Abstract
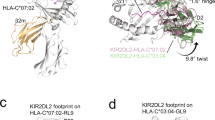
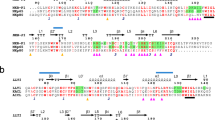
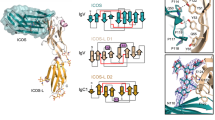
Inhibitory natural killer (NK) cell receptors down-regulate the cytotoxicity of NK cells upon recognition of specific class I major histocompatibility complex (MHC) molecules on target cells. We report here the crystal structure of the inhibitory human killer cell immunoglobulin-like receptor 2DL1 (KIR2DL1) bound to its class I MHC ligand, HLA-Cw4. The KIR2DL1–HLA-Cw4 interface exhibits charge and shape complementarity. Specificity is mediated by a pocket in KIR2DL1 that hosts the Lys80 residue of HLA-Cw4. Many residues conserved in HLA-C and in KIR2DL receptors make different interactions in KIR2DL1–HLA-Cw4 and in a previously reported KIR2DL2–HLA-Cw3 complex. A dimeric aggregate of KIR–HLA-C complexes was observed in one KIR2DL1–HLA-Cw4 crystal. Most of the amino acids that differ between human and chimpanzee KIRs with HLA-C specificities form solvent-accessible clusters outside the KIR-HLA interface, which suggests undiscovered interactions by KIRs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Biron, C. A., Nguyen, K. B., Pien, G. C., Cousens, L. P. & Salazar-Mather, T. P. Natural killer cells in antiviral defense: Function and regulation by innate cytokines. Annu. Rev. Immunol. 17, 189–220 (1999).
Scharton-Kersten, T. M. & Sher, A. Role of natural killer cells in innate resistance to protozoan infections. Curr. Opin. Immunol. 9, 44–51 (1997).
Lanier, L. L. NK cell receptors. Annu. Rev. Immunol. 16, 359–393 (1998).
Khakoo, S. I. et al. Rapid evolution of NK cell receptor systems demonstrated by comparison of chimpanzees and humans. Immunity 12, 687–698 (2000).
Yokoyama, W. M. in Fundamental Immunology. (ed. Paul, W.E.) 575–603 (Lippincott-Raven, Philadelphia, 1999).
Westgaard, I. H., Berg, S. F., Orstavik, S., Fossum, S. & Dissen, E. Identification of a human member of the Ly-49 multigene family. Eur. J. Immunol. 28, 1839–1846 (1998).
Barten, R. & Trowsdale, J. The human Ly-49L gene. Immunogenetics 49, 731–734 (1999).
Long, E. O. & Wagtmann, N. Natural killer cell receptors. Curr. Opin. Immunol. 9, 344–350 (1997).
Sibley, C. G., Comstock, J. A. & Ahlquist, J. E. DNA hybridization evidence of hominoid phylogeny: a reanalysis of the data. J. Mol. Evol. 30, 202–236 (1990).
Colonna, M., Borsellino, G., Falco, M., Ferrara, G. B. & Strominger, J. L. HLA-C is the inhibitory ligand that determines dominant resistance to lysis by NK1- and NK2-specific natural killer cells. Proc. Natl Acad. Sci. USA 90, 12000–12004 (1993).
Moretta, A. et al. p58 molecules as putative receptors for major histocompatibility complex (MHC) class I molecules in human natural killer (NK) cells: anti-p58 antibodies reconstitute lysis of MHC class I-protected cells in NK clones displaying different specificities. J. Exp. Med. 178, 597–604 (1993).
Colonna, M. & Samaridis, J. Cloning of Ig-superfamily members associated with HLA-C and HLA-B recognition by human NK cells. Science 268, 405–408 (1995).
Wagtmann, N. et al. Molecular clones of the p58 natural killer cell receptor reveal Ig-related molecules with diversity in both the extra- and intracellular domains. Immunity 2, 439–449 (1995).
Mandelboim, O. et al. Protection from lysis by natural killer cells of group 1 and 2 specificity is mediated by residue 80 in human histocompatibility leukocyte antigen C alleles and also occurs with empty major histocompatibility complex molecules. J. Exp. Med. 184, 913–922 (1996).
Winter, C. C. & Long, E. O. A single amino acid in the p58 killer cell inhibitory receptor controls the ability of natural killer cells to discriminate between the two groups of HLA-C allotypes. J. Immunol. 158, 4026–4028 (1997).
