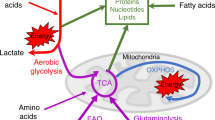
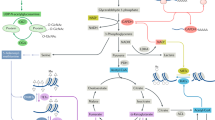
Abstract
Cells of the immune system are auxotrophs for most amino acids, including several nonessential ones. Arginine and tryptophan are used within the regulatory immune networks to control proliferation and function through pathways that actively deplete the amino acid from the microenvironment or that create regulatory molecules such as nitric oxide or kynurenines. How immune cells integrate information about essential amino acid supplies and then transfer these signals to growth and activation pathways remains unclear but has potential for pathway discovery about amino sensing. In applied research, strategies to harness amino acid auxotrophy so as to block cancerous lymphocyte growth have been attempted for decades with limited success. Emerging insights about amino acid metabolism may lead to new strategies in clinical medicine whereby both amino acid auxotrophy and the immunoregulatory pathways controlled by amino acids can be manipulated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Currie, G.A. Activated macrophages kill tumour cells by releasing arginase. Nature 273, 758–759 (1978).
Currie, G.A., Gyure, L. & Cifuentes, L. Microenvironmental arginine depletion by macrophages in vivo. Br. J. Cancer 39, 613–620 (1979).
Meltzer, M.S., Ruco, L.P. & Leonard, E.J. Macrophage activation for tumor cytotoxicity: mechanisms of macrophage activation by lymphokines. Adv. Exp. Med. Biol. 121B, 381–398 (1979).
Ruco, L.P. & Meltzer, M.S. Macrophage activation for tumor cytotoxicity: development of macrophage cytotoxic activity requires completion of a sequence of short-lived intermediary reactions. J. Immunol. 121, 2035–2042 (1978).
Keller, R., Geiges, M. & Keist, R. L-Arginine-dependent reactive nitrogen intermediates as mediators of tumor cell killing by activated macrophages. Cancer Res. 50, 1421–1425 (1990).
Mills, C.D. Molecular basis of “suppressor” macrophages. Arginine metabolism via the nitric oxide synthetase pathway. J. Immunol. 146, 2719–2723 (1991).
McGaha, T.L. et al. Amino acid catabolism: a pivotal regulator of innate and adaptive immunity. Immunol. Rev. 249, 135–157 (2012).
Boulland, M.L. et al. Human IL4I1 is a secreted L-phenylalanine oxidase expressed by mature dendritic cells that inhibits T-lymphocyte proliferation. Blood 110, 220–227 (2007).
Mason, J.M. et al. IL-4-induced gene-1 is a leukocyte L-amino acid oxidase with an unusual acidic pH preference and lysosomal localization. J. Immunol. 173, 4561–4567 (2004).
Cousin, C. et al. The immunosuppressive enzyme IL4I1 promotes FoxP3+ regulatory T lymphocyte differentiation. Eur. J. Immunol. 45, 1772–1782 (2015).
Santarlasci, V. et al. Rarity of human T helper 17 cells is due to retinoic acid orphan receptor-dependent mechanisms that limit their expansion. Immunity 36, 201–214 (2012).
Vonk, F.J. et al. The king cobra genome reveals dynamic gene evolution and adaptation in the snake venom system. Proc. Natl. Acad. Sci. USA 110, 20651–20656 (2013).
Thompson, R.W. et al. Cationic amino acid transporter-2 regulates immunity by modulating arginase activity. PLoS Pathog. 4, e1000023 (2008).
Grohmann, U. & Bronte, V. Control of immune response by amino acid metabolism. Immunol. Rev. 236, 243–264 (2010).
Mills, C.D. M1 and M2 macrophages: oracles of health and disease. Crit. Rev. Immunol. 32, 463–488 (2012).
Murray, P.J. & Wynn, T.A. Obstacles and opportunities for understanding macrophage polarization. J. Leukoc. Biol. 89, 557–563 (2011).
Murray, P.J. & Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 11, 723–737 (2011).
Murray, P.J. et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41, 14–20 (2014).
Gordon, S. & Martinez, F.O. Alternative activation of macrophages: mechanism and functions. Immunity 32, 593–604 (2010).
Stein, M., Keshav, S., Harris, N. & Gordon, S. Interleukin 4 potently enhances murine macrophage mannose receptor activity: a marker of alternative immunologic macrophage activation. J. Exp. Med. 176, 287–292 (1992).
