Abstract
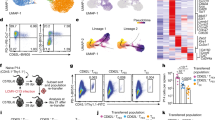
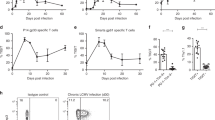
T cell exhaustion often occurs during chronic infection and prevents optimal viral control. The molecular pathways involved in T cell exhaustion remain poorly understood. Here we show that exhausted CD8+ T cells are subject to complex layers of negative regulation resulting from the coexpression of multiple inhibitory receptors. Exhausted CD8+ T cells expressed up to seven inhibitory receptors. Coexpression of multiple distinct inhibitory receptors was associated with greater T cell exhaustion and more severe infection. Regulation of T cell exhaustion by various inhibitory pathways was nonredundant, as blockade of the T cell inhibitory receptors PD-1 and LAG-3 simultaneously and synergistically improved T cell responses and diminished viral load in vivo. Thus, CD8+ T cell responses during chronic viral infections are regulated by complex patterns of coexpressed inhibitory receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Williams, M.A. & Bevan, M.J. Effector and memory CTL differentiation. Annu. Rev. Immunol. 25, 171–192 (2007).
Kaech, S.M. & Wherry, E.J. Heterogeneity and cell-fate decisions in effector and memory CD8+ T cell differentiation during viral infection. Immunity 27, 393–405 (2007).
Wherry, E.J. & Ahmed, R. Memory CD8 T-cell differentiation during viral infection. J. Virol. 78, 5535–5545 (2004).
Shin, H. & Wherry, E.J. CD8 T cell dysfunction during chronic viral infection. Curr. Opin. Immunol. 19, 408–415 (2007).
Zajac, A.J. et al. Viral immune evasion due to persistence of activated T cells without effector function. J. Exp. Med. 188, 2205–2213 (1998).
Barber, D.L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006).
Sharpe, A.H., Wherry, E.J., Ahmed, R. & Freeman, G.J. The function of programmed cell death 1 and its ligands in regulating autoimmunity and infection. Nat. Immunol. 8, 239–245 (2007).
Day, C.L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Petrovas, C. et al. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J. Exp. Med. 203, 2281–2292 (2006).
Trautmann, L. et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat. Med. 12, 1198–1202 (2006).
Zhang, J.Y. et al. PD-1 up-regulation is correlated with HIV-specific memory CD8+ T-cell exhaustion in typical progressors but not in long-term nonprogressors. Blood 109, 4671–4678 (2007).
Urbani, S. et al. PD-1 expression in acute hepatitis C virus (HCV) infection is associated with HCV-specific CD8 exhaustion. J. Virol. 80, 11398–11403 (2006).
Radziewicz, H. et al. Liver-infiltrating lymphocytes in chronic human hepatitis C virus infection display an exhausted phenotype with high levels of PD-1 and low levels of CD127 expression. J. Virol. 81, 2545–2553 (2006).
Boni, C. et al. Characterization of hepatitis B virus (HBV)-specific T-cell dysfunction in chronic HBV infection. J. Virol. 81, 4215–4225 (2007).
Boettler, T. et al. Expression of the interleukin-7 receptor alpha chain (CD127) on virus-specific CD8+ T cells identifies functionally and phenotypically defined memory T cells during acute resolving hepatitis B virus infection. J. Virol. 80, 3532–3540 (2006).
Wherry, E.J. et al. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 27, 670–684 (2007).
Kaufmann, D.E. et al. Upregulation of CTLA-4 by HIV-specific CD4+ T cells correlates with disease progression and defines a reversible immune dysfunction. Nat. Immunol. 8, 1246–1254 (2007).
Golub, T.R. et al. Molecular classification of cancer: class discovery and class prediction by gene expression monitoring. Science 286, 531–537 (1999).
McNerney, M.E., Lee, K.M. & Kumar, V. 2B4 (CD244) is a non-MHC binding receptor with multiple functions on natural killer cells and CD8+ T cells. Mol. Immunol. 42, 489–494 (2005).
Cai, G. et al. CD160 inhibits activation of human CD4+ T cells through interaction with herpesvirus entry mediator. Nat. Immunol. 9, 176–185 (2008).
Triebel, F. LAG-3: a regulator of T-cell and DC responses and its use in therapeutic vaccination. Trends Immunol. 24, 619–622 (2003).
