Trends Cell Biol. 9, 350–355 (1999).
Nobes, C. D. & Hall, A. Rho, rac, and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamellipodia, and filopodia. Cell 81, 53–62 (1995).
Hall, A. Rho GTPases and the actin cytoskeleton. Science 279, 509–514 (1998).
Lamarche, N. et al. Rac and Cdc42 induce actin polymerization and G1 cell cycle progression independently of p65PAK and the JNK/SAPK MAP kinase cascade. Cell 87, 519–529 (1996).
Westwick, J. K. et al. Rac regulation of transformation, gene expression, and actin organization by multiple, PAK-independent pathways. Mol. Cell. Biol. 17, 1324–1335 (1997).
Zhumabekov, T., Corbella, P., Tolaini, M. & Kioussis, D. Improved version of a human CD2 minigene based vector for T cell-specific expression in transgenic mice. J. Immunol. Methods 185, 133–140 (1995).
Mombaerts, P. et al. RAG-1-deficient mice have no mature B and T lymphocytes. Cell 68, 869–877 (1992).
Bubeck Wardenburg, J. et al. Regulation of PAK activation and the T cell cytoskeleton by the linker protein SLP-76. Immunity 9, 607–616 (1998).
Swat, W., Shinakai, Y., Cheng, H.-L., Davidson, L. & Alt, F. W. Activated Ras signals differentiation and expansion of CD4+8+ thymocytes. Proc. Natl Acad. Sci. USA 93, 4683–4687 (1996).
Gartner, F. et al. Immature thymocytes employ distinct signaling pathways for allelic exclusion versus differentiation and expansion. Immunity 10, 537–546 (1999).
Ashton-Rickardt, P. G. et al. Evidence for a differential avidity model of T cell selection in the thymus. Cell 76, 651–663 (1994).
Sebzda, E. et al. Positive and negative thymocyte selection induced by different concentrations of a single peptide. Science 263, 1615–1618 (1994).
Evan, G. I., Lewis, G. K., Ramsay, G. & Bishop, J. M. Isolation of monoclonal antibodies specific for human c-Myc proto-oncogene product. Mol. Cell. Biol. 5, 3610–3616 (1985).
Acknowledgements
We thank I. Rosewell for injection of transgenic constructs, and T. Crafton, S. Hoskins, J. Bee and G. Hutchinson for animal care. Supported by the Imperial Cancer Research Fund, European Community Training and Mobility of Researchers Program (to M. G.; ERBFMICT 972245) and the Medical Research Council (to V. T.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gomez, M., Tybulewicz, V. & Cantrell, D. Control of pre-T cell proliferation and differentiation by the GTPase Rac-1. Nat Immunol 1, 348–352 (2000). https://doi.org/10.1038/79808
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/79808
This article is cited by
-
Vav1
AfCS-Nature Molecule Pages (2010)
-
Rho family GTPases and their regulators in lymphocytes
Nature Reviews Immunology (2009)
-
T-cell lymphomas in T-cell-specific Pten-deficient mice originate in the thymus
Leukemia (2008)
-
Lymphocyte calcium signaling from membrane to nucleus
Nature Immunology (2006)
-
Protein kinase C and beyond
Nature Immunology (2004)