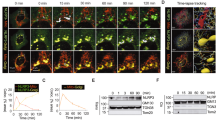
Abstract
NLRP3 forms an inflammasome with its adaptor ASC, and its excessive activation can cause inflammatory diseases. However, little is known about the mechanisms that control assembly of the inflammasome complex. Here we show that microtubules mediated assembly of the NLRP3 inflammasome. Inducers of the NLRP3 inflammasome caused aberrant mitochondrial homeostasis to diminish the concentration of the coenzyme NAD+, which in turn inactivated the NAD+-dependent α-tubulin deacetylase sirtuin 2; this resulted in the accumulation of acetylated α-tubulin. Acetylated α-tubulin mediated the dynein-dependent transport of mitochondria and subsequent apposition of ASC on mitochondria to NLRP3 on the endoplasmic reticulum. Therefore, in addition to direct activation of NLRP3, the creation of optimal sites for signal transduction by microtubules is required for activation of the entire NLRP3 inflammasome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Núñez, G. Intracellular sensors of microbes and danger. Immunol. Rev. 243, 5–8 (2011).
Strowig, T., Henao-Mejia, J., Elinav, E. & Flavell, R. Inflammasomes in health and disease. Nature 481, 278–286 (2012).
Martinon, F. et al. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Masters, S.L. et al. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat. Immunol. 11, 897–904 (2010).
Vandanmagsar, B. et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 17, 179–188 (2011).
Wen, H. et al. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 12, 408–415 (2011).
Cronstein, B.N. & Terkeltaub, R. The inflammatory process of gout and its treatment. Arthritis Res. Ther. 8, S3 (2006).
Allen, J.N. et al. Colchicine has opposite effects on interleukin-1β and tumor necrosis factor-alpha production. Am. J. Physiol. 26, 315–21 (1991).
Carta, S. et al. Histone deacetylase inhibitors prevent exocytosis of interleukin-1β-containing secretory lysosomes: role of microtubules. Blood 108, 1618–1626 (2006).
Zhou, R., Yazdi, A.S., Menu, P. & Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 469, 221–225 (2011).
Howard, J. & Hyman, A.A. Dynamics and mechanics of the microtubule plus end. Nature 422, 753–758 (2003).
Kardon, J.R. & Vale, R.D. Regulators of the cytoplasmic dynein motor. Nat. Rev. Mol. Cell Biol. 10, 854–865 (2009).
Firestone, A.J. et al. Small-molecule inhibitors of the AAA+ ATPase motor cytoplasmic dynein. Nature 484, 125–129 (2012).
Naik, E. & Dixit, V.M. Mitochondrial reactive oxygen species drive proinflammatory cytokine production. J. Exp. Med. 208, 417–420 (2011).
Wloga, D. & Gaertig, J. Post-translational modifications of microtubules. J. Cell Sci. 123, 3447–3455 (2010).
Reed, N.A. et al. Microtubule acetylation promotes kinesin-1 binding and transport. Curr. Biol. 16, 2166–2172 (2006).
Dompierre, J.P. et al. Histone deacetylase 6 inhibition compensates for the transport deficit in Huntington's disease by increasing tubulin acetylation. J. Neurosci. 27, 3571–3583 (2007).
Friedman, J. et al. ER sliding dynamics and ER-mitochondrial contacts occur on acetylated microtubules. J. Cell Biol. 190, 363–375 (2010).
Akella, J. et al. MEC-17 is an α-tubulin acetyltransferase. Nature 467, 218–222 (2010).
North, B. et al. The human ortholog, SIRT2, is an NAD+-dependent tubulin deacetylase. Mol. Cell 11, 437–444 (2003).
Hubbert, C. et al. HDAC6 is a microtubule-associated deacetylase. Nature 417, 455–458 (2002).
Nunnari, J. & Suomalainen, A. Mitochondria: in sickness and in health. Cell 148, 1145–1159 (2012).
Hentze, H., Lin, X.Y., Choi, M.S.K. & Porter, A.G. Critical role of cathepsin B in mediating caspase-1-dependent interleukin-18 maturation and caspase-1-independent necrosis triggered by the microbial toxin negericin. Cell Death Differ. 10, 956–968 (2003).
Gracia-Marcos, M. et al. Role of sodium in mitochondrial depolarization induced by P2X7 receptor activation in submandibular glands. FEBS Lett. 579, 5407–5413 (2005).
Li, Z. et al. The lysosomal-mitochondria axis in free fatty acid-induced hepatic lipotoxicity. Hepatology 47, 1495–1503 (2011).
