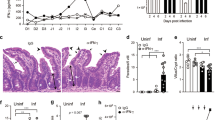
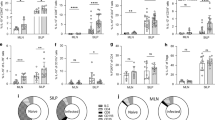
Abstract
Activation of Toll-like receptors (TLRs) by pathogens triggers cytokine production and T cell activation, immune defense mechanisms that are linked to immunopathology. Here we show that IFN-γ production by CD4+ TH1 cells during mucosal responses to the protozoan parasite Toxoplasma gondii resulted in dysbiosis and the elimination of Paneth cells. Paneth cell death led to loss of antimicrobial peptides and occurred in conjunction with uncontrolled expansion of the Enterobacteriaceae family of Gram-negative bacteria. The expanded intestinal bacteria were required for the parasite-induced intestinal pathology. The investigation of cell type–specific factors regulating TH1 polarization during T. gondii infection identified the T cell–intrinsic TLR pathway as a major regulator of IFN-γ production in CD4+ T cells responsible for Paneth cell death, dysbiosis and intestinal immunopathology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kawai, T. & Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34, 637–650 (2011).
Iwasaki, A. & Medzhitov, R. Regulation of adaptive immunity by the innate immune system. Science 327, 291–295 (2010).
Beutler, B. Microbe sensing, positive feedback loops, and the pathogenesis of inflammatory diseases. Immunol. Rev. 227, 248–263 (2009).
Medzhitov, R., Schneider, D.S. & Soares, M.P. Disease tolerance as a defense strategy. Science 335, 936–941 (2012).
Gazzinelli, R.T. & Denkers, E.Y. Protozoan encounters with Toll-like receptor signalling pathways: implications for host parasitism. Nat. Rev. Immunol. 6, 895–906 (2006).
Sukhumavasi, W. et al. TLR adaptor MyD88 is essential for pathogen control during oral Toxoplasma gondii infection but not adaptive immunity induced by a vaccine strain of the parasite. J. Immunol. 181, 3464–3473 (2008).
Scanga, C.A. et al. Cutting edge: MyD88 is required for resistance to Toxoplasma gondii infection and regulates parasite-induced IL-12 production by dendritic cells. J. Immunol. 168, 5997–6001 (2002).
Scharton-Kersten, T.M. et al. In the absence of endogenous IFN-gamma, mice develop unimpaired IL-12 responses to Toxoplasma gondii while failing to control acute infection. J. Immunol. 157, 4045–4054 (1996).
Lieberman, L.A. et al. IL-23 provides a limited mechanism of resistance to acute toxoplasmosis in the absence of IL-12. J. Immunol. 173, 1887–1893 (2004).
Suzuki, Y., Orellana, M.A., Schreiber, R.D. & Remington, J.S. Interferon-gamma: the major mediator of resistance against Toxoplasma gondii. Science 240, 516–518 (1988).
Gazzinelli, R.T. et al. In the absence of endogenous IL-10, mice acutely infected with Toxoplasma gondii succumb to a lethal immune response dependent on CD4+ T cells and accompanied by overproduction of IL-12, IFN-gamma and TNF-alpha. J. Immunol. 157, 798–805 (1996).
Jankovic, D. et al. Conventional T-bet+Foxp3– Th1 cells are the major source of host-protective regulatory IL-10 during intracellular protozoan infection. J. Exp. Med. 204, 273–283 (2007).
Jankovic, D., Kugler, D.G. & Sher, A. IL-10 production by CD4+ effector T cells: a mechanism for self-regulation. Mucosal Immunol. 3, 239–246 (2010).
Egan, C.E., Cohen, S.B. & Denkers, E.Y. Insights into inflammatory bowel disease using Toxoplasma gondii as an infectious trigger. Immunol. Cell Biol. 90, 668–675 (2012).
Jankovic, D. et al. In the absence of IL-12, CD4+ T cell responses to intracellular pathogens fail to default to a Th2 pattern and are host protective in an IL-10−/− setting. Immunity 16, 429–439 (2002).
