Abstract
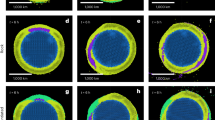
Exposed rocks at underwater volcanoes and ridges host complex, abundant and diverse microbial communities1,2,3. The volcanic glasses associated with these features constitute one of the most geochemically reactive components of the Earth’s crust. The most commonly held hypothesis is that their oxidation in sea water provides the energy necessary to establish a seafloor biosphere4,5,6,7. However, this hypothesis has yet to be directly tested. Here we used synchrotron-based X-ray microprobe mapping, X-ray absorption spectroscopy and high-resolution scanning and transmission electron microscopy techniques to examine the initial chemical changes that occur as the glassy rims of young pillow basalts are colonized by microbial organisms at Loihi seamount, Hawaii. We found little evidence of basalt dissolution. Instead, microbial biofilms were intimately associated with Fe(III)- and Mn(IV)-oxides that had precipitated from sea water onto the fresh basalt surfaces. These accumulations of secondary minerals probably represent the earliest stages of ferromanganese crust formation. We suggest that fluid-derived energy sources, such as dissolved and particulate Fe(II), Mn(II) and organic matter, may support the microbial communities colonizing seafloor rocks to a greater degree than local rock dissolution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Santelli, C. M. et al. Abundance and diversity of microbial life in ocean crust. Nature 453, 653–656 (2008).
Mason, O. U. et al. The phylogeny of endolithic microbes associated with marine basalts. Environ. Microbiol. 9, 2539–2550 (2007).
Lysnes, K. et al. Microbial community diversity in seafloor basalt from the Arctic spreading ridges. FEMS Microbiol. Ecol. 50, 213–230 (2004).
Fisk, M. R., Giovannoni, S. J. & Thorseth, I. H. Alteration of volcanic glass: Textural evidence of microbial activity. Science 281, 978–979 (1998).
Thorseth, H. I. et al. Diversity of life in ocean floor basalt. Earth Planet. Sci. Lett. 194, 31–37 (2001).
Bach, W. & Edwards, K. J. Iron and sulfide oxidation within the basaltic ocean crust: Implications for chemolithoautotrophic microbial biomass production. Geochim. Cosmochim. Acta 67, 3871–3887 (2003).
Staudigel, H. et al. 3.5-billion years of glass bioalteration: Volcanic rocks as a basis for microbial life? Earth Sci. Rev. 89, 156–176 (2008).
Einen, J., Thorseth, I. H. & Ovreas, L. Enumeration of Archaea and Bacteria in seafloor basalt using real-time quantitative PCR and fluorescence microscopy. FEMS Microbiol. Lett. 282, 182–187 (2008).
Templeton, A. S., Staudigel, H. & Tebo, B. M. Diverse Mn(II)-oxidizing bacteria isolated from submarine basalts at Loihi Seamount. Geomicrobiol. J. 22, 127–139 (2005).
Thorseth, H. I., Pedersen, R. B. & Christie, D. M. Microbial alteration of 0–30 Ma seafloor and subseafloor basaltic glasses from the Australian–Antarctic Discordance. Earth Planet. Sci. Lett. 215, 237–247 (2003).
Staudigel, H. & Hart, S. R. Alteration of basaltic glass: Mechanisms and significance for the oceanic crust-seawater budget. Geochim. Cosmochim. Acta 47, 337–350 (1983).
Kruber, C., Thorseth, I. H. & Pedersen, R. B. Seafloor alteration of basaltic glass: Textures, geochemistry, and endolithic microorganisms. Geochem. Geophys. Geosyst. 9, Q12002 (2008).
Webb, S. M., Tebo, B. M. & Bargar, J. R. Structural characterization of biogenic Mn oxides produced in seawater by the marine bacillus sp strain SG-1. Am. Mineral. 90, 1342–1357 (2005).
Hansel, C. M., Benner, S. G., Nico, P. & Fendorf, S. Structural constraints of ferric (hydr)oxides on dissimilatory iron reduction and the fate of Fe(II). Geochim. Cosmochim. Acta 68, 3217–3229 (2004).
Moore, J. G., Clague, D. A. & Normark, W. R. Diverse basalt types from Loihi seamount, Hawaii. Geology 10, 88–92 (1982).
De Carlo, E. H., McMurtry, G. M. & Yeh, H.-W. Geochemistry of hydrothermal deposits from Loihi submarine volcano, Hawaii. Earth Planet. Sci. Lett. 66, 438–449 (1983).
Karl, D. M., McMurtry, G. M., Malahoff, A. & Garcia, M. O. Loihi Seamount, Hawaii: A mid-plate volcano with a distinctive hydrothermal system. Nature 335, 533–535 (1988).
Emerson, D. & Moyer, C. L. Neutrophilic Fe-oxidizing bacteria are abundant at the Loihi seamount hydrothermal vents and play a major role in Fe-oxide deposition. Appl. Environ. Microbiol. 68, 3085–3093 (2002).
Malahoff, A., Kolotyrkina, I. Y., Midson, B. P. & Massoth, G. J. A decade of exploring a submarine intraplate volcano: Hydrothermal manganese and iron at Lo’ihi volcano, Hawai’i. Geochem. Geophys. Geosyst. 7, Q06002 (2006).
Lupton, J. E. A far-field hydrothermal plume from Loihi Seamount. Science 272, 976–979 (1996).