Ahmed, R. et al. Selection of genetic variants of lymphocytic choriomeningitis virus in spleens of persistently infected mice. Role in suppression of cytotoxic T lymphocyte response and viral persistence. J. Exp. Med. 160, 521–540 (1984).
Wherry, E.J. et al. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 77, 4911–4927 (2003).
Wherry, E.J. et al. Antigen-independent memory CD8 T cells do not develop during chronic viral infection. Proc. Natl. Acad. Sci. USA 101, 16004–16009 (2004).
Workman, C.J. & Vignali, D.A. The CD4-related molecule, LAG-3 (CD223), regulates the expansion of activated T cells. Eur. J. Immunol. 33, 970–979 (2003).
Takai, T. Paired immunoglobulin-like receptors and their MHC class I recognition. Immunology 115, 433–440 (2005).
Rojo, S., Burshtyn, D.N., Long, E.O. & Wagtmann, N. Type I transmembrane receptor with inhibitory function in mouse mast cells and NK cells. J. Immunol. 158, 9–12 (1997).
Lau, L.L., Jamieson, B.D., Somasundaram, T. & Ahmed, R. Cytotoxic T-cell memory without antigen. Nature 369, 648–652 (1994).
Matloubian, M., Concepcion, R.J. & Ahmed, R. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J. Virol. 68, 8056–8063 (1994).
Kaech, S.M., Hemby, S., Kersh, E. & Ahmed, R. Molecular and functional profiling of memory CD8 T cell differentiation. Cell 111, 837–851 (2002).
Wherry, E.J. et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nat. Immunol. 4, 225–234 (2003).
Badovinac, V.P., Haring, J.S. & Harty, J.T. Initial T cell receptor transgenic cell precursor frequency dictates critical aspects of the CD8+ T cell response to infection. Immunity 26, 827–841 (2007).
Shin, H., Blackburn, S.D., Blattman, J.N. & Wherry, E.J. Viral antigen and extensive division maintain virus-specific CD8 T cells during chronic infection. J. Exp. Med. 204, 941–949 (2007).
Fuller, M.J. et al. Cutting edge: emergence of CD127high functionally competent memory T cells is compromised by high viral loads and inadequate T cell help. J. Immunol. 174, 5926–5930 (2005).
Betts, M.R. et al. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107, 4781–4789 (2006).
Blackburn, S.D., Shin, H., Freeman, G.J. & Wherry, E.J. Selective expansion of a subset of exhausted CD8 T cells by αPD-L1 blockade. Proc. Natl. Acad. Sci. USA 105, 15016–15021 (2008).
Hermans, I.F. et al. The VITAL assay: a versatile fluorometric technique for assessing CTL- and NKT-mediated cytotoxicity against multiple targets in vitro and in vivo. J. Immunol. Methods 285, 25–40 (2004).
Chlewicki, L.K. et al. Molecular basis of the dual functions of 2B4 (CD244). J. Immunol. 180, 8159–8167 (2008).
Butte, M.J. et al. Programmed death-1 ligand 1 interacts specifically with the B7–1 costimulatory molecule to inhibit T cell responses. Immunity 27, 111–122 (2007).
Kostense, S. et al. High viral burden in the presence of major HIV-specific CD8+ T cell expansions: evidence for impaired CTL effector function. Eur. J. Immunol. 31, 677–686 (2001).
Wherry, E.J., Blattman, J.N. & Ahmed, R. Low CD8 T-cell proliferative potential and high viral load limit the effectiveness of therapeutic vaccination. J. Virol. 79, 8960–8968 (2005).
Streeck, H. et al. Antigen load and viral sequence diversification determine the functional profile of HIV-1-specific CD8+ T cells. PLoS Med. 5, e100 (2008).
Freeman, G.J., Wherry, E.J., Ahmed, R. & Sharpe, A.H. Reinvigorating exhausted HIV-specific T cells via PD-1-PD-1 ligand blockade. J. Exp. Med. 203, 2223–2227 (2006).
Baixeras, E. et al. Characterization of the lymphocyte activation gene 3-encoded protein. A new ligand for human leukocyte antigen class II antigens. J. Exp. Med. 176, 327–337 (1992).
