Abstract
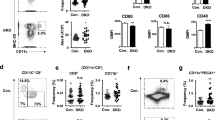
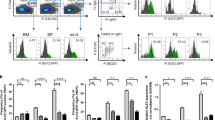
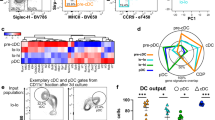
Spleen-resident dendritic cell (DC) populations occupy sentinel positions for the capture and presentation of blood-borne antigens. Here we found a difference in expression of the chemotactic receptor EBI2 (GPR183) on splenic DC subsets and that EBI2 regulated the positioning and homeostasis of DCs in the spleen. EBI2 and its main ligand, 7α,25-OHC, were required for the generation of the splenic CD4+ DC subset and the localization of DCs in bridging channels. Absence of EBI2 from DCs resulted in defects in both the activation of CD4+ T cells and the induction of antibody responses. Regulated expression of EBI2 on DC populations is therefore critical for the generation and correct positioning of splenic DCs and the initiation of immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
22 April 2013
In the version of this article initially published, the top graph in Figure 7g was incorrect. The error has been corrected in the HTML and PDF versions of the article.
References
Shortman, K. & Liu, Y.J. Mouse and human dendritic cell subtypes. Nat. Rev. Immunol. 2, 151–161 (2002).
Villadangos, J.A. & Schnorrer, P. Intrinsic and cooperative antigen-presenting functions of dendritic-cell subsets in vivo. Nat. Rev. Immunol. 7, 543–555 (2007).
Satpathy, A.T., Wu, X., Albring, J.C. & Murphy, K.M. Re(de)fining the dendritic cell lineage. Nat. Immunol. 13, 1145–1154 (2012).
Pulendran, B., Tang, H. & Denning, T.L. Division of labor, plasticity, and crosstalk between dendritic cell subsets. Curr. Opin. Immunol. 20, 61–67 (2008).
Vremec, D., Pooley, J., Hochrein, H., Wu, L. & Shortman, K. CD4 and CD8 expression by dendritic cell subtypes in mouse thymus and spleen. J. Immunol. 164, 2978–2986 (2000).
Dudziak, D. et al. Differential antigen processing by dendritic cell subsets in vivo. Science 315, 107–111 (2007).
Luber, C.A. et al. Quantitative proteomics reveals subset-specific viral recognition in dendritic cells. Immunity 32, 279–289 (2010).
Mount, A.M. et al. Multiple dendritic cell populations activate CD4+ T cells after viral stimulation. PLoS One 3, e1691 (2008).
den Haan, J.M. & Bevan, M.J. Constitutive versus activation-dependent cross-presentation of immune complexes by CD8+ and CD8− dendritic cells in vivo. J. Exp. Med. 196, 817–827 (2002).
Pooley, J.L., Heath, W.R. & Shortman, K. Cutting edge: intravenous soluble antigen is presented to CD4 T cells by CD8− dendritic cells, but cross-presented to CD8 T cells by CD8+ dendritic cells. J. Immunol. 166, 5327–5330 (2001).
Chappell, C.P., Draves, K.E., Giltiay, N.V. & Clark, E.A. Extrafollicular B cell activation by marginal zone dendritic cells drives T cell-dependent antibody responses. J. Exp. Med. 209, 1825–1840 (2012).
den Haan, J.M., Lehar, S.M. & Bevan, M.J. CD8+ but not CD8− dendritic cells cross-prime cytotoxic T cells in vivo. J. Exp. Med. 192, 1685–1696 (2000).
Schnorrer, P. et al. The dominant role of CD8+ dendritic cells in cross-presentation is not dictated by antigen capture. Proc. Natl. Acad. Sci. USA 103, 10729–10734 (2006).
Caminschi, I. et al. The dendritic cell subtype-restricted C-type lectin Clec9A is a target for vaccine enhancement. Blood 112, 3264–3273 (2008).
