Abstract
Microbial infections often precede the onset of autoimmunity. How infections trigger autoimmunity remains poorly understood. We investigated the possibility that infection might create conditions that allow the stimulatory presentation of self peptides themselves and that this might suffice to elicit autoreactive T cell responses that lead to autoimmunity. Self-reactive CD4+ T cells are major drivers of autoimmune disease, but their activation is normally prevented through regulatory mechanisms that limit the immunostimulatory presentation of self antigens. Here we found that the apoptosis of infected host cells enabled the presentation of self antigens by major histocompatibility complex class II molecules in an inflammatory context. This was sufficient for the generation of an autoreactive TH17 subset of helper T cells, prominently associated with autoimmune disease. Once induced, the self-reactive TH17 cells promoted auto-inflammation and autoantibody generation. Our findings have implications for how infections precipitate autoimmunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cho, J.H. & Gregersen, P.K. Genomics and the multifactorial nature of human autoimmune disease. N. Engl. J. Med. 365, 1612–1623 (2011).
Marrack, P., Kappler, J. & Kotzin, B.L. Autoimmune disease: why and where it occurs. Nat. Med. 7, 899–905 (2001).
Sakaguchi, S., Powrie, F. & Ransohoff, R.M. Re-establishing immunological self-tolerance in autoimmune disease. Nat. Med. 18, 54–58 (2012).
Suurmond, J. & Diamond, B. Autoantibodies in systemic autoimmune diseases: specificity and pathogenicity. J. Clin. Invest. 125, 2194–2202 (2015).
Cho, J.H. & Feldman, M. Heterogeneity of autoimmune diseases: pathophysiologic insights from genetics and implications for new therapies. Nat. Med. 21, 730–738 (2015).
Blander, J.M., Torchinsky, M.B. & Campisi, L. Revisiting the old link between infection and autoimmune disease with commensals and T helper 17 cells. Immunol. Res. 54, 50–68 (2012).
Pordeus, V., Szyper-Kravitz, M., Levy, R.A., Vaz, N.M. & Shoenfeld, Y. Infections and autoimmunity: a panorama. Clin. Rev. Allergy Immunol. 34, 283–299 (2008).
Sfriso, P. et al. Infections and autoimmunity: the multifaceted relationship. J. Leukoc. Biol. 87, 385–395 (2010).
Rosenblum, M.D., Remedios, K.A. & Abbas, A.K. Mechanisms of human autoimmunity. J. Clin. Invest. 125, 2228–2233 (2015).
Root-Bernstein, R. & Fairweather, D. Complexities in the relationship between infection and autoimmunity. Curr. Allergy Asthma Rep. 14, 407 (2014).
Blander, J.M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Nair-Gupta, P. et al. TLR signals induce phagosomal MHC-I delivery from the endosomal recycling compartment to allow cross-presentation. Cell 158, 506–521 (2014).
Torchinsky, M.B., Garaude, J., Martin, A.P. & Blander, J.M. Innate immune recognition of infected apoptotic cells directs TH17 cell differentiation. Nature 458, 78–82 (2009).
Brereton, C.F. & Blander, J.M. The unexpected link between infection-induced apoptosis and a TH17 immune response. J. Leukoc. Biol. 89, 565–576 (2011).
Hirota, K. et al. Fate mapping of IL-17-producing T cells in inflammatory responses. Nat. Immunol. 12, 255–263 (2011).
Mowat, A.M. & Agace, W.W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685 (2014).
Esplugues, E. et al. Control of TH17 cells occurs in the small intestine. Nature 475, 514–518 (2011).
McGeachy, M.J. et al. TGF-β and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell–mediated pathology. Nat. Immunol. 8, 1390–1397 (2007).
Berer, K. et al. Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature 479, 538–541 (2011).
Basu, R. et al. IL-1 signaling modulates activation of STAT transcription factors to antagonize retinoic acid signaling and control the TH17 cell–iTreg cell balance. Nat. Immunol. 16, 286–295 (2015).