Hou, B., Benson, A., Kuzmich, L., DeFranco, A.L. & Yarovinsky, F. Critical coordination of innate immune defense against Toxoplasma gondii by dendritic cells responding via their Toll-like receptors. Proc. Natl. Acad. Sci. USA 108, 278–283 (2011).
Yarovinsky, F. et al. TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science 308, 1626–1629 (2005).
Erridge, C., Duncan, S.H., Bereswill, S. & Heimesaat, M.M. The induction of colitis and ileitis in mice is associated with marked increases in intestinal concentrations of stimulants of TLRs 2, 4, and 5. PLoS ONE 5, e9125 (2010).
Heimesaat, M.M. et al. Gram-negative bacteria aggravate murine small intestinal Th1-type immunopathology following oral infection with Toxoplasma gondii. J. Immunol. 177, 8785–8795 (2006).
Garrett, W.S. et al. Enterobacteriaceae act in concert with the gut microbiota to induce spontaneous and maternally transmitted colitis. Cell Host Microbe 8, 292–300 (2010).
Willing, B.P. et al. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology 139, 1844–1854 (2010).
Bevins, C.L. & Salzman, N.H. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nat. Rev. Microbiol. 9, 356–368 (2011).
Wehkamp, J., Koslowski, M., Wang, G. & Stange, E.F. Barrier dysfunction due to distinct defensin deficiencies in small intestinal and colonic Crohn's disease. Mucosal Immunol. 1 (suppl. 1), S67–S74 (2008).
Vora, P. et al. Beta-defensin-2 expression is regulated by TLR signaling in intestinal epithelial cells. J. Immunol. 173, 5398–5405 (2004).
Vaishnava, S., Behrendt, C.L., Ismail, A.S., Eckmann, L. & Hooper, L.V. Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface. Proc. Natl. Acad. Sci. USA 105, 20858–20863 (2008).
Brandl, K., Plitas, G., Schnabl, B., DeMatteo, R.P. & Pamer, E.G. MyD88-mediated signals induce the bactericidal lectin RegIII gamma and protect mice against intestinal Listeria monocytogenes infection. J. Exp. Med. 204, 1891–1900 (2007).
Yuan, J. & Kroemer, G. Alternative cell death mechanisms in development and beyond. Genes Dev. 24, 2592–2602 (2010).
Benson, A., Pifer, R., Behrendt, C.L., Hooper, L.V. & Yarovinsky, F. Gut commensal bacteria direct a protective immune response against Toxoplasma gondii. Cell Host Microbe 6, 187–196 (2009).
Plattner, F. et al. Toxoplasma profilin is essential for host cell invasion and TLR11-dependent induction of an interleukin-12 response. Cell Host Microbe 3, 77–87 (2008).
Sun, D. & Ding, A. MyD88-mediated stabilization of interferon-gamma-induced cytokine and chemokine mRNA. Nat. Immunol. 7, 375–381 (2006).
Han, J. MyD88 beyond Toll. Nat. Immunol. 7, 370–371 (2006).
Mellman, I. & Steinman, R.M. Dendritic cells: specialized and regulated antigen processing machines. Cell 106, 255–258 (2001).
Hou, B., Reizis, B. & DeFranco, A.L. Toll-like receptors activate innate and adaptive immunity by using dendritic cell-intrinsic and -extrinsic mechanisms. Immunity 29, 272–282 (2008).
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004).
Vaishnava, S. et al. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011).
Abreu, M.T. Toll-like receptor signalling in the intestinal epithelium: how bacterial recognition shapes intestinal function. Nat. Rev. Immunol. 10, 131–144 (2010).
Kirkland, D. et al. B cell-intrinsic MyD88 signaling prevents the lethal dissemination of commensal bacteria during colonic damage. Immunity 36, 228–238 (2012).
Hou, B. et al. Selective utilization of Toll-like receptor and MyD88 signaling in B cells for enhancement of the antiviral germinal center response. Immunity 34, 375–384 (2011).
Damsker, J.M., Hansen, A.M. & Caspi, R.R. Th1 and Th17 cells: adversaries and collaborators. Ann. NY Acad. Sci. 1183, 211–221 (2010).