Munder, M., Eichmann, K. & Modolell, M. Alternative metabolic states in murine macrophages reflected by the nitric oxide synthase/arginase balance: competitive regulation by CD4+ T cells correlates with Th1/Th2 phenotype. J. Immunol. 160, 5347–5354 (1998).
Munder, M. et al. Th1/Th2-regulated expression of arginase isoforms in murine macrophages and dendritic cells. J. Immunol. 163, 3771–3777 (1999).
Adams, D.O. & Hamilton, T.A. The cell biology of macrophage activation. Annu. Rev. Immunol. 2, 283–318 (1984).
MacKaness, G.B. The immunological basis of acquired cellular resistance. J. Exp. Med. 120, 105–120 (1964).
Mills, C.D., Kincaid, K., Alt, J.M., Heilman, M.J. & Hill, A.M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J. Immunol. 164, 6166–6173 (2000).
Martinez, F.O. & Gordon, S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep. 6, 13 (2014).
Shi, O., Morris, S.M. Jr., Zoghbi, H., Porter, C.W. & O'Brien, W.E. Generation of a mouse model for arginase II deficiency by targeted disruption of the arginase II gene. Mol. Cell. Biol. 21, 811–813 (2001).
Gobert, A.P. et al. Helicobacter pylori arginase inhibits nitric oxide production by eukaryotic cells: a strategy for bacterial survival. Proc. Natl. Acad. Sci. USA 98, 13844–13849 (2001).
Rutschman, R. et al. Cutting edge: Stat6-dependent substrate depletion regulates nitric oxide production. J. Immunol. 166, 2173–2177 (2001).
Gray, M.J., Poljakovic, M., Kepka-Lenhart, D. & Morris, S.M. Jr. Induction of arginase I transcription by IL-4 requires a composite DNA response element for STAT6 and C/EBPbeta. Gene 353, 98–106 (2005).
Pauleau, A.L. et al. Enhancer-mediated control of macrophage-specific arginase I expression. J. Immunol. 172, 7565–7573 (2004).
Lang, R., Patel, D., Morris, J.J., Rutschman, R.L. & Murray, P.J. Shaping gene expression in activated and resting primary macrophages by IL-10. J. Immunol. 169, 2253–2263 (2002).
Chawla, A. Control of macrophage activation and function by PPARs. Circ. Res. 106, 1559–1569 (2010).
Nguyen, K.D. et al. Alternatively activated macrophages produce catecholamines to sustain adaptive thermogenesis. Nature 480, 104–108 (2011).
El Kasmi, K.C. et al. Toll-like receptor-induced arginase 1 in macrophages thwarts effective immunity against intracellular pathogens. Nat. Immunol. 9, 1399–1406 (2008).
Pesce, J.T. et al. Arginase-1-expressing macrophages suppress Th2 cytokine-driven inflammation and fibrosis. PLoS Pathog. 5, e1000371 (2009).
Barron, L. et al. Role of arginase 1 from myeloid cells in th2-dominated lung inflammation. PLoS ONE 8, e61961 (2013).
Herbert, D.R. et al. Arginase I suppresses IL-12/IL-23p40-driven intestinal inflammation during acute schistosomiasis. J. Immunol. 184, 6438–6446 (2010).
Wynn, T.A. Fibrotic disease and the T(H)1/T(H)2 paradigm. Nat. Rev. Immunol. 4, 583–594 (2004).
Morris, S.M. Jr. Arginine metabolism: boundaries of our knowledge. J. Nutr. 137 (suppl. 2), 1602S–1609S (2007).
Fletcher, M. et al. L-Arginine depletion blunts antitumor T-cell responses by inducing myeloid-derived suppressor cells. Cancer Res. 75, 275–283 (2015).
Rodriguez, P.C., Quiceno, D.G. & Ochoa, A.C. L-arginine availability regulates T-lymphocyte cell-cycle progression. Blood 109, 1568–1573 (2007).
Rodriguez, P.C. et al. L-arginine consumption by macrophages modulates the expression of CD3 zeta chain in T lymphocytes. J. Immunol. 171, 1232–1239 (2003).
Esser-von Bieren, J. et al. Antibodies trap tissue migrating helminth larvae and prevent tissue damage by driving IL-4Rα-independent alternative differentiation of macrophages. PLoS Pathog. 9, e1003771 (2013).
Albina, J.E., Mills, C.D., Henry, W.L. Jr. & Caldwell, M.D. Temporal expression of different pathways of 1-arginine metabolism in healing wounds. J. Immunol. 144, 3877–3880 (1990).