Marks, B.R. et al. Thymic self-reactivity selects natural interleukin 17–producing T cells that can regulate peripheral inflammation. Nat. Immunol. 10, 1125–1132 (2009).
Simons, D.M. et al. How specificity for self-peptides shapes the development and function of regulatory T cells. J. Leukoc. Biol. 88, 1099–1107 (2010).
Zehn, D. & Bevan, M.J. T cells with low avidity for a tissue-restricted antigen routinely evade central and peripheral tolerance and cause autoimmunity. Immunity 25, 261–270 (2006).
Malissen, M. et al. Regulation of TCR α and β gene allelic exclusion during T-cell development. Immunol. Today 13, 315–322 (1992).
Padovan, E. et al. Expression of two T cell receptor α chains: dual receptor T cells. Science 262, 422–424 (1993).
Jalkanen, S., Nash, G.S., De los Toyos, J., MacDermott, R.P. & Butcher, E.C. Human lamina propria lymphocytes bear homing receptors and bind selectively to mucosal lymphoid high endothelium. Eur. J. Immunol. 19, 63–68 (1989).
Shimizu, Y., Van Seventer, G.A., Siraganian, R., Wahl, L. & Shaw, S. Dual role of the CD44 molecule in T cell adhesion and activation. J. Immunol. 143, 2457–2463 (1989).
Mandl, J.N., Monteiro, J.P., Vrisekoop, N. & Germain, R.N. T cell-positive selection uses self-ligand binding strength to optimize repertoire recognition of foreign antigens. Immunity 38, 263–274 (2013).
Grossman, Z. & Paul, W.E. Autoreactivity, dynamic tuning and selectivity. Curr. Opin. Immunol. 13, 687–698 (2001).
Atarashi, K. et al. Th17 cell induction by adhesion of microbes to intestinal epithelial cells. Cell 163, 367–380 (2015).
Fonseca, D.M. et al. Microbiota-dependent sequelae of acute infection compromise tissue-specific immunity. Cell 163, 354–366 (2015).
Di Sabatino, A., Lenti, M.V., Giuffrida, P., Vanoli, A. & Corazza, G.R. New insights into immune mechanisms underlying autoimmune diseases of the gastrointestinal tract. Autoimmun. Rev. 14, 1161–1169 (2015).
Sweet, R.A., Lee, S.K. & Vinuesa, C.G. Developing connections amongst key cytokines and dysregulated germinal centers in autoimmunity. Curr. Opin. Immunol. 24, 658–664 (2012).
Richards, D.M., Kyewski, B. & Feuerer, M. Re-examining the nature and function of self-reactive t cells. Trends Immunol. 37, 114–125 (2016).
Hogquist, K.A. & Jameson, S.C. The self-obsession of T cells: how TCR signaling thresholds affect fate 'decisions' and effector function. Nat. Immunol. 15, 815–823 (2014).
Moon, J.J. et al. Quantitative impact of thymic selection on Foxp3+ and Foxp3− subsets of self-peptide/MHC class II-specific CD4+ T cells. Proc. Natl. Acad. Sci. USA 108, 14602–14607 (2011).
Yu, W. et al. Clonal deletion prunes but does not eliminate self-specific αβ CD8+ T lymphocytes. Immunity 42, 929–941 (2015).
Rizzuto, G.A. et al. Self-antigen-specific CD8+ T cell precursor frequency determines the quality of the antitumor immune response. J. Exp. Med. 206, 849–866 (2009).
Walker, L.S. & Abbas, A.K. The enemy within: keeping self-reactive T cells at bay in the periphery. Nat. Rev. Immunol. 2, 11–19 (2002).
Abadie, V., Sollid, L.M., Barreiro, L.B. & Jabri, B. Integration of genetic and immunological insights into a model of celiac disease pathogenesis. Annu. Rev. Immunol. 29, 493–525 (2011).