Strober, W. & Fuss, I.J. Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology 140, 1756–1767 (2011).
Benson, A., Pifer, R., Behrendt, C.L., Hooper, L.V. & Yarovinsky, F. Gut commensal bacteria direct a protective immune response against Toxoplasma gondii. Cell Host Microbe 6, 187–196 (2009).
Yarovinsky, F. et al. TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science 308, 1626–1629 (2005).
Kirkland, D. et al. B cell-intrinsic MyD88 signaling prevents the lethal dissemination of commensal bacteria during colonic damage. Immunity 36, 228–238 (2012).
Vaishnava, S. et al. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011).
Jansen, G.J. et al. Rapid identification of bacteria in blood cultures by using fluorescently labeled oligonucleotide probes. J. Clin. Microbiol. 38, 814–817 (2000).
Rozen, S. & Skaletsky, H. Primer3 on the WWW for general users and for biologist programmers. Methods Mol. Biol. 132, 365–386 (2000).
Bacchetti De Gregoris, T., Aldred, N., Clare, A.S. & Burgess, J.G. Improvement of phylum- and class-specific primers for real-time PCR quantification of bacterial taxa. J. Microbiol. Methods 86, 351–356 (2011).
Guo, X. et al. Development of a real-time PCR method for Firmicutes and Bacteroidetes in faeces and its application to quantify intestinal population of obese and lean pigs. Lett. Appl. Microbiol. 47, 367–373 (2008).
Doré, J., Sghir, A., Hannequart-Gramet, G., Corthier, G. & Pochart, P. Design and evaluation of a 16S rRNA-targeted oligonucleotide probe for specific detection and quantitation of human faecal Bacteroides populations. Syst. Appl. Microbiol. 21, 65–71 (1998).
Barman, M. et al. Enteric salmonellosis disrupts the microbial ecology of the murine gastrointestinal tract. Infect. Immun. 76, 907–915 (2008).
Hou, B., Reizis, B. & DeFranco, A.L. Toll-like receptors activate innate and adaptive immunity by using dendritic cell-intrinsic and -extrinsic mechanisms. Immunity 29, 272–282 (2008).
Acknowledgements
This research was supported by US National Institutes of Health (NIH) grants R01 AI085263 to F.Y. and R01 DK070855 to L.V.H. and by the Howard Hughes Medical Institute (L.V.H.). C.R.S. was supported in part by NIH grant A1005284-33. We would also like to thank C. Behrendt and C. Clements for germ-free mouse husbandry.
Author information
Authors and Affiliations
Contributions
F.Y. conceived the project. M.R., S.-h.H., C.L.W., D.K., A.B., C.R.S., J.M., S.V. and C.J.G. performed the experiments. A.L.D. and B.H. provided Myd88fl/fl mice, and L.V.H. provided germ-free mice. M.R., S.-h.H., C.L.W. and F.Y. analyzed data. M.R., C.L.W. and F.Y. wrote the manuscript, with all authors contributing to the writing and providing advice.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 2026 kb)
Rights and permissions
About this article
Cite this article
Raetz, M., Hwang, Sh., Wilhelm, C. et al. Parasite-induced TH1 cells and intestinal dysbiosis cooperate in IFN-γ-dependent elimination of Paneth cells. Nat Immunol 14, 136–142 (2013). https://doi.org/10.1038/ni.2508
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2508
This article is cited by
-
Gut microbiota-related metabolite alpha-linolenic acid mitigates intestinal inflammation induced by oral infection with Toxoplasma gondii
Microbiome (2023)
-
Gut commensal bacteria exacerbate toxoplasmosis associated with TgSheepCHn5 (ToxoDB#2) and TgRedpandaCHn1 (ToxoDB#20) through Th1 immune response
Parasitology Research (2023)
-
Toxoplasma gondii infection and its implications within the central nervous system
Nature Reviews Microbiology (2021)
-
Reg4 and complement factor D prevent the overgrowth of E. coli in the mouse gut
Communications Biology (2020)
-
Alarmin S100A11 initiates a chemokine response to the human pathogen Toxoplasma gondii
Nature Immunology (2019)