Abstract
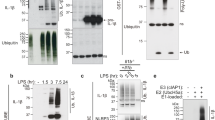
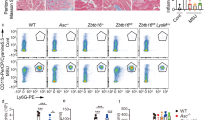
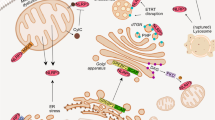
Particulate ligands, including cholesterol crystals and amyloid fibrils, induce production of interleukin 1β (IL-1β) dependent on the cytoplasmic sensor NLRP3 in atherosclerosis, Alzheimer's disease and diabetes. Soluble endogenous ligands, including oxidized low-density lipoprotein (LDL), amyloid-β and amylin peptides, accumulate in such diseases. Here we identify an endocytic pathway mediated by the pattern-recognition receptor CD36 that coordinated the intracellular conversion of those soluble ligands into crystals or fibrils, which resulted in lysosomal disruption and activation of the NLRP3 inflammasome. Consequently, macrophages that lacked CD36 failed to elicit IL-1β production in response to those ligands, and targeting CD36 in atherosclerotic mice resulted in lower serum concentrations of IL-1β and accumulation of cholesterol crystals in plaques. Collectively, our findings highlight the importance of CD36 in the accrual and nucleation of NLRP3 ligands from within the macrophage and position CD36 as a central regulator of inflammasome activation in sterile inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Abela, G.S. Cholesterol crystals piercing the arterial plaque and intima trigger local and systemic inflammation. J. Clin. Lipidol. 4, 156–164 (2010).
Meyer-Luehmann, M. et al. Rapid appearance and local toxicity of amyloid-β plaques in a mouse model of Alzheimer's disease. Nature 451, 720–724 (2008).
Westermark, G.T., Westermark, P., Berne, C. & Korsgren, O. Nordic Network for Clinical Islet T. Widespread amyloid deposition in transplanted human pancreatic islets. N. Engl. J. Med. 359, 977–979 (2008).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Halle, A. et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-beta. Nat. Immunol. 9, 857–865 (2008).
Masters, S.L. et al. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat. Immunol. 11, 897–904 (2010).
Rajamäki, K. et al. Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: a novel link between cholesterol metabolism and inflammation. PLoS ONE 5, e11765 (2010).
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1β converting enzyme. Science 267, 2000–2003 (1995).
Keller, M., Ruegg, A., Werner, S. & Beer, H.D. Active caspase-1 is a regulator of unconventional protein secretion. Cell 132, 818–831 (2008).
Bauernfeind, F. et al. Reactive oxygen species inhibitors block priming, but not activation, of the NLRP3 inflammasome. J. Immunol. 187, 613–617 (2011).
Bauernfeind, F.G. et al. NF-κB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. 183, 787–791 (2009).
Embry, C.A., Franchi, L., Nunez, G. & Mitchell, T.C. Mechanism of impaired NLRP3 inflammasome priming by monophosphoryl lipid A. Sci. Signal. 4, ra28 (2011).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 9, 847–856 (2008).
Zhou, R., Yazdi, A.S., Menu, P. & Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 469, 221–225 (2011).
Williams, K.J. & Tabas, I. The response-to-retention hypothesis of early atherogenesis. Arterioscler. Thromb. Vasc. Biol. 15, 551–561 (1995).
Lim, R.S. et al. Identification of cholesterol crystals in plaques of atherosclerotic mice using hyperspectral CARS imaging. J. Lipid Res. 52, 2177–2186 (2011).
Haass, C. et al. Amyloid β-peptide is produced by cultured cells during normal metabolism. Nature 359, 322–325 (1992).
Kirschner, D.A. et al. Synthetic peptide homologous to β protein from Alzheimer disease forms amyloid-like fibrils in vitro. Proc. Natl. Acad. Sci. USA 84, 6953–6957 (1987).
Seubert, P. et al. Isolation and quantification of soluble Alzheimer's β-peptide from biological fluids. Nature 359, 325–327 (1992).
Clark, A. et al. Islet amyloid formed from diabetes-associated peptide may be pathogenic in type-2 diabetes. Lancet 2, 231–234 (1987).
Cooper, G.J. et al. Purification and characterization of a peptide from amyloid-rich pancreases of type 2 diabetic patients. Proc. Natl. Acad. Sci. USA 84, 8628–8632 (1987).
