Abstract
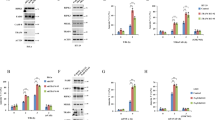
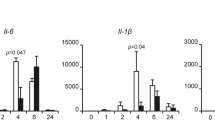
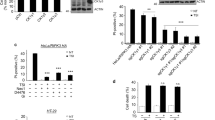
Despite rapid progress in elucidating the molecular mechanisms of activation of the kinase IKK, the processes that regulate IKK deactivation are still unknown. Here we demonstrate that CUE domain–containing 2 (CUEDC2) interacted with IKKα and IKKβ and repressed activation of the transcription factor NF-κB by decreasing phosphorylation and activation of IKK. Notably, CUEDC2 also interacted with GADD34, a regulatory subunit of protein phosphatase 1 (PP1). We found that IKK, CUEDC2 and PP1 existed in a complex and that IKK was released from the complex in response to inflammatory stimuli such as tumor necrosis factor. CUEDC2 deactivated IKK by recruiting PP1 to the complex. Therefore, CUEDC2 acts as an adaptor protein to target IKK for dephosphorylation and inactivation by recruiting PP1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chen, G. & Goeddel, D.V. TNF-R1 signaling: a beautiful pathway. Science 296, 1634–1635 (2002).
Wajant, H., Pfizenmaier, K. & Scheurich, P. Tumor necrosis factor signaling. Cell Death Differ. 10, 45–65 (2003).
Karin, M. & Ben-Neriah, Y. Phosphorylation meets ubiquitination: the control of NF-κB activity. Annu. Rev. Immunol. 18, 621–663 (2000).
Maniatis, T. A ubiquitin ligase complex essential for the NF-κB, Wnt/Wingless, and Hedgehog signaling pathways. Genes Dev. 13, 505–510 (1999).
Häcker, H. & Karin, M. Regulation and function of IKK and IKK-related kinases. Sci. STKE 357, re13 (2006).
Li, Q., Estepa, G., Memet, S., Israel, A. & Verma, I.M. Complete lack of NF-κB activity in IKK1 and IKK2 double deficient mice: additional defect in neurulation. Genes Dev. 14, 1729–1733 (2000).
Ghosh, S. & Karin, M. Missing pieces in the NF-κB puzzle. Cell 109, S81–S96 (2002).
Yamaoka, S. et al. Complementation cloning of NEMO, a component of the IκB kinase complex essential for NF-κB activation. Cell 93, 1231–1240 (1998).
Delhase, M., Hayakawa, M., Chen, Y. & Karin, M. Positive and negative regulation of IκB kinase activity through IKKβ subunit phosphorylation. Science 284, 309–313 (1999).
Mercurio, F. et al. IKK-1 and IKK-2: cytokine-activated IκB kinases essential for NF-κB activation. Science 278, 860–866 (1997).
Jackman, J., Alamo, I. & Fornace, A.J. Jr. Genotoxic stress confers preferential and coordinate messenger RNA stability on the five gadd genes. Cancer Res. 54, 5656–5662 (1994).
Aggen, J.B., Nairn, A.C. & Chamberlin, R. Regulation of protein phosphatase-1. Chem. Biol. 7, R13–R23 (2000).
Bollen, M. Combinatorial control of protein phosphatase-1. Trends Biochem. Sci. 26, 426–431 (2001).
Egloff, M.P. et al. Structural basis for the recognition of regulatory subunits by the catalytic subunit of protein phosphatase 1. EMBO J. 16, 1876–1887 (1997).
Donaldson, K.M., Yin, H., Gekakis, N., Supek, F. & Hoazeiro, C.A. Ubiquitin signals protein trafficking via interaction with a novel ubquitin binding domain in the membrane fusion regulator, Vps9p. Curr. Biol. 13, 258–262 (2003).
Ponting, C.P. Proteins of the endoplasmic-retivulum-associated degradation pathway: domain detection and function prediction. Biochem. J. 351, 527–535 (2000).
Shih, S.C. et al. A ubiquitin-binding motif required for intramolecular monoubiquitylation, the CUE domain. EMBO J. 22, 1273–1281 (2003).
Zhang, P.J. et al. CUE domain containing 2 regulates degradation of progesterone receptor by ubiquitin–proteasome. EMBO J. 26, 1831–1842 (2007).
Bender, K., Gottlicher, M., Whiteside, S., Rahmsdorf, H.J. & Herrlich, P. Sequential DNA damage-independent and -dependent activation of NF-κB by UV. EMBO J. 17, 5170–5181 (1998).
Li, N. & Karin, M. Ionizing radiation and short wavelength UV activate NF-κB through two distinct mechanisms. Proc. Natl. Acad. Sci. USA 95, 13012–13017 (1998).
DiDonato, J.A., Hayakawa, M., Rothwarf, D.M., Zandi, E. & Karin, M. A cytokine-responsive IκB kinase that activates the transcription factor NF-κB. Nature 388, 548–554 (1997).
Devin, A. et al. The distinct roles of TRAF2 and RIP in IKK activation by TNF-R1: TRAF2 recruits IKK to TNF-R1 while RIP mediates IKK activation. Immunity 12, 419–429 (2000).
Devin, A. et al. The α and β subunits of IκB kinase (IKK) mediate TRAF2-dependent IKK recruitment to tumor necrosis factor (TNF) receptor 1 in response to TNF. Mol. Cell. Biol. 21, 3986–3994 (2001).