Boyle, E. A., Bergquist, B. A., Kayser, R. A. & Mahowald, N. Iron, manganese, and lead at Hawaii Ocean Time-series station ALOHA: Temporal variability and an intermediate water hydrothermal plume. Geochim. Cosmochim. Acta 69, 933–952 (2005).
Koschinsky, A. & Halbach, P. Sequential leaching of marine ferromanganese precipitates: Genetic implications. Geochim. Cosmochim. Acta 59, 5113–5132 (1995).
Hein, J. R. et al. Iron and manganese oxide mineralization in the Pacific. Geol. Soc. Lond. Spec. Pub. 119, 123–138 (1997).
Fortin, D., Ferris, F. G. & Scott, S. D. Formation of Fe-silicate and Fe-oxides on bacterial surfaces in samples collected near hydrothermal vents on the Southern Explorer Ridge in the northeast Pacific Ocean. Am. Min. 83, 1399–1408 (1998).
Alt, J. C. Hydrothermal oxide and nontronite deposits on seamounts in the eastern Pacific. Mar. Geol. 81, 227–239 (1988).
Toner, B. M. et al. Preservation of iron(II) by carbon-rich matrices in a hydrothermal plume. Nature Geosci. 2, 197–201 (2009).
Hein, J. R., Koschinsky, A. & Halliday, A. N. Global occurrence of tellurium-rich ferromanganese crusts and a model for the enrichment of tellurium. Geochim. Cosmochim. Acta 67, 1117–1127 (2003).
Sunda, W. G. & Kieber, D. J. Oxidation of humic substances by manganese oxides yields low-molecular-weight organic substrates. Nature 367, 62–64 (1994).
Mason, O. U. et al. Prokaryotic diversity, distribution, and insights into their role in biogeochemical cycling in marine basalts. ISME J. 3, 231–242 (2008).
Santelli, C. M., Edgcomb, V. P., Bach, W. & Edwards, K. J. The diversity and abundance of bacteria inhabiting seafloor lavas positively correlate with rock alteration. Environ. Microbiol. 11, 86–98 (2009).
Webb, S. M. SIXpack: A graphical user interface for XAS analysis using IFEFFIT. Phys. Scr. 2005, 1011–1014 (2005).
Acknowledgements
We thank C. Sheehan (OGI) and the crews of the RV Ka’imikai-o-Kanaloa, RV Kilo Moana and Pisces V (Hawaii Undersea Research Lab), RV Thomas G. Thompson (University of Washington) and ROV Jason II (Woods Hole Oceanographic Institution), as well as the principal investigators and collaborators associated with FeMO, the NSF Fe Microbial Observatory at Loihi seamount (MCB-0348668). We also thank C. Hansel at Harvard University for providing reference EXAFS spectra of Fe-bearing model compounds. This work was directly supported by the National Science Foundation grant OCE-0433629 (H.S., B.M.T., A.S.T.), the David and Lucille Packard Foundation (A.S.T.) and a NASA Predoctoral Fellowship (E.J.K.). The synchrotron work was conducted on beamlines 2-3 and 11-2 at the Stanford Synchrotron Radiation Lightsource (SSRL), a national user facility operated by Stanford University on behalf of the Department of Energy, Office of Basic Energy Sciences, through the Structural Molecular Biology Program, supported by DOE Office of Biological and Environmental Research and the National Institutes of Health. The FIB-milling and high-resolution SEM and TEM analyses were conducted at the Environmental Molecular Sciences Laboratory (EMSL), a national scientific user facility at the Pacific Northwest National Laboratory also supported by the DOE Office of Biological and Environmental Research. A. Buxbaum, FEI Corp., also provided valuable technical assistance in the FIB-SEM preparation.
Author information
Authors and Affiliations
Contributions
A.S.T., E.J.K., D.L.E., B.E.B., B.M.T. and H.S. all participated in sample planning and collection. A.S.T., E.J.K., D.L.E. and S.M.W. each led aspects of the synchrotron-based X-ray spectroscopy and microprobe mapping data collection and analysis. B.W.A. and A.C.D. conducted the electron microscopy with input from A.S.T. and B.M.T. A.S.T. wrote the manuscript with input from all of the authors.
Corresponding author
Supplementary information
Supplementary Information
Supplementary Information (PDF 1643 kb)
Rights and permissions
About this article
Cite this article
Templeton, A., Knowles, E., Eldridge, D. et al. A seafloor microbial biome hosted within incipient ferromanganese crusts. Nature Geosci 2, 872–876 (2009). https://doi.org/10.1038/ngeo696
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ngeo696
This article is cited by
-
Microbial Diversity of Deep-Sea Ferromanganese Crust Field in the Rio Grande Rise, Southwestern Atlantic Ocean
Microbial Ecology (2021)
-
Modern precipitation of hydrogenetic ferromanganese minerals during on-site 15-year exposure tests
Scientific Reports (2020)
-
Significant contribution of subseafloor microparticles to the global manganese budget
Nature Communications (2019)
-
Diversity patterns and isolation of Planctomycetes associated with metalliferous deposits from hydrothermal vent fields along the Valu Fa Ridge (SW Pacific)
Antonie van Leeuwenhoek (2018)
-
Under the sea: microbial life in volcanic oceanic crust
Nature Reviews Microbiology (2011)