Westermark, P. et al. Amyloid fibrils in human insulinoma and islets of Langerhans of the diabetic cat are derived from a neuropeptide-like protein also present in normal islet cells. Proc. Natl. Acad. Sci. USA 84, 3881–3885 (1987).
Badman, M.K., Pryce, R.A., Charge, S.B., Morris, J.F. & Clark, A. Fibrillar islet amyloid polypeptide (amylin) is internalised by macrophages but resists proteolytic degradation. Cell Tissue Res. 291, 285–294 (1998).
de Koning, E.J. et al. Macrophages and pancreatic islet amyloidosis. Amyloid 5, 247–254 (1998).
Haass, C., Koo, E.H., Mellon, A., Hung, A.Y. & Selkoe, D.J. Targeting of cell-surface β-amyloid precursor protein to lysosomes: alternative processing into amyloid-bearing fragments. Nature 357, 500–503 (1992).
Hartmann, T. et al. Distinct sites of intracellular production for Alzheimer's disease Aβ40/42 amyloid peptides. Nat. Med. 3, 1016–1020 (1997).
Silverstein, R.L. & Febbraio, M. CD36, a scavenger receptor involved in immunity, metabolism, angiogenesis, and behavior. Sci. Signal. 2, re3 (2009).
Kunjathoor, V.V. et al. Scavenger receptors class A-I/II and CD36 are the principal receptors responsible for the uptake of modified low density lipoprotein leading to lipid loading in macrophages. J. Biol. Chem. 277, 49982–49988 (2002).
Moore, K.J. et al. A CD36-initiated signaling cascade mediates inflammatory effects of β-amyloid. J. Biol. Chem. 277, 47373–47379 (2002).
Wilkinson, K., Boyd, J.D., Glicksman, M., Moore, K.J. & El Khoury, J. A high content drug screen identifies ursolic acid as an inhibitor of amyloid beta protein interactions with its receptor CD36. J. Biol. Chem. 286, 34914–34922 (2011).
Stewart, C.R. et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 11, 155–161 (2010).
El Khoury, J.B. et al. CD36 mediates the innate host response to β-amyloid. J. Exp. Med. 197, 1657–1666 (2003).
Febbraio, M. et al. Targeted disruption of the class B scavenger receptor CD36 protects against atherosclerotic lesion development in mice. J. Clin. Invest. 105, 1049–1056 (2000).
Moore, K.J. et al. Loss of receptor-mediated lipid uptake via scavenger receptor A or CD36 pathways does not ameliorate atherosclerosis in hyperlipidemic mice. J. Clin. Invest. 115, 2192–2201 (2005).
Goudriaan, J.R. et al. CD36 deficiency increases insulin sensitivity in muscle, but induces insulin resistance in the liver in mice. J. Lipid Res. 44, 2270–2277 (2003).
Hajri, T., Han, X.X., Bonen, A. & Abumrad, N.A. Defective fatty acid uptake modulates insulin responsiveness and metabolic responses to diet in CD36-null mice. J. Clin. Invest. 109, 1381–1389 (2002).
Kennedy, D.J. et al. A CD36-dependent pathway enhances macrophage and adipose tissue inflammation and impairs insulin signalling. Cardiovasc. Res. 89, 604–613 (2011).
Binder, C.J. et al. Pneumococcal vaccination decreases atherosclerotic lesion formation: molecular mimicry between Streptococcus pneumoniae and oxidized LDL. Nat. Med. 9, 736–743 (2003).
Schmitz, G. & Grandl, M. Endolysosomal phospholipidosis and cytosolic lipid droplet storage and release in macrophages. Biochim. Biophys. Acta 1791, 524–539 (2009).
Rosenbaum, A.I. et al. Chemical screen to reduce sterol accumulation in Niemann-Pick C disease cells identifies novel lysosomal acid lipase inhibitors. Biochim. Biophys. Acta 1791, 1155–1165 (2009).
Heneka, M.T. et al. NLRP3 is activated in Alzheimer's disease and contributes to pathology in APP/PS1 mice. Nature 493, 674–678 (2013).
Friedrich, R.P. et al. Mechanism of amyloid plaque formation suggests an intracellular basis of Abeta pathogenicity. Proc. Natl. Acad. Sci. USA 107, 1942–1947 (2010).
Walsh, D.M., Tseng, B.P., Rydel, R.E., Podlisny, M.B. & Selkoe, D.J. The oligomerization of amyloid β-protein begins intracellularly in cells derived from human brain. Biochemistry 39, 10831–10839 (2000).
