Abstract
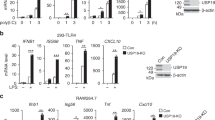
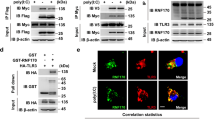
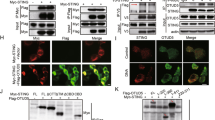
Balanced production of type I interferons and proinflammatory cytokines after engagement of Toll-like receptors (TLRs), which signal through adaptors containing a Toll–interleukin 1 receptor (TIR) domain, such as MyD88 and TRIF, has been proposed to control the pathogenesis of autoimmune disease and tumor responses to inflammation. Here we show that TRAF3, a ubiquitin ligase that interacts with both MyD88 and TRIF, regulated the production of interferon and proinflammatory cytokines in different ways. Degradative ubiquitination of TRAF3 during MyD88-dependent TLR signaling was essential for the activation of mitogen-activated protein kinases (MAPKs) and production of inflammatory cytokines. In contrast, TRIF-dependent signaling triggered noncanonical TRAF3 self-ubiquitination that activated the interferon response. Inhibition of degradative ubiquitination of TRAF3 prevented the expression of all proinflammatory cytokines without affecting the interferon response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Banchereau, J. & Pascual, V. Type I interferon in systemic lupus erythematosus and other autoimmune diseases. Immunity 25, 383–392 (2006).
Luo, J.L., Maeda, S., Hsu, L.C., Yagita, H. & Karin, M. Inhibition of NF-κB in cancer cells converts inflammation- induced tumor growth mediated by TNFα to TRAIL-mediated tumor regression. Cancer Cell 6, 297–305 (2004).
Lin, W.W. & Karin, M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Invest. 117, 1175–1183 (2007).
Akira, S. & Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511 (2004).
Karin, M., Lawrence, T. & Nizet, V. Innate immunity gone awry: linking microbial infections to chronic inflammation and cancer. Cell 124, 823–835 (2006).
Pasare, C. & Medzhitov, R. Toll-like receptors: linking innate and adaptive immunity. Adv. Exp. Med. Biol. 560, 11–18 (2005).
Vogel, S.N., Fitzgerald, K.A. & Fenton, M.J. TLRs: differential adapter utilization by toll-like receptors mediates TLR-specific patterns of gene expression. Mol. Interv. 3, 466–477 (2003).
Doyle, S. et al. IRF3 mediates a TLR3/TLR4-specific antiviral gene program. Immunity 17, 251–263 (2002).
Medzhitov, R. et al. MyD88 is an adaptor protein in the hToll/IL-1 receptor family signaling pathways. Mol. Cell 2, 253–258 (1998).
Yamamoto, M. et al. Cutting edge: a novel Toll/IL-1 receptor domain-containing adapter that preferentially activates the IFN-β promoter in the Toll-like receptor signaling. J. Immunol. 169, 6668–6672 (2002).
Hoebe, K. et al. Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature 424, 743–748 (2003).
Bin, L.H., Xu, L.G. & Shu, H.B. TIRP, a novel Toll/interleukin-1 receptor (TIR) domain-containing adapter protein involved in TIR signaling. J. Biol. Chem. 278, 24526–24532 (2003).
Yamamoto, M. et al. TRAM is specifically involved in the Toll-like receptor 4-mediated MyD88-independent signaling pathway. Nat. Immunol. 4, 1144–1150 (2003).
Fitzgerald, K.A. et al. Mal (MyD88-adapter-like) is required for Toll-like receptor-4 signal transduction. Nature 413, 78–83 (2001).
Horng, T., Barton, G.M. & Medzhitov, R. TIRAP: an adapter molecule in the Toll signaling pathway. Nat. Immunol. 2, 835–841 (2001).
Kawai, T. & Akira, S. TLR signaling. Semin. Immunol. 19, 24–32 (2007).
Hacker, H. et al. Specificity in Toll-like receptor signalling through distinct effector functions of TRAF3 and TRAF6. Nature 439, 204–207 (2006).
Oganesyan, G. et al. Critical role of TRAF3 in the Toll-like receptor-dependent and -independent antiviral response. Nature 439, 208–211 (2006).
Matsuzawa, A. et al. Essential cytoplasmic translocation of a cytokine receptor-assembled signaling complex. Science 321, 663–668 (2008).
Inoue, J., Gohda, J. & Akiyama, T. Characteristics and biological functions of TRAF6. Adv. Exp. Med. Biol. 597, 72–79 (2007).
Petersen, S.L. et al. Autocrine TNFα signaling renders human cancer cells susceptible to Smac-mimetic-induced apoptosis. Cancer Cell 12, 445–456 (2007).
Li, L. et al. A small molecule Smac mimic potentiates TRAIL- and TNFα-mediated cell death. Science 305, 1471–1474 (2004).
Vallabhapurapu, S. et al. Nonredundant and complementary functions of TRAF2 and TRAF3 in a ubiquitination cascade that activates NIK-dependent alternative NF-κB signaling. Nat. Immunol. 9, 1364–1370 (2008).
Werneburg, B.G., Zoog, S.J., Dang, T.T., Kehry, M.R. & Crute, J.J. Molecular characterization of CD40 signaling intermediates. J. Biol. Chem. 276, 43334–43342 (2001).
Kagan, J.C. et al. TRAM couples endocytosis of Toll-like receptor 4 to the induction of interferon-β. Nat. Immunol. 9, 361–368 (2008).
Núñez Miguel, R. et al. A dimer of the Toll-like receptor 4 cytoplasmic domain provides a specific scaffold for the recruitment of signalling adaptor proteins. PLoS One 2, e788 (2007).