Boyington, J. C., Motyka, S. A., Schuck, P., Brooks, A. G. & Sun, P. D. Crystal structure of an NK cell immunoglobulin-like receptor in complex with its class I MHC ligand. Nature 405, 537–543 (2000).
Fan, Q. R. et al. Direct binding of a soluble natural killer cell inhibitory receptor to a soluble human leukocyte antigen-Cw4 class I major histocompatibility complex molecule. Proc. Natl Acad. Sci. USA 93, 7178–7183 (1996).
Vales-Gomez, M., Reyburn, H. T., Mandelboim, M. & Strominger, J. L. Kinetics of interaction of HLA-C ligands with natural killer cell inhibitory receptors. Immunity 9, 337–344 (1998).
Maenaka, K. et al. Killer cell immunoglobulin receptors and T cell receptors bind peptide-major histocompatibility complex class I with distinct thermodynamic and kinetic properties. J. Biol. Chem. 274, 28329–28334 (1999).
Fan, Q. R. et al. Structure of the inhibitory receptor for human natural killer cells resembles haematopoietic receptors. Nature 389, 96–100 (1997).
Fan, Q. R. & Wiley, D. C. Structure of human leukocyte antigen (HLA)-Cw4, a ligand for the KIR2D natural killer cell inhibitory receptor. J. Exp. Med. 190, 113–123 (1999).
Rajagopalan, S. & Long, E. O. The direct binding of a p58 killer cell inhibitory receptor to human histocompatibility leukocyte antigen (HLA)-Cw4 exhibits peptide selectivity. J. Exp. Med. 185, 1523–1528 (1997).
Lawrence, M. C. & Coleman, P. M. Shape complementarity at protein/protein interfaces. J. Mol. Biol. 234, 946–950 (1993).
Tormo, J., Natarajan, K., Margulles, D. H. & Mariuzza, R. A. Crystal structure of a lectin-like natural killer cell receptor bound to its MHC class I ligand. Nature 402, 623–631 (1999).
Matsumoto, N., Mitsuki, M., Tajima, K., Yokoyama, W. M. & Yamamoto, K. The functional binding site for the C-type lectin-like natural killer cell receptor Ly49A spans three domains of its major histocompatibility complex class I ligand. J. Exp. Med. 193, 147–158 (2001).
Baker, B. M., Ding, Y. -H., Garboczi, D. N., Biddison, W. E. & Wiley, D. C. Structural, biochemical and biophysical studies of HLA-A2/altered peptide ligands binding to viral-peptide specific human T-cell receptors. Cold Spring Harb. Symp. Quant. Biol. 64, 235–241 (1999).
Garcia, K. C. et al. Structural basis of plasticity in T cell receptor recognition of a self peptide-MHC antigen. Science 279, 1166–1172 (1998).
Garcia, K. C., Teyton, L. & Wilson, I. A. Structural basis of T cell recognition. Annu. Rev. Immunol. 17, 369–397 (1999).
Ysern, X., Li, H. & Mariuzza, R. A. Imperfect interfaces. Nature Struct. Biol. 5, 412–414 (1998).
Reinherz, E. L. et al. The crystal structure of a T cell receptor in complex with peptide and MHC class II. Science 286, 1913–1921 (1999).
Clackson, T. & Wells, J. A. A hot spot of binding energy in a hormone-receptor interface. Science 267, 383 (1995).
Clackson, T., Ultsch, M. H., Wells, J. A. & de Vos, A. M. Structural and functional analysis of the 1:1 growth hormone:receptor complex reveals the molecular basis for receptor affinity. J. Mol. Biol. 277, 1111–1128 (1998).
de Vos, A. M., Ultsch, M. & Kossiakoff, A. A. Human growth hormone and extracellular domain of its receptor: crystal structure of the complex. Science 255, 306–312 (1992).
Somers, W., Ultsch, M., de Vos, A. M. & Kossiakoff, A. A. The X-ray structure of a growth hormone-prolactin receptor complex. Nature 372, 478–481 (1994).
Livnah, O. et al. Funtional mimicry of a protein hormone by a peptide agonist: the EPO receptor complex at 2.8 Å. Science 273, 464–471 (1996).
Snyder, G. A., Brooks, A. G. & Sun, P. D. Crystal structure of the HLA-Cw3 allotype-specific killer cell inhibitory receptor KIR2DL2. Proc. Natl Acad. Sci. USA 96, 3864–3869 (1999).