Vader, W. et al. The HLA-DQ2 gene dose effect in celiac disease is directly related to the magnitude and breadth of gluten-specific T cell responses. Proc. Natl. Acad. Sci. USA 100, 12390–12395 (2003).
Ito, Y. et al. Detection of T cell responses to a ubiquitous cellular protein in autoimmune disease. Science 346, 363–368 (2014).
Klein, L., Kyewski, B., Allen, P.M. & Hogquist, K.A. Positive and negative selection of the T cell repertoire: what thymocytes see (and don't see). Nat. Rev. Immunol. 14, 377–391 (2014).
Burkett, P.R., Meyer zu Horste, G. & Kuchroo, V.K. Pouring fuel on the fire: Th17 cells, the environment, and autoimmunity. J. Clin. Invest. 125, 2211–2219 (2015).
Ghoreschi, K., Laurence, A., Yang, X.P., Hirahara, K. & O'Shea, J.J. T helper 17 cell heterogeneity and pathogenicity in autoimmune disease. Trends Immunol. 32, 395–401 (2011).
Kleinewietfeld, M. & Hafler, D.A. The plasticity of human Treg and Th17 cells and its role in autoimmunity. Semin. Immunol. 25, 305–312 (2013).
Gurung, P. et al. FADD and caspase-8 mediate priming and activation of the canonical and noncanonical Nlrp3 inflammasomes. J. Immunol. 192, 1835–1846 (2014).
Legoux, F.P. et al. CD4+ T cell tolerance to tissue-restricted self antigens is mediated by antigen-specific regulatory T cells rather than deletion. Immunity 43, 896–908 (2015).
Hirota, K. et al. Plasticity of TH17 cells in Peyer's patches is responsible for the induction of T cell-dependent IgA responses. Nat. Immunol. 14, 372–379 (2013).
Kazemi-Shirazi, L. et al. IgA autoreactivity: a feature common to inflammatory bowel and connective tissue diseases. Clin. Exp. Immunol. 128, 102–109 (2002).
Viret, C. & Janeway, C.A. Jr. Functional and phenotypic evidence for presentation of Eα 52-68 structurally related self-peptide(s) in I-Eα-deficient mice. J. Immunol. 164, 4627–4634 (2000).
Nagai, T., Abe, A. & Sasakawa, C. Targeting of enteropathogenic Escherichia coli EspF to host mitochondria is essential for bacterial pathogenesis: critical role of the 16th leucine residue in EspF. J. Biol. Chem. 280, 2998–3011 (2005).
Nougayrède, J.P. & Donnenberg, M.S. Enteropathogenic Escherichia coli EspF is targeted to mitochondria and is required to initiate the mitochondrial death pathway. Cell. Microbiol. 6, 1097–1111 (2004).
Vallance, B.A., Deng, W., Jacobson, K. & Finlay, B.B. Host susceptibility to the attaching and effacing bacterial pathogen Citrobacter rodentium. Infect. Immun. 71, 3443–3453 (2003).
Sham, H.P. et al. Attaching and effacing bacterial effector NleC suppresses epithelial inflammatory responses by inhibiting NF-κB and p38 mitogen-activated protein kinase activation. Infect. Immun. 79, 3552–3562 (2011).
Choi, K.H. et al. A Tn7-based broad-range bacterial cloning and expression system. Nat. Methods 2, 443–448 (2005).
Ferrières, L. et al. Silent mischief: bacteriophage Mu insertions contaminate products of Escherichia coli random mutagenesis performed using suicidal transposon delivery plasmids mobilized by broad-host-range RP4 conjugative machinery. J. Bacteriol. 192, 6418–6427 (2010).
Ding, Y., Shen, S., Lino, A.C., Curotto de Lafaille, M.A. & Lafaille, J.J. β-catenin stabilization extends regulatory T cell survival and induces anergy in nonregulatory T cells. Nat. Med. 14, 162–169 (2008).