Campbell, L., Saville, C.R., Murray, P.J., Cruickshank, S.M. & Hardman, M.J. Local arginase 1 activity is required for cutaneous wound healing. J. Invest. Dermatol. 133, 2461–2470 (2013).
Bogdan, C. Nitric oxide and the immune response. Nat. Immunol. 2, 907–916 (2001).
Schindler, H. & Bogdan, C. NO as a signaling molecule: effects on kinases. Int. Immunopharmacol. 1, 1443–1455 (2001).
Nicholson, B., Manner, C.K., Kleeman, J. & MacLeod, C.L. Sustained nitric oxide production in macrophages requires the arginine transporter CAT2. J. Biol. Chem. 276, 15881–15885 (2001).
Qualls, J.E. et al. Sustained generation of nitric oxide and control of mycobacterial infection requires argininosuccinate synthase 1. Cell Host Microbe 12, 313–323 (2012).
Everts, B. et al. Commitment to glycolysis sustains survival of NO-producing inflammatory dendritic cells. Blood 120, 1422–1431 (2012).
Modolell, M., Corraliza, I.M., Link, F., Soler, G. & Eichmann, K. Reciprocal regulation of the nitric oxide synthase/arginase balance in mouse bone marrow-derived macrophages by TH1 and TH2 cytokines. Eur. J. Immunol. 25, 1101–1104 (1995).
Qualls, J.E. et al. Arginine usage in mycobacteria-infected macrophages depends on autocrine-paracrine cytokine signaling. Sci. Signal. 3, ra62 (2010).
Mills, C.D. Macrophage arginine metabolism to ornithine/urea or nitric oxide/citrulline: a life or death issue. Crit. Rev. Immunol. 21, 399–425 (2001).
El-Gayar, S., Thüring-Nahler, H., Pfeilschifter, J., Röllinghoff, M. & Bogdan, C. Translational control of inducible nitric oxide synthase by IL-13 and arginine availability in inflammatory macrophages. J. Immunol. 171, 4561–4568 (2003).
Lee, J., Ryu, H., Ferrante, R.J., Morris, S.M. Jr. & Ratan, R.R. Translational control of inducible nitric oxide synthase expression by arginine can explain the arginine paradox. Proc. Natl. Acad. Sci. USA 100, 4843–4848 (2003).
Reece, S.T. et al. Serine protease activity contributes to control of Mycobacterium tuberculosis in hypoxic lung granulomas in mice. J. Clin. Invest. 120, 3365–3376 (2010).
Duque-Correa, M.A. et al. Macrophage arginase-1 controls bacterial growth and pathology in hypoxic tuberculosis granulomas. Proc. Natl. Acad. Sci. USA 111, E4024–E4032 (2014).
Mori, M. Regulation of nitric oxide synthesis and apoptosis by arginase and arginine recycling. J. Nutr. 137 (suppl. 2), 1616S–1620S (2007).
Rapovy, S.M. et al. Differential requirements for l-citrulline and l-arginine during antimycobacterial macrophage activity. J. Immunol. 195, 3293–3300 (2015).
Jha, A.K. et al. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity 42, 419–430 (2015).
Bessede, A. et al. Aryl hydrocarbon receptor control of a disease tolerance defence pathway. Nature 511, 184–190 (2014).
Munn, D.H. & Mellor, A.L. Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol. 34, 137–143 (2013).
Platten, M., Wick, W. & Van den Eynde, B.J. Tryptophan catabolism in cancer: beyond IDO and tryptophan depletion. Cancer Res. 72, 5435–5440 (2012).
Stone, T.W., Stoy, N. & Darlington, L.G. An expanding range of targets for kynurenine metabolites of tryptophan. Trends Pharmacol. Sci. 34, 136–143 (2013).
Moffett, J.R. & Namboodiri, M.A. Tryptophan and the immune response. Immunol. Cell Biol. 81, 247–265 (2003).
Thomas, S., DuHadaway, J., Prendergast, G.C. & Laury-Kleintop, L. Specific in situ detection of murine indoleamine 2, 3-dioxygenase. J. Cell. Biochem. 115, 391–396 (2014).
Schmidt, S.K. et al. Influence of tryptophan contained in 1-Methyl-Tryptophan on antimicrobial and immunoregulatory functions of indoleamine 2,3-dioxygenase. PLoS ONE 7, e44797 (2012).