Hannier, S., Tournier, M., Bismuth, G. & Triebel, F. CD3/TCR complex-associated lymphocyte activation gene-3 molecules inhibit CD3/TCR signaling. J. Immunol. 161, 4058–4065 (1998).
Huang, C.T. et al. Role of LAG-3 in regulatory T cells. Immunity 21, 503–513 (2004).
Grosso, J.F. et al. LAG-3 regulates CD8+ T cell accumulation and effector function in murine self- and tumor-tolerance systems. J. Clin. Invest. 117, 3383–3392 (2007).
Workman, C.J. & Vignali, D.A. Negative regulation of T cell homeostasis by lymphocyte activation gene-3 (CD223). J. Immunol. 174, 688–695 (2005).
Workman, C.J. et al. Lymphocyte activation gene-3 (CD223) regulates the size of the expanding T cell population following antigen activation in vivo. J. Immunol. 172, 5450–5455 (2004).
Byun, H.J. et al. Proliferation of activated CD1d-restricted NKT cells is down-modulated by lymphocyte activation gene-3 signaling via cell cycle arrest in S phase. Cell Biol. Int. 31, 257–262 (2007).
Katz, H.R. et al. Mouse mast cell gp49B1 contains two immunoreceptor tyrosine-based inhibition motifs and suppresses mast cell activation when coligated with the high-affinity Fc receptor for IgE. Proc. Natl. Acad. Sci. USA 93, 10809–10814 (1996).
Laouar, A. et al. Cutting edge: distinct NK receptor profiles are imprinted on CD8 T cells in the mucosa and periphery during the same antigen challenge: role of tissue-specific factors. J. Immunol. 178, 652–656 (2007).
Kumar, V. & McNerney, M.E. A new self: MHC-class-I-independent natural-killer-cell self-tolerance. Nat. Rev. Immunol. 5, 363–374 (2005).
Ha, S.J. et al. Enhancing therapeutic vaccination by blocking PD-1-mediated inhibitory signals during chronic infection. J. Exp. Med. 205, 543–555 (2008).
Brooks, D.G. et al. IL-10 blockade facilitates DNA vaccine-induced T cell responses and enhances clearance of persistent virus infection. J. Exp. Med. 205, 533–541 (2008).
Kotturi, M.F. et al. The CD8+ T-cell response to lymphocytic choriomeningitis virus involves the L antigen: uncovering new tricks for an old virus. J. Virol. 81, 4928–4940 (2007).
Acknowledgements
We thank E. Long, V. Kumar and S. Reiner for comments and suggestions, and B. Laidlaw for critically reading the manuscript. Supported by the National Institute of Allergy and Infectious Diseases (AI071309 to E.J.W and HHSN26620050030C to E.J.W. and G.J.F.) and the Bill and Melinda Gates Foundation Grand Challenge in Global Health (G.J.F. and E.J.W.).
Author information
Authors and Affiliations
Contributions
S.D.B. and E.J.W. designed the experiments; S.D.B. did the experiments with assistance from H.S., T.Z. and A.P.; S.D.B. and E.J.W. analyzed results with input from W.N.H. and M.R.B. and wrote the manuscript; and C.J.W., G.J.F. and D.A.A.V. provided crucial reagents and intellectual input.
Corresponding author
Ethics declarations
Competing interests
G.J.F. has patents on the PD-1 pathway and receives royalties from those patents.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–3 (PDF 2740 kb)
Rights and permissions
About this article
Cite this article
Blackburn, S., Shin, H., Haining, W. et al. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat Immunol 10, 29–37 (2009). https://doi.org/10.1038/ni.1679
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1679
This article is cited by
-
Inhibition of SFTSV replication in humanized mice by a subcutaneously administered anti-PD1 nanobody
EMBO Molecular Medicine (2024)
-
Turning up the heat: CTLA4 blockade in urothelial cancer
Nature Reviews Urology (2024)
-
Evaluation of changes in exhausted T lymphocytes and miRNAs expression in the different trimesters of pregnancy in pregnant women
Molecular Biology Reports (2024)
-
Focus on T cell exhaustion: new advances in traditional Chinese medicine in infection and cancer
Chinese Medicine (2023)
-
The “Great Debate” at Melanoma Bridge 2022, Naples, December 1st–3rd, 2022
Journal of Translational Medicine (2023)