Naka, T., Nishimoto, N. & Kishimoto, T. The paradigm of IL-6: from basic science to medicine. Arthritis Res. 4, S233–S242 (2002).
Yang, J. et al. The essential role of MEKK3 in TNF-induced NF-κB activation. Nat. Immunol. 2, 620–624 (2001).
Huang, Q. et al. Differential regulation of interleukin 1 receptor and Toll-like receptor signaling by MEKK3. Nat. Immunol. 5, 98–103 (2004).
Hsu, H., Huang, J., Shu, H.B., Baichwal, V. & Goeddel, D.V. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity 4, 387–396 (1996).
Nakano, H. et al. Differential regulation of IκB kinase α and β by two upstream kinases, NF-κB-inducing kinase and mitogen-activated protein kinase/ERK kinase kinase-1. Proc. Natl. Acad. Sci. USA 95, 3537–3542 (1998).
Brummelkamp, T.R., Nijman, S.M.B., Dirac, A.M.G. & Bernards, R. Loss of the cylindromatosis tumour suppressor inhibits apoptosis by activating NF-κB. Nature 424, 797–801 (2003).
Kovalenko, A. et al. The tumour suppressor CYLD negatively regulates NF-κB signaling by deubiquitination. Nature 424, 801–805 (2003).
Lee, E.G. et al. Failure to regulate TNF-induced NF-κB and cell death responses in A20-deficient mice. Science 289, 2350–2354 (2000).
Trompouki, E. et al. CYLD is a deubiquitinating enzyme that negatively regulates NF-κB activation by TNFR family members. Nature 424, 793–796 (2003).
Wertz, I.E. et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-κB signaling. Nature 430, 694–699 (2004).
Jobin, C. & Sartor, R.B. The IκB/NF-κB system: a key determinant of mucosalinflammation and protection. Am. J. Physiol. Cell Physiol. 278, C451–C462 (2000).
Li, Q. & Verma, I.M. NF-κB regulation in the immune system. Nat. Rev. Immunol. 2, 725–734 (2002).
Prajapati, S.S., Verma, U., Yamamoto, Y.M., Kwak, Y.T. & Gaynor, R.B. Protein phosphatase 2Cβ association with the IκB kinase complex is involved in regulating NF-κB activity. J. Biol. Chem. 279, 1739–1746 (2004).
Li, S.T., Wang, L.Y., Berman, M.A., Zhang, Y. & Dorf, M.E. RNAi screen in mouse astrocytes identifies phosphatases that regulate NF-κB signaling. Mol. Cell 24, 497–509 (2006).
Man, J.H. et al. PIAS3 induction of PRB sumoylation represses PRB transactivation by destabilizing its retention in the nucleus. Nucleic Acids Res. 34, 5552–5566 (2006).
Pan, X. et al. Ubc9 interacts with SOX4 and represses its transcriptional activity. Biochem. Biophys. Res. Commun. 344, 727–734 (2006).
Acknowledgements
We thank Z.G. Liu (National Cancer Institute, National Institutes of Health) for human IKKα and IKKβ plasmids and TRAF2-deficient MEFs; M.W. Mayo (University of Virginia) for the p65 expression plasmid; X. Lin (Anderson Cancer Center) for the RIP expression vector; X.X. Li (Cleveland Clinic Foundation) for construction of pE-selectin-Luc; G. Haegeman (University of Gent, Gent) for the pIL-6-Luc plasmid; and Z.G. Liu for discussions and technical help. Supported by the National Natural Science Foundation of China (30525021, 30672357, 30500583 and 30321003), the Major State Basic Research Development Program of China (973 Program; 2004CB518800) and the National High Technology Research and Development Program of China (2006AA02Z340).
Author information
Authors and Affiliations
Contributions
H.-Y.L., A.-L.L. and X.-M.Z. contributed to all studies; H.L. contributed to the yeast two-hybrid experiments; C.-H.W. and J.-H.M. participated in study design and analyzed data; J.Z., W.-H.L., and X.P. developed the stable cell lines; T.Z. and W.-L.G. contributed to the preparation of antibodies; and Y.-F.G., J.-Y.Z. and P.-J.Z. did the primary macrophage study.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Table 1 and Supplementary Methods (PDF 465 kb)
Rights and permissions
About this article
Cite this article
Li, HY., Liu, H., Wang, CH. et al. Deactivation of the kinase IKK by CUEDC2 through recruitment of the phosphatase PP1. Nat Immunol 9, 533–541 (2008). https://doi.org/10.1038/ni.1600
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1600
This article is cited by
-
Emerging roles of the Protein Phosphatase 1 (PP1) in the context of viral infections
Cell Communication and Signaling (2024)
-
Protein phosphatase 1 regulatory subunit 15 A promotes translation initiation and induces G2M phase arrest during cuproptosis in cancers
Cell Death & Disease (2024)
-
Phosphatases in toll-like receptors signaling: the unfairly-forgotten
Cell Communication and Signaling (2021)
-
Protein phosphatases in TLR signaling
Cell Communication and Signaling (2021)
-
Elimination of negative feedback in TLR signalling allows rapid and hypersensitive detection of microbial contaminants
Scientific Reports (2021)