Powrie, F., Carlino, J., Leach, M.W., Mauze, S. & Coffman, R.L. A critical role for transforming growth factor-β but not interleukin 4 in the suppression of T helper type 1-mediated colitis by CD45RBlow CD4+ T cells. J. Exp. Med. 183, 2669–2674 (1996).
Acknowledgements
We thank S. Lira, G. Furtado, H. Xiong, G. Yeretssian, M.K. Jenkins and members of the Blander laboratory for discussions; D. Amsen and R.J. Cummings for critical reading of the manuscript; A. Rialdi for help with statistical analyses; J. Ochando and C. Bare for flow cytometry; A. Soto and M.J. Suarez (the NIH Tetramer Core Facility at Emory University) for I-Ab–OVA(328–337) and control tetramers; H. Xiong (The Icahn School of Medicine at Mount Sinai) for LM-OVA bacteria; A. Morelli (University of Pittsburgh) for 1H3.1 mice; B. Finlay and M. Croxen (University of British Columbia) for wild-type and ΔEspF C. rodentium and plasmid pMAC5; H.P. Schweizer (Colorado State University) for plasmid pTNS2; D. Mazel (Institut Pasteur) for MFDpir bacteria; and I. Marazzi, V. Verhasselt, S.E.F. Campisi, G.C. Chiesa, Jr., M.A. Blander, S.J. Blander and the late L. Mayer for advice and support. Supported by the National Institute of Diabetes and Digestive and Kidney Diseases (DK072201 to J.M.B.), the National Institute of Allergy and Infectious Diseases (AI073899, AI080959 and AI095245 to J.M.B.), the Arthritis Foundation (L.C.), the Crohn's and Colitis Foundation of America (G.B.), the Burroughs Wellcome Fund (J.M.B.), the Irma Hirschl and Monique Weill-Caulier Charitable Trust Funds (J.M.B.), the American Cancer Society (J.M.B.) and the Leukemia and Lymphoma Society (J.M.B.).
Author information
Authors and Affiliations
Contributions
L.C. and J.M.B designed and directed the study and wrote the manuscript; L.C. conducted all experiments; G.B. assisted with T cell–sorting experiments; Y.D. conducted the histological and pathological assessments of colonic tissues; E.E. and R.A.F. provided the IL-17–eGFP reporter mice; and J.M.B conceived of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Presentation of cellular antigens from LPS–B cell blasts in vitro and Eα peptide in vivo.
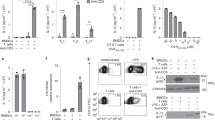
(a) Proliferation of splenic OT-II CD4+ T cells co-cultured for 5 days with BMDCs previously incubated with Eα(52-68) or OVA(329-337) peptides, heat killed (HK) E. coli (EC) bacteria expressing OVA or Eα or apoptotic splenic B cells from Act-mOVA BALB/c mice cultured with or without LPS. Apoptosis was induced by UV irradiation. (b) Schematic showing experimental strategy for assessing proliferation of OT-II and 1H3.1 CD4+ T cells in response to different cargo stimulated bone marrow derived DCs (BMDCs). (c) Schematic showing experimental strategy used in Figure 1b. 5x105 each OT-II and 1H3.1 CD4+ T cells were adoptively transferred together into chimeric actin (Act)-mOVA or wild-type (WT) mice reconstituted with CD11c-DTR BM cells. Mice were infected with WT or ΔEspF Citrobacter and injected with BrdU daily starting at 24 hours post-infection. Some mice received diphtheria toxin (DT) daily starting at 24 hours before infection. (d) Proliferation of splenic Vβ6+CD45.2 CD4+ T cells from 1H3.1 transgenic mice transferred into CD45.1 congenic C57BL/6J mice at day 5 after injection of 109 HK EC-Eα. (a) Data represent 3 independent experiments. (d) n=4 individual mice.