Sancho, D. et al. Tumor therapy in mice via antigen targeting to a novel, DC-restricted C-type lectin. J. Clin. Invest. 118, 2098–2110 (2008).
Kabashima, K. et al. Intrinsic lymphotoxin-beta receptor requirement for homeostasis of lymphoid tissue dendritic cells. Immunity 22, 439–450 (2005).
Naik, S.H. et al. Intrasplenic steady-state dendritic cell precursors that are distinct from monocytes. Nat. Immunol. 7, 663–671 (2006).
Diao, J. et al. In situ replication of immediate dendritic cell (DC) precursors contributes to conventional DC homeostasis in lymphoid tissue. J. Immunol. 176, 7196–7206 (2006).
Liu, K. et al. Origin of dendritic cells in peripheral lymphoid organs of mice. Nat. Immunol. 8, 578–583 (2007).
Belz, G.T. & Nutt, S.L. Transcriptional programming of the dendritic cell network. Nat. Rev. Immunol. 12, 101–113 (2012).
Gunn, M.D. et al. Mice lacking expression of secondary lymphoid organ chemokine have defects in lymphocyte homing and dendritic cell localization. J. Exp. Med. 189, 451–460 (1999).
Reis e Sousa, C. et al. In vivo microbial stimulation induces rapid CD40 ligand-independent production of interleukin 12 by dendritic cells and their redistribution to T cell areas. J. Exp. Med. 186, 1819–1829 (1997).
Ohl, L. et al. CCR7 governs skin dendritic cell migration under inflammatory and steady-state conditions. Immunity 21, 279–288 (2004).
Czeloth, N. et al. Sphingosine-1 phosphate signaling regulates positioning of dendritic cells within the spleen. J. Immunol. 179, 5855–5863 (2007).
Hannedouche, S. et al. Oxysterols direct immune cell migration via EBI2. Nature 475, 524–527 (2011).
Liu, C. et al. Oxysterols direct B-cell migration through EBI2. Nature 475, 519–523 (2011).
Yi, T. et al. Oxysterol gradient generation by lymphoid stromal cells guides activated B cell movement during humoral responses. Immunity 37, 535–548 (2012).
Gatto, D., Paus, D., Basten, A., Mackay, C.R. & Brink, R. Guidance of B cells by the orphan G protein-coupled receptor EBI2 shapes humoral immune responses. Immunity 31, 259–269 (2009).
Pereira, J.P., Kelly, L.M., Xu, Y. & Cyster, J.G. EBI2 mediates B cell segregation between the outer and centre follicle. Nature 460, 1122–1126 (2009).
Gatto, D., Wood, K. & Brink, R. EBI2 operates independently of but in cooperation with CXCR5 and CCR7 to direct B cell migration and organization in follicles and the germinal center. J. Immunol. 187, 4621–4628 (2011).
Kelly, L.M., Pereira, J.P., Yi, T., Xu, Y. & Cyster, J.G. EBI2 guides serial movements of activated B cells and ligand activity is detectable in lymphoid and nonlymphoid tissues. J. Immunol. 187, 3026–3032 (2011).
Shortman, K. & Naik, S.H. Steady-state and inflammatory dendritic-cell development. Nat. Rev. Immunol. 7, 19–30 (2007).
Kamath, A.T. et al. The development, maturation, and turnover rate of mouse spleen dendritic cell populations. J. Immunol. 165, 6762–6770 (2000).
Türeci, O. et al. Cascades of transcriptional induction during dendritic cell maturation revealed by genome-wide expression analysis. FASEB J. 17, 836–847 (2003).
Paus, D. et al. Antigen recognition strength regulates the choice between extrafollicular plasma cell and germinal center B cell differentiation. J. Exp. Med. 203, 1081–1091 (2006).
Brink, R., Phan, T.G., Paus, D. & Chan, T.D. Visualizing the effects of antigen affinity on T-dependent B-cell differentiation. Immunol. Cell Biol. 86, 31–39 (2008).