McGuire, K. et al. Adenovirus type 5 rupture of lysosome leads to cathepsin B-dependent mitochondrial stress and production of reactive oxygen species. J. Virol. 85, 10806–10813 (2011).
Tanaka, A. Parkin-mediated selective mitochondrial autophagy, mitophagy: Parkin purges damaged organelles from the vital mitochondrial network. FEBS Lett. 584, 1386–1392 (2010).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 9, 847–856 (2008).
Dostert, C. et al. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 320, 674–677 (2008).
Ichinohe, T. et al. Influenza virus activates inflammasomes via its intracellular M2 ion channel. Nat. Immunol. 11, 404–410 (2010).
Shenoy, A. et al. GBP5 promotes NLRP3 inflammasome assembly and immunity in mammals. Science 336, 481–485 (2012).
Hornung, V. et al. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 458, 514–518 (2009).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Broz, P. et al. Redundant roles for inflammasome receptors NLRP3 and NLRC4 in host defense against Salmonella. J. Exp. Med. 207, 1745–1755 (2010).
Li., Z. et al. The lysosomal-mitochondria axis in free fatty acid-induced hepatic lipotoxicity. Hepatology 47, 1495–503 (2008).
Yoshino, J., Mills, K.F., Yoon, M.J. & Imai, S. Nicotinamide mononucleotide, a key NAD+ intermediate, treats pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 14, 528–536 (2011).
Sorbara, M.T. & Girardin, S.E. Mitochondrial ROS fuel the inflammasome. Cell Res. 21, 558–560 (2011).
Saitoh, T. & Akira, S. Regulation of innate immune responses by autophagy-related proteins. J. Cell Biol. 189, 925–935 (2010).
Itakura, E. et al. Structures containing Atg9A and the ULK1 complex independently target depolarized mitochondria at initial stages of Parkin-mediated mitophagy. J. Cell Sci. 125, 1488–1499 (2012).
Zhao, Y. et al. Cytosolic FoxO1 is essential for the induction of autophagy and tumour suppressor activity. Nat. Cell Biol. 12, 665–675 (2010).
Kume, S. et al. Calorie restriction enhances cell adaption to hypoxia through Sirt1-dependent mitochondrial autophagy in mouse aged kidney. J. Clin. Invest. 120, 1043–1055 (2010).
Chen, D. et al. Tissue specific regulation of SIRT1 by calorie restriction. Genes Dev. 22, 1753–1757 (2008).
Saitoh, T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456, 264–268 (2008).
Acknowledgements
We thank T. Kitamura (Tokyo University) for Plat-E packaging cells; H. Osada and T. Saito (RIKEN Natural Products Depository) for the authentic chemical compound library of the RIKEN Natural Products Depository; T. Yoshimori and K. Ikegami for critical reading of the manuscript; and members of the Laboratory of Host Defense for assistance. Supported by Japan Society for the Promotion of Science Grant-in-Aid for Specially Promoted Research (S.A.), Japan Society for the Promotion of Science Funding Program for World-Leading Innovative R&D on Science and Technology 'FIRST Program' (S.A.) and Japan Science and Technology Agency Core Research for Evolutional Science and Technology (T.S.).
Author information
Authors and Affiliations
Contributions
T.M. did most of the experiments and analyzed the data; M.T., T.K., H.L. and J.Z. helped with the experiments; T.M. and T.S. designed most of the experiments and wrote the manuscript; and S.A. supervised the overall research project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 2198 kb)
Rights and permissions
About this article
Cite this article
Misawa, T., Takahama, M., Kozaki, T. et al. Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome. Nat Immunol 14, 454–460 (2013). https://doi.org/10.1038/ni.2550
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2550
This article is cited by
-
Nuclear receptor coactivator 6 is a critical regulator of NLRP3 inflammasome activation and gouty arthritis
Cellular & Molecular Immunology (2024)
-
The NADase CD38 is a central regulator in gouty inflammation and a novel druggable therapeutic target
Inflammation Research (2024)
-
Inflammatory signalling in atrial cardiomyocytes: a novel unifying principle in atrial fibrillation pathophysiology
Nature Reviews Cardiology (2023)
-
Possible therapeutic targets for NLRP3 inflammasome-induced breast cancer
Discover Oncology (2023)
-
Role of NLRP3 Inflammasome and Its Inhibitors as Emerging Therapeutic Drug Candidate for Alzheimer’s Disease: a Review of Mechanism of Activation, Regulation, and Inhibition
Inflammation (2023)