Supplementary Figure 2 The C. rodentium–specific CD4+ T cell response.
Mice were orogastrically infected with C. rodentium (CR) or left untreated. (a-c) and (e) Gated on Aqua−B220−CD45.2+CD3+ CD4+ T cells following ex-vivo stimulation as indicated. (a) Cytokine production and Foxp3 expression in large intestinal lamina propria (LI LP) CD4+ T cells from uninfected and infected C57BL/6J mice. (b) T-bet expression in LI LP and MLN IL-17+ CD4 T cells from infected C57BL/6J mice. (c) Percentages of IL-10+ LI LP CD4+ T cells from uninfected and infected IL-10-eGFP mice after ex-vivo re-stimulation with phorbol 12-myristate 13-acetate (PMA) and ionomycin. (d) Left, Pictures of large intestines from uninfected or WT or ΔEspF CR infected mice. Right, Colony-forming units (CFU) in the stools and colons of infected mice (log10 of CFU normalized by gram of stools or colons). (e) Foxp3, RORγt and T-bet expression by LI LP CD4 T cells from uninfected or day 9 infected mice. Mean and standard deviations (s.d.) are shown; p-values calculated using a t-test. ns=not significant. Data in (a) n=9 mice represent 3 independent experiments, (b) n=4, (c) n=5, (e) n=6 and (d) n=4 (left) and n=9 (right) represent 2 experiments each. Mean and standard deviations (s.d.) are shown; **P=0.0052 (c) and ns=not significant (d) were calculated using a t-test.
Supplementary Figure 3 Characterization of DTg mice.
(a) OT-II were crossed to Act-mOVA mice to generate double transgenic mice (DTg). (b, c) Aqua−B220−CD45+CD3+ MLN and LI LP CD4+ T cells analyzed in uninfected OT-II and DTg mice as indicated. (b) Top, Percentages of Vα2hiCD3+CD4+ T cells. Bottom, Vβ5hi T cells within the Vα2hiCD3+ CD4+ T cells are encircled in blue. (c) Left, I-Ab- OVA(328-337) (HAAHAEINEA) tetramer+ or control unrelated CLIP tetramer+ CD4+ T cells. Tetramer staining MFIs shown below corresponding dot plots. Right, Percentages of self-reactive CD4+ T cells detected with antibodies to Vα2 and Vβ5 or I-Ab-OVA(328-337) tetramer staining. Data in (b, c) n= 6 mice represent 3 experiments. Mean and standard deviations (s.d.) are shown.
Supplementary Figure 4 Intact immune response to C. rodentium in DTg mice.
Indicated mouse strains were infected with WT Citrobacter (a, b) CFU in the stools and colons on days 9 (a) or 30 (b) post-infection (log10 of CFU/ g of stools or colon). (a) Right, Colitis scores in WT and DTg mice on day 9 post-infection. (c) Detection of intracellular cytokines IL-17 and IL-22 in polyclonal Vα2−Vβ5− LI LP CD4+ T cells from DTg (UI and day 9 Inf) and Act-mOVA (Inf) mice after ex vivo restimulation with PMA+I. Gated on Aqua−B220−CD45+CD3+ CD4+ T cells. Data in (a, left) n=9 (OT-II and DTg), n=6 (WT), n=4 (OT-II and DTg, CFU in the colon). Data in (a, right) n= 8 (WT), n=7 (DTg), (b) n=8 (WT and DTg), n=7 (OT-II), n=6 (Act-mOVA) and (c) n=4 individual mice represent 2 experiments each. Mean and standard deviations (s.d.) are shown; NS= not significant calculated using One-way ANOVA test and Dunnet post-test (a, left), t-test (a, right) and One-way ANOVA test and Tukey’s Multiple Comparison post-test (b).