Opitz, C.A. et al. The indoleamine-2,3-dioxygenase (IDO) inhibitor 1-methyl-D-tryptophan upregulates IDO1 in human cancer cells. PLoS ONE 6, e19823 (2011).
Degterev, A., Maki, J.L. & Yuan, J. Activity and specificity of necrostatin-1, small-molecule inhibitor of RIP1 kinase. Cell Death Differ. 20, 366 (2013).
Röhrig, U.F., Majjigapu, S.R., Vogel, P., Zoete, V. & Michielin, O. Challenges in the discovery of indoleamine 2,3-dioxygenase 1 (IDO1) inhibitors. J. Med. Chem. http://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.5b00326 (23 September 2015).
Munn, D.H. et al. Inhibition of T cell proliferation by macrophage tryptophan catabolism. J. Exp. Med. 189, 1363–1372 (1999).
Munn, D.H. et al. GCN2 kinase in T cells mediates proliferative arrest and anergy induction in response to indoleamine 2,3-dioxygenase. Immunity 22, 633–642 (2005).
Cobbold, S.P. et al. Infectious tolerance via the consumption of essential amino acids and mTOR signaling. Proc. Natl. Acad. Sci. USA 106, 12055–12060 (2009).
Smith, C. et al. IDO is a nodal pathogenic driver of lung cancer and metastasis development. Cancer Discov. 2, 722–735 (2012).
Jasperson, L.K. et al. Indoleamine 2,3-dioxygenase is a critical regulator of acute graft-versus-host disease lethality. Blood 111, 3257–3265 (2008).
Holmgaard, R.B., Zamarin, D., Munn, D.H., Wolchok, J.D. & Allison, J.P. Indoleamine 2,3-dioxygenase is a critical resistance mechanism in antitumor T cell immunotherapy targeting CTLA-4. J. Exp. Med. 210, 1389–1402 (2013).
Wang, R. & Green, D.R. Metabolic checkpoints in activated T cells. Nat. Immunol. 13, 907–915 (2012).
Carr, E.L. et al. Glutamine uptake and metabolism are coordinately regulated by ERK/MAPK during T lymphocyte activation. J. Immunol. 185, 1037–1044 (2010).
Nakaya, M. et al. Inflammatory T cell responses rely on amino acid transporter ASCT2 facilitation of glutamine uptake and mTORC1 kinase activation. Immunity 40, 692–705 (2014).
Sinclair, L.V. et al. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat. Immunol. 14, 500–508 (2013).
MacIver, N.J., Michalek, R.D. & Rathmell, J.C. Metabolic regulation of T lymphocytes. Annu. Rev. Immunol. 31, 259–283 (2013).
Klysz, D. et al. Glutamine-dependent α-ketoglutarate production regulates the balance between T helper 1 cell and regulatory T cell generation. Sci. Signal. 8, ra97 (2015).
Delgoffe, G.M. et al. The mTOR kinase differentially regulates effector and regulatory T cell lineage commitment. Immunity 30, 832–844 (2009).
Zoncu, R., Efeyan, A. & Sabatini, D.M. mTOR: from growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 12, 21–35 (2011).
Sancak, Y. et al. Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141, 290–303 (2010).
Rebsamen, M. et al. SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1. Nature 519, 477–481 (2015).
Wang, S. et al. Metabolism. Lysosomal amino acid transporter SLC38A9 signals arginine sufficiency to mTORC1. Science 347, 188–194 (2015).
Yang, K. et al. T cell exit from quiescence and differentiation into Th2 cells depend on Raptor-mTORC1-mediated metabolic reprogramming. Immunity 39, 1043–1056 (2013).
Zeng, H. et al. mTORC1 couples immune signals and metabolic programming to establish Treg-cell function. Nature 499, 485–490 (2013).
Pollizzi, K.N. & Powell, J.D. Regulation of T cells by mTOR: the known knowns and the known unknowns. Trends Immunol. 36, 13–20 (2015).
Procaccini, C. et al. An oscillatory switch in mTOR kinase activity sets regulatory T cell responsiveness. Immunity 33, 929–941 (2010).
Weichhart, T., Hengstschläger, M. & Linke, M. Regulation of innate immune cell function by mTOR. Nat. Rev. Immunol. 15, 599–614 (2015).
Broome, J.D. Studies on the mechanism of tumor inhibition by L-asparaginase. Effects of the enzyme on asparagine levels in the blood, normal tissues, and 6C3HED lymphomas of mice: differences in asparagine formation and utilization in asparaginase-sensitive and -resistant lymphoma cells. J. Exp. Med. 127, 1055–1072 (1968).
