Abstract
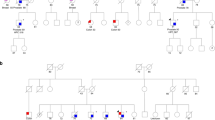
Although prostate cancer is the most common non-cutaneous malignancy diagnosed in men in the United States1,2, little is known about inherited factors that influence its genetic predisposition3,4,5. Here we report that germline mutations in the gene encoding 2′-5′-oligoadenylate(2-5A)–dependent RNase L (RNASEL)6,7,8 segregate in prostate cancer families that show linkage to the HPC1 (hereditary prostate cancer 1) region at 1q24–25 (ref. 9). We identified RNASEL by a positional cloning/candidate gene method, and show that a nonsense mutation and a mutation in an initiation codon of RNASEL segregate independently in two HPC1-linked families. Inactive RNASEL alleles are present at a low frequency in the general population. RNASEL regulates cell proliferation and apoptosis through the interferon-regulated 2-5A pathway and has been suggested to be a candidate tumor suppressor gene10,11,12. We found that microdissected tumors with a germline mutation showed loss of heterozygosity and loss of RNase L protein, and that RNASEL activity was reduced in lymphoblasts from heterozyogous individuals compared with family members who were homozygous with respect to the wildtype allele. Thus, germline mutations in RNASEL may be of diagnostic value, and the 2-5A pathway might provide opportunities for developing therapies for those with prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hankey, B.F. et al. Cancer surveillance series: interpreting trends in prostate cancer—part I: evidence of the effects of screening in recent prostate cancer incidence, mortality, and survival rates. J. Natl Cancer Inst. 91, 1017–1024 (1999).
Dennis, L.K. & Resnick, M.I. Analysis of recent trends in prostate cancer incidence and mortality. Prostate 42, 247–252 (2000).
Steinberg, G.D., Carter, B.S., Beaty, T.H., Childs, B. & Walsh, P.C. Family history and the risk of prostate cancer. Prostate 17, 337–347 (1990).
Carter, B.S., Beaty, T.H., Steinberg, G.D., Childs, B. & Walsh, P.C. Mendelian inheritance of familial prostate cancer. Proc. Natl Acad. Sci. USA 89, 3367–3371 (1992).
Ostrander, E.A. & Stanford, J.L. Genetics of prostate cancer: too many loci, too few genes. Am. J. Hum. Genet. 67, 1367–1375 (2000).
Clemens, M.J. & Williams, B.R. Inhibition of cell-free protein synthesis by pppA2′p5′A2′p5′A: a novel oligonucleotide synthesized by interferon-treated L cell extracts. Cell 13, 565–572 (1978).
Floyd-Smith, G., Slattery, E. & Lengyel, P. Interferon action: RNA cleavage pattern of a (2′-5′)oligoadenylate-dependent endonuclease. Science 212, 1030–1032 (1981).
Zhou, A., Hassel, B.A. & Silverman, R.H. Expression cloning of 2-5A-dependent RNAase: a uniquely regulated mediator of interferon action. Cell 72, 753–765 (1993).
Smith, J.R. et al. Major susceptibility locus for prostate cancer on chromosome 1 suggested by a genome-wide search. Science 274, 1371–1374 (1996).
Hassel, B.A., Zhou, A., Sotomayor, C., Maran, A. & Silverman, R.H. A dominant negative mutant of 2-5A-dependent RNase suppresses antiproliferative and antiviral effects of interferon. EMBO J. 12, 3297–3304 (1993).
Zhou A., et al. Interferon action and apoptosis are defective in mice devoid of 2′,5′- oligoadenylate-dependent RNase L. EMBO J. 16, 6355–6363 (1997).
Lengyel, P. Tumor-suppressor genes: news about the interferon connection. Proc. Natl Acad. Sci. USA 90, 5893–5895 (1993).
Carpten, J.D. et al. A 6-Mb high-resolution physical and transcription map encompassing the hereditary prostate cancer 1 (HPC1) region. Genomics 64, 1–14 (2000).