Supplementary Figure 5 Differentiation of self-reactive CD4+ T cells during infection with C. rodentium.
(a) Left, IL-10-eGFP and Foxp3/IL-17 expression in IL-10-eGFP LI LP CD4+ T cells in DTg mice crossed to IL-10-eGFP reporter mice. Right, Percentages of Foxp3+Vα2hiVβ5+ CD4+ T cells in DTg mice. (b) Left, Schematic showing experimental strategy used in Figure 5a: CD45.1+CD45.2+ (CD45.1.2) Act-mOVA chimeric mice reconstituted with BM cells from CD45.1 C57BL/6J and CD45.2+ Tcrα−/− OT-II mice. Right, Percentages of thymic single positive (SP) CD4+ T cells in naïve chimeric mice. R1, R2, R3 represent WT, endogenous, and Tcrα−/− OT-II CD4 T cell populations, respectively. (c) Detection of intracellular IL-17 and Foxp3 at day 9 post-infection with WT Citrobacter in LI LP Vα2hiVβ5+ CD4+ T cells from DTg mice, treated or not with the pan-caspase inhibitor Q-VD-OPH. (a, c) Gated on Aqua−B220−CD45+CD3+ LI LP CD4+ T cells. Data in (a, left and c) n=4 and (b) n=6 represent 2 experiments each, and (a, right) n=8 (uninfected) and n=10 (infected) represent 3 experiments. ***P ≤0.001 calculated using the Mann-Whitney test. Mean and standard deviations (s.d.) are shown.
Supplementary Figure 6 Autoantibody response in DTg mice after infection with C. rodentium.
(a,c) Optical densities (OD) of anti-OVA IgM and IgG1 in the serum of indicated mice on day 40 post-infection. (b) CD45.1+CD45.2+ Act-mOVA chimeric mice reconstituted with BM cells from CD45.1 C57BL/6J and CD45.2 Tcrα−/− OT-II mice and left untreated or infected with WT Citrobacter. (d) Colitis scores on day 40 post-infection in Act-mOVA chimeric mice left untreated or injected with anti-Thy1.1 antibody 24 hours before C. rodentium infection. (a) n=11 (IgM in DTg) and n=8 represent 3 independent experiments; (c, d) n=8 (untreated) and n=7 (treated) represent 2 experiments. Mean and standard deviations (s.d.) are shown. NS=not statistically significant (with P=0.08 for d) and *** P≤ 0.001 calculated using t- test. (e) Model for self-reactive CD4+ T cell activation and differentiation into TH17 cells upon dendritic cell phagocytosis of infected apoptotic host cells. TLR signals from phagosomes carrying infected apoptotic cells lead to MHC class II presentation of both apoptotic cell-derived self and pathogen-derived non-self peptides present within the same phagosome. Activation within the resultant TGF-β and IL-6 rich inflammatory milieu instructs differentiation of both pathogen-specific and self-specific CD4+ T cells into TH17 cells. Self-reactive TH17 cells promote intestinal inflammation and autoantibody production (likely through autoreactive plasma cells).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 1366 kb)
Rights and permissions
About this article
Cite this article
Campisi, L., Barbet, G., Ding, Y. et al. Apoptosis in response to microbial infection induces autoreactive TH17 cells. Nat Immunol 17, 1084–1092 (2016). https://doi.org/10.1038/ni.3512
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3512
This article is cited by
-
The oral microbiome in autoimmune diseases: friend or foe?
Journal of Translational Medicine (2023)
-
Clonally expanded CD8 T cells characterize amyotrophic lateral sclerosis-4
Nature (2022)
-
Gut Microbiota in Lupus: a Butterfly Effect?
Current Rheumatology Reports (2021)
-
Efferocytosis in health and disease
Nature Reviews Immunology (2020)
-
Primary biliary cholangitis: pathogenesis and therapeutic opportunities
Nature Reviews Gastroenterology & Hepatology (2020)