Maenaka, K., Juji, T., Stuart, D. I. & Jones, E. Y. Crystal structure of the human p58 killer cell inhibitory receptor (KIR2DL3) specific for HLA-Cw3-related MHC class I. Structure 7, 391–398 (1999).
Davis, D. M. et al. The human natural killer cell immune synapse. Proc. Natl Acad. Sci. USA 96, 15062–16067 (1999).
Adams, E. J., Thomson, G. & Parham, P. Evidence for an HLA-C-like locus in the orangutan Pongo pygmaeus. Immunogenetics 49, 865–871 (1999).
Litwin, V., Gumperz, J., Parham, P., Phillips, J. H. & Lanier, L. L. NKB1: a natural killer cell receptor involved in the recognition of polymorphic HLA-B molecules. J. Exp. Med. 180, 537–543 (1994).
Gumperz, J. E., Litwin, V., Phillips, J. H., Lanier, L. L. & Parham, P. The Bw4 public epitope of HLA-B molecules confers reactivity with NK cell clones that express NKB1, a putative HLA receptor. J. Exp. Med. 181, 113–114 (1995).
Wagtmann, N., Rajagopalan, S., Winter, C. C., Peruzzi, M. & Long, E. O. Killer cell inhibitory receptors specific for HLA-C and HLA-B identified by direct binding and by functional transfer. Immunity 3, 801–809 (1995).
Sidney, J. et al. Several HLA alleles share overlapping peptide specificities. J. Immunol. 154, 247–259 (1995).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Meth. Enzymol. 276, 307–326 (1997).
Navaza, J. AMoRe: an automated package for molecular replacement. Acta Crystallogr. A 50, 157–163 (1994).
Brünger, A. T. et al. Crystallography,NMR system: A new softwear suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in the models. Acta Crystallogr. A 47, 110–119 (1991).
Collaborative computational project Number 4. The CCP4 Suite: Programs for protein crystallography. Acta Crystallogr. D 50, 760–776 (1994).
Hendrickson, W. A. Transformations to optimize the superposition of similar structures. Acta Crystallogr. A 35, 158–163 (1979).
Stauber, D. J., DiGabriele, A. D. & Hendrickson, W. A. Structural interactions of fibroblast growth factor receptor with its ligand. Proc. Natl Acad. Sci. USA 97, 49–54 (2000).
Kraulis, P. J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Cryst. 24, 946–950 (1991).
Nicholls, A., Charp, K. A. & Honig, B. Protein folding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins 11, 281–296 (1991).
Carson, M. Ribbon models of macromolecules. J. Mol. Graph 5, 103–106 (1987).
Acknowledgements
We thank Y. Tao for help with data collection, D. N. Garboczi for helpful advice, A. Haykov for inclusion body preparation, N. Siniskaya for peptide and DNA synthesis, D. J. Stauber and W. A. Hendrickson for the program TOSS, C. Watzl for the Lig.1 antibody, I. Vakser for molecular modeling studies, P. Sun for the coordinates of KIR2DL2–HLA-Cw3 complex structure and a preprint of the manuscript, R. Mariuzza for discussion of the Ly49A–H-2Dd complex structure before publication, R. Rajalingam for a preprint of the manuscript on Pygmy Chimpanzee KIR, P. Sliz, K. Mahan, B. Harris and R. Crouse for technical assistance and staff at the X25 beamline of NSLS and F-1 beamline at CHESS for help with data collection. Supported by the Howard Hughes Medical Institute, National Institute of Health and the National Science Foundation (Q. R. F.). D. C. W. is supported by HHMI.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, Q., Long, E. & Wiley, D. Crystal structure of the human natural killer cell inhibitory receptor KIR2DL1–HLA-Cw4 complex. Nat Immunol 2, 452–460 (2001). https://doi.org/10.1038/87766
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/87766
This article is cited by
-
Structural basis for the activity and specificity of the immune checkpoint inhibitor lirilumab
Scientific Reports (2024)
-
Structural plasticity of KIR2DL2 and KIR2DL3 enables altered docking geometries atop HLA-C
Nature Communications (2021)
-
Conservation of sequence motifs suggests that the nonclassical MHC class I lineages CD1/PROCR and UT were established before the emergence of tetrapod species
Immunogenetics (2018)
-
Specificity of inhibitory KIRs enables NK cells to detect changes in an altered peptide environment
Immunogenetics (2018)
-
MHC-I peptides get out of the groove and enable a novel mechanism of HIV-1 escape
Nature Structural & Molecular Biology (2017)