Miao, E.A. et al. Caspase-1-induced pyroptosis is an innate immune effector mechanism against intracellular bacteria. Nat. Immunol. 11, 1136–1142 (2010).
Kamari, Y. et al. Reduced atherosclerosis and inflammatory cytokines in apolipoprotein-E-deficient mice lacking bone marrow-derived interleukin-1α. Biochem. Biophys. Res. Commun. 405, 197–203 (2011).
Kuchibhotla, S. et al. Absence of CD36 protects against atherosclerosis in ApoE knock-out mice with no additional protection provided by absence of scavenger receptor A I/II. Cardiovasc. Res. 78, 185–196 (2008).
Chen, C.J. et al. Identification of a key pathway required for the sterile inflammatory response triggered by dying cells. Nat. Med. 13, 851–856 (2007).
Rasmussen, L.T. & Seljelid, R. The modulatory effect of lipoproteins on the release of interleukin 1 by human peritoneal macrophages stimulated with β-1,3-D-polyglucose derivatives. Scand. J. Immunol. 29, 477–484 (1989).
Thomas, C.E., Jackson, R.L., Ohlweiler, D.F. & Ku, G. Multiple lipid oxidation products in low density lipoproteins induce interleukin-1β release from human blood mononuclear cells. J. Lipid Res. 35, 417–427 (1994).
Moyer, C.F., Sajuthi, D., Tulli, H. & Williams, J.K. Synthesis of IL-1α and IL-1β by arterial cells in atherosclerosis. Am. J. Pathol. 138, 951–960 (1991).
Guarda, G. & So, A. Regulation of inflammasome activity. Immunology 130, 329–336 (2010).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Boltz-Nitulescu, G. et al. Differentiation of rat bone marrow cells into macrophages under the influence of mouse L929 cell supernatant. J. Leuk. Biol. 41, 83–91 (1987).
Stuart, L.M. et al. Response to Staphylococcus aureus requires CD36-mediated phagocytosis triggered by the COOH-terminal cytoplasmic domain. J. Cell Biol. 170, 477–485 (2005).
Acknowledgements
We thank F. Maxfield (Weill Cornell Medical College) for Lalistat. Supported by the US National Institutes of Health (R01HL117334 and R01AG032349 to K.J.M.; U24 AI082660 to L.M.S. and K.J.M.; R01 AI079198 to L.M.S.; 5R01HL093262-02 and 1R01HL112661-01 to E.L.; and AI083713 to K.A.F. and E.L.) and the American Heart Association (11POST7400075 to F.J.S.).
Author information
Authors and Affiliations
Contributions
F.J.S. designed, did and analyzed experiments; A.G. analyzed cholesterol crystals in atherosclerotic plaques; K.J.R. and H.N.E. assisted with mouse atherosclerosis experiments. P.K., B.R., S.B.C. and C.E.B. assisted with microscopy experiments; A.E.M. provided CD36-specific ASOs and contributed to experimental design; D.T.G., L.M.S., E.L. and K.A.F. contributed to study design, data analysis, and manuscript preparation; and K.J.M. conceived of the study, designed and analyzed experiments and, along with F.J.S., prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
A.E.M. is an employee of Isis Pharmaceuticals, a biotechnology company that develops ASO therapies.
Supplementary information
Supplementary Figures and Tables
Supplementary Figures 1–6 and Supplementary Table 1 (PDF 9146 kb)
Rights and permissions
About this article
Cite this article
Sheedy, F., Grebe, A., Rayner, K. et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat Immunol 14, 812–820 (2013). https://doi.org/10.1038/ni.2639
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2639
This article is cited by
-
The role of inflammasomes in human diseases and their potential as therapeutic targets
Signal Transduction and Targeted Therapy (2024)
-
TNEA therapy promotes the autophagic degradation of NLRP3 inflammasome in a transgenic mouse model of Alzheimer’s disease via TFEB/TFE3 activation
Journal of Neuroinflammation (2023)
-
The NLRP3 inflammasome: contributions to inflammation-related diseases
Cellular & Molecular Biology Letters (2023)
-
Cerebral organoids with chromosome 21 trisomy secrete Alzheimer’s disease-related soluble aggregates detectable by single-molecule-fluorescence and super-resolution microscopy
Molecular Psychiatry (2023)
-
Drug screen identifies verteporfin as a regulator of lipid metabolism in macrophage foam cells
Scientific Reports (2023)