Lahoud, M.H. et al. Signal regulatory protein molecules are differentially expressed by CD8− dendritic cells. J. Immunol. 177, 372–382 (2006).
Garciá De Vinuesa, C. et al. Dendritic cells associated with plasmablast survival. Eur. J. Immunol. 29, 3712–3721 (1999).
Ansel, K.M. et al. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 406, 309–314 (2000).
Lahoud, M.H. et al. The C-type lectin Clec12A present on mouse and human dendritic cells can serve as a target for antigen delivery and enhancement of antibody responses. J. Immunol. 182, 7587–7594 (2009).
Rosenkilde, M.M. et al. Molecular pharmacological phenotyping of EBI2. An orphan seven-transmembrane receptor with constitutive activity. J. Biol. Chem. 281, 13199–13208 (2006).
Phan, T.G. et al. B cell receptor-independent stimuli trigger immunoglobulin (Ig) class switch recombination and production of IgG autoantibodies by anergic self-reactive B cells. J. Exp. Med. 197, 845–860 (2003).
Barnden, M.J., Allison, J., Heath, W.R. & Carbone, F.R. Defective TCR expression in transgenic mice constructed using cDNA-based α- and β-chain genes under the control of heterologous regulatory elements. Immunol. Cell Biol. 76, 34–40 (1998).
Caton, M.L., Smith-Raska, M.R. & Reizis, B. Notch-RBP-J signaling controls the homeostasis of CD8− dendritic cells in the spleen. J. Exp. Med. 204, 1653–1664 (2007).
Bauman, D.R. et al. 25-Hydroxycholesterol secreted by macrophages in response to Toll-like receptor activation suppresses immunoglobulin A production. Proc. Natl. Acad. Sci. USA 106, 16764–16769 (2009).
Phan, T.G., Gardam, S., Basten, A. & Brink, R. Altered migration, recruitment, and somatic hypermutation in the early response of marginal zone B cells to T cell-dependent antigen. J. Immunol. 174, 4567–4578 (2005).
Allen, C.D. et al. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nat. Immunol. 5, 943–952 (2004).
Acknowledgements
We thank the MLC Garvan Flow Cytometry Facility for cell sorting; the staff of Australian Bioresources and the Garvan Institute animal facilities; and J. Villadangos, K. Shortman and S. Tangye for comments on the manuscript. Supported by the National Health and Medical Research Council of Australia (427620 to R.B. and 1003025 to D.G.)
Author information
Authors and Affiliations
Contributions
D.G. and R.B. conceived of the project and wrote the manuscript; D.G. and K.W. did most of the experiments; I.C. provided antibody to Sirpβ1 and analyzed anti-rat IgG responses; D.M.-D. produced and analyzed cells transfected to express EBI2; P.S. and D.C. organized the production and purification of antibody to EBI2; and G.K. helped analyze responses in EBI2-deficient mice.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 3476 kb)
Rights and permissions
About this article
Cite this article
Gatto, D., Wood, K., Caminschi, I. et al. The chemotactic receptor EBI2 regulates the homeostasis, localization and immunological function of splenic dendritic cells. Nat Immunol 14, 446–453 (2013). https://doi.org/10.1038/ni.2555
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2555
This article is cited by
-
The transcription factor Zeb1 controls homeostasis and function of type 1 conventional dendritic cells
Nature Communications (2023)
-
An epithelial cell-derived metabolite tunes immunoglobulin A secretion by gut-resident plasma cells
Nature Immunology (2023)
-
Immune microenvironment-related gene mapping predicts immunochemotherapy response and prognosis in diffuse large B-cell lymphoma
Medical Oncology (2022)
-
Dendritic cell subsets in T cell programming: location dictates function
Nature Reviews Immunology (2019)
-
Chemokine and chemotactic signals in dendritic cell migration
Cellular & Molecular Immunology (2018)