Chan, W.K. et al. The glutaminase activity of L-asparaginase is not required for anticancer activity against ASNS-negative cells. Blood 123, 3596–3606 (2014).
Zhang, J. et al. Asparagine plays a critical role in regulating cellular adaptation to glutamine depletion. Mol. Cell 56, 205–218 (2014).
Pui, C.H. & Evans, W.E. Treatment of acute lymphoblastic leukemia. N. Engl. J. Med. 354, 166–178 (2006).
Chen, S.H. et al. A genome-wide approach identifies that the aspartate metabolism pathway contributes to asparaginase sensitivity. Leukemia 25, 66–74 (2011).
Yau, T. et al. A phase 1 dose-escalating study of pegylated recombinant human arginase 1 (Peg-rhArg1) in patients with advanced hepatocellular carcinoma. Invest. New Drugs 31, 99–107 (2013).
Munder, M. Arginase: an emerging key player in the mammalian immune system. Br. J. Pharmacol. 158, 638–651 (2009).
Sahin, E. et al. Macrophage PTEN regulates expression and secretion of arginase I modulating innate and adaptive immune responses. J. Immunol. 193, 1717–1727 (2014).
Gabrilovich, D.I., Ostrand-Rosenberg, S. & Bronte, V. Coordinated regulation of myeloid cells by tumours. Nat. Rev. Immunol. 12, 253–268 (2012).
Qin, S. et al. “Infectious” transplantation tolerance. Science 259, 974–977 (1993).
Beatty, W.L., Belanger, T.A., Desai, A.A., Morrison, R.P. & Byrne, G.I. Tryptophan depletion as a mechanism of gamma interferon-mediated chlamydial persistence. Infect. Immun. 62, 3705–3711 (1994).
Gaur, U. et al. An effect of parasite-encoded arginase on the outcome of murine cutaneous leishmaniasis. J. Immunol. 179, 8446–8453 (2007).
Price, J.V. & Vance, R.E. The macrophage paradox. Immunity 41, 685–693 (2014).
Mussai, F. et al. Neuroblastoma arginase activity creates an immunosuppressive microenvironment that impairs autologous and engineered immunity. Cancer Res. 75, 3043–3053 (2015).
Scott, L., Lamb, J., Smith, S. & Wheatley, D.N. Single amino acid (arginine) deprivation: rapid and selective death of cultured transformed and malignant cells. Br. J. Cancer 83, 800–810 (2000).
Cama, E. et al. Human arginase II: crystal structure and physiological role in male and female sexual arousal. Biochemistry 42, 8445–8451 (2003).
Pawelek, P.D. et al. The structure of L-amino acid oxidase reveals the substrate trajectory into an enantiomerically conserved active site. EMBO J. 19, 4204–4215 (2000).
Colegio, O.R. et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513, 559–563 (2014).
Csóka, B. et al. Adenosine promotes alternative macrophage activation via A2A and A2B receptors. FASEB J. 26, 376–386 (2012).
El Kasmi, K.C. et al. Adventitial fibroblasts induce a distinct proinflammatory/profibrotic macrophage phenotype in pulmonary hypertension. J. Immunol. 193, 597–609 (2014).
Acknowledgements
Research in the author's laboratory is supported by grants from the US National Institutes of Health, Calithera Biosciences, National Cancer Institute Cancer Core grant P30 CA21765 and the American Lebanese Associated Charities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author's laboratory has a collaborative agreement with Calithera Biosciences to investigate aspects of arginine metabolism in immune responses. No personal funds are derived from this agreement.
Rights and permissions
About this article
Cite this article
Murray, P. Amino acid auxotrophy as a system of immunological control nodes. Nat Immunol 17, 132–139 (2016). https://doi.org/10.1038/ni.3323
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3323
This article is cited by
-
Arg-tRNA synthetase links inflammatory metabolism to RNA splicing and nuclear trafficking via SRRM2
Nature Cell Biology (2023)
-
Arginyl-tRNA synthetase in inflammation
Nature Cell Biology (2023)
-
Asparagine restriction enhances CD8+ T cell metabolic fitness and antitumoral functionality through an NRF2-dependent stress response
Nature Metabolism (2023)
-
Protein synthesis, degradation, and energy metabolism in T cell immunity
Cellular & Molecular Immunology (2022)
-
Clinical perspectives on the age-related increase of immunosuppressive activity
Journal of Molecular Medicine (2022)