Sood, R. et al. Cloning and characterization of 13 novel transcripts and the human rgs8 gene from the 1q25 region encompassing the hereditary prostate cancer (hpc1) locus. Genomics 73, 211–222 (2001).
Kerr, I.M. & Brown, R.E. pppA2′p5′A2′p5′A: an inhibitor of protein synthesis synthesized with an enzyme fraction from interferon-treated cells. Proc. Natl Acad. Sci. USA 75, 256–260 (1978).
Sherman, F., McKnight, G. & Stewart, J.W. AUG is the only initiation codon in eukaryotes. Biochim. Biophys. Acta. 609, 343–346 (1980).
Hartge, P., Struewing, J.P., Wacholder, S., Brody, L.C. & Tucker, M.A. The prevalence of common BRCA1 and BRCA2 mutations among Ashkenazi Jews. Am. J. Hum. Genet. 64, 963–970 (1999).
Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 411, 603–606 (2001).
Dong, B. & Silverman, R.H. 2-5A-dependent RNase molecules dimerize during activation by 2-5A. J. Biol. Chem. 270, 4133–4137 (1995).
Silverman, R.H., Skehel, J.J., James, T.C., Wreschner, D.H. & Kerr, I.M. rRNA cleavage as an index of ppp(A2′p)nA activity in interferon-treated encephalomyocarditis virus-infected cells. J. Virol. 46, 1051–1055 (1983).
Tnani, M. & Bayard, B.A. Lack of 2′,5′-oligoadenylate-dependent RNase expression in the human hepatoma cell line HepG2. Biochim. Biophys. Acta 1402, 139–150 (1998).
Isaacs, J.T., Furuya, Y. & Berges, R. The role of androgen in the regulation of programmed cell death/apoptosis in normal and malignant prostatic tissue. Semin. Cancer Biol. 5, 391–400 (1994).
Kaighn, M.E., Narayan, K.S., Ohnuki, Y., Lechner, J.F. & Jones, L.W. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest. Urol. 17, 16–23 (1979).
Acknowledgements
We wish to thank the affected individuals and their family members who made this study possible. We thank D. Freije, H. Suzuki, E. Wilkens, A. Kibel, G. Bova, S. Gregory and T. Bonner for contributions to earlier phases of this work; F. S. Collins for input and comments; J. Qian for FISH analyses; M. Emmert-Buck for help in laser-capture microdissection; J. Hicks for immunohistochemistry; and J.R. Okicki for synthesizing the fluorescein-tagged 2-5A.This work was supported in part by grants from the PHS (SPORE), DOD, CaPCURE (W.I.) and the Fund for Research and Progress in Urology, the Johns Hopkins University, the Swedish Cancer Society and the SSF Genome Program (H.G.), the V Foundation for Cancer Research (J.S.), the Finnish Cultural Foundation, the Helsingin Sanomat Foundation, the Paulo Foundation, the Ella & Georg Ehrnrooth Foundation and the Maud Kuistila Foundation (N.N.), the NIH (R.J. and R.H.S.), and DOD (J.X.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Carpten, J., Nupponen, N., Isaacs, S. et al. Germline mutations in the ribonuclease L gene in families showing linkage with HPC1. Nat Genet 30, 181–184 (2002). https://doi.org/10.1038/ng823
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng823
This article is cited by
-
CRISPR/Cas9 model of prostate cancer identifies Kmt2c deficiency as a metastatic driver by Odam/Cabs1 gene cluster expression
Nature Communications (2024)
-
Carriage of mutations R462Q (rs 486907) and D541E (rs 627928) of the RNASEL gene and risk factors in patients with prostate cancer in Burkina Faso
BMC Medical Genomics (2022)
-
Is RNASEL:p.Glu265* a modifier of early-onset breast cancer risk for carriers of high-risk mutations?
BMC Cancer (2018)
-
IFNL4-ΔG is associated with prostate cancer among men at increased risk of sexually transmitted infections
Communications Biology (2018)
-
Exome screening to identify loss-of-function mutations in the rhesus macaque for development of preclinical models of human disease
BMC Genomics (2016)