Macia, E. et al. Dynasore, a cell-permeable inhibitor of dynamin. Dev. Cell 10, 839–850 (2006).
Lamothe, B. et al. Site-specific Lys-63-linked tumor necrosis factor receptor-associated factor 6 auto-ubiquitination is a critical determinant of IκB kinase activation. J. Biol. Chem. 282, 4102–4112 (2007).
Vaux, D.L. & Silke, J. IAPs, RINGs and ubiquitylation. Nat. Rev. Mol. Cell Biol. 6, 287–297 (2005).
Li, X., Yang, Y. & Ashwell, J.D. TNF-RII and c-IAP1 mediate ubiquitination and degradation of TRAF2. Nature 416, 345–347 (2002).
Uematsu, S. & Akira, S. Toll-like receptors and type I interferons. J. Biol. Chem. 282, 15319–15323 (2007).
O'Neill, L.A. & Bowie, A.G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat. Rev. Immunol. 7, 353–364 (2007).
Pineda, G., Ea, C.K. & Chen, Z.J. Ubiquitination and TRAF signaling. Adv. Exp. Med. Biol. 597, 80–92 (2007).
Yamamoto, M. et al. Key function for the Ubc13 E2 ubiquitin-conjugating enzyme in immune receptor signaling. Nat. Immunol. 7, 962–970 (2006).
Sato, S. et al. Essential function for the kinase TAK1 in innate and adaptive immune responses. Nat. Immunol. 6, 1087–1095 (2005).
Kawai, T., Adachi, O., Ogawa, T., Takeda, K. & Akira, S. Unresponsiveness of MyD88-deficient mice to endotoxin. Immunity 11, 115–122 (1999).
Keats, J.J. et al. Promiscuous mutations activate the noncanonical NF-κB pathway in multiple myeloma. Cancer Cell 12, 131–144 (2007).
Park, J.M. et al. Signaling pathways and genes that inhibit pathogen-induced macrophage apoptosis–CREB and NF-κB as key regulators. Immunity 23, 319–329 (2005).
Micheau, O. & Tschopp, J. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114, 181–190 (2003).
Lawrence, T., Bebien, M., Liu, G.Y., Nizet, V. & Karin, M. IKKα limits macrophage NF-κB activation and contributes to the resolution of inflammation. Nature 434, 1138–1143 (2005).
Beaumelle, B.D., Gibson, A. & Hopkins, C.R. Isolation and preliminary characterization of the major membrane boundaries of the endocytic pathway in lymphocytes. J. Cell Biol. 111, 1811–1823 (1990).
Johnson, G.L. & Bourne, H.R. Influence of cholera toxin on the regulation of adenylate cyclase by GTP. Biochem. Biophys. Res. Commun. 78, 792–798 (1977).
Iwamura, T. et al. Induction of IRF-3/-7 kinase and NF-κB in response to double-stranded RNA and virus infection: common and unique pathways. Genes Cells 6, 375–388 (2001).
Acknowledgements
We thank H. Ichijo (University of Tokyo) for providing A.M. with space and support for some of this work described above; S. Akira (Osaka University) for Myd88−/− mice; B. Beutler (Scripps Research Institute) for TrifLps2/Lps2 mice; R. Fonseca (Mayo Clinic) for multiple myeloma cells; X. Wang (University of Texas Southwestern Medical Center) for SM; H. Wang (St. Jude Children's Research Hospital) for generating monoclonal antibody HWA4C4, specific for K63-linked ubiquitin; I. Verma (Salk Institute) for pLV-CMV-delta 8.2; and Millipore for the antibody to K48-linked polyubiquitin. Supported by the National Institutes of Health (AI043477 to M.K. and AI52199 to D.A.A.V.), the American Cancer Society (M.K.), the American Lung Association of California (P.-H.T.), the Global Center of Excellence program (A.M.), the Toyobo Biotechnology Foundation (T.M.), the National Cancer Institute (CA21765 to D.A.A.V.) and the American Lebanese Syrian Associated Charities (D.A.A.V.).
Author information
Authors and Affiliations
Contributions
P.-H.T. and M.K. planned and designed all experiments and wrote the manuscript; P.-H.T. and A.M. did most experiments; W.Z. and T.M. helped with cell cultures, TRAF3 mutants and immunoprecipitation experiments; and D.A.A.V. provided the HWA4C4 K63-specific antibody to ubiquitin.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9, Supplementary Table 1 and Supplementary Methods (PDF 1490 kb)
Rights and permissions
About this article
Cite this article
Tseng, PH., Matsuzawa, A., Zhang, W. et al. Different modes of ubiquitination of the adaptor TRAF3 selectively activate the expression of type I interferons and proinflammatory cytokines. Nat Immunol 11, 70–75 (2010). https://doi.org/10.1038/ni.1819
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1819
This article is cited by
-
Targeted inhibition of PTPN22 is a novel approach to alleviate osteogenic responses in aortic valve interstitial cells and aortic valve lesions in mice
BMC Medicine (2023)
-
The Interaction of OTUB1 and TRAF3 Mediates NLRP3 Inflammasome Activity to Regulate TGF-β1-induced BEAS-2B Cell Injury
Applied Biochemistry and Biotechnology (2023)
-
Extracellular fibrinogen-binding protein released by intracellular Staphylococcus aureus suppresses host immunity by targeting TRAF3
Nature Communications (2022)
-
Influenza A virus use of BinCARD1 to facilitate the binding of viral NP to importin α7 is counteracted by TBK1-p62 axis-mediated autophagy
Cellular & Molecular Immunology (2022)
-
The toxicity of perfluorodecanoic acid is mainly manifested as a deflected immune function
Molecular Biology Reports (2022)