Abstract
Idiopathic congenital nystagmus is characterized by involuntary, periodic, predominantly horizontal oscillations of both eyes. We identified 22 mutations in FRMD7 in 26 families with X-linked idiopathic congenital nystagmus. Screening of 42 singleton cases of idiopathic congenital nystagmus (28 male, 14 females) yielded three mutations (7%). We found restricted expression of FRMD7 in human embryonic brain and developing neural retina, suggesting a specific role in the control of eye movement and gaze stability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
06 June 2011
In the version of this article initially published, the author Andrew Bastawrous was omitted from the author list. The error has been corrected in the HTML and PDF versions of the article.
References
Stayte, M., Reeves, B. & Wortham, C. Br. J. Ophthalmol. 77, 228–232 (1993).
Pilling, R.F., Thompson, J.R. & Gottlob, I. Br. J. Ophthalmol. 89, 1278–1281 (2005).
Jacobs, J.B. & Dell'Osso, L.F. J. Vis. 4, 604–625 (2004).
Gottlob, I.Nystagmus. Curr. Opin. Ophthalmol. 12, 378–383 (2001).
Kerrison, J.B., Vagefi, M.R., Barmada, M.M. & Maumenee, I.H. Am. J. Hum. Genet. 64, 600–607 (1999).
Kerrison, J.B., Giorda, R., Lenart, T.D., Drack, A.V. & Maumenee, I.H. Ophthalmic Genet. 22, 241–248 (2001).
Zhang, B. et al. Hum. Genet. 116, 128–131 (2005).
Cabot, A. et al. Am. J. Hum. Genet. 64, 1141–1146 (1999).
Tarpey, P. et al. Am. J. Hum. Genet. 75, 318–324 (2004).
Berman, H., Henrick, K. & Nakamura, H. Nat. Struct. Biol. 10, 980 (2003).
Han, B.G., Nunomura, W., Takakuwa, Y., Mohandas, N. & Jap, B.K. Nat. Struct. Biol. 7, 871–875 (2000).
Emsley, P. & Cowtan, K. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Kubo, T. et al. J. Neurosci. 22, 8504–8513 (2002).
Toyofuku, T. et al. Nat. Neurosci. 8, 1712–1719 (2005).
Acknowledgements
This project was funded by the Wellcome Trust, Medisearch Leicester and The Ulverscroft Foundation. The human embryonic material was provided by the Joint MRC-Wellcome Trust Human Developmental Biology Resource at the Institute of Human Genetics, Newcastle upon Tyne, UK (http://www.hdbr.org).
Author information
Authors and Affiliations
Contributions
This study was designed by A.B., I.G., R.C.T., F.L.R., M.R.S., S.T. and N.S.; phenotype assessment was performed by I.G., S.T., N.S., E.O.R., M.A., M.S., R.J.M., R.D.R., A.L., S.L., M.K., G.W., R.P.G., C.D., K.D., I.A., A.A.Z., C.P., O.C.B., S.J., M.C.B., D.G.H. and R.W.H.; DNA extraction, linkage analysis, sequencing, in situ hybridization and molecular modeling were performed by F.L.R., P.T., S.T., R.C.T., U.M., S.L., C.J.T., E.O.R., M.S., C.D.V., R.D.M., L.B., C.S.C., R.J.R., S.E., S.O., A.P., C.S., J.T., R.W. and P.A.F. and the paper was written by F.L.R., I.G., P.T., M.R.S. and S.T.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
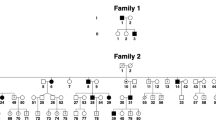
Pedigrees N2–N16 showing number and gender of affected individuals who showed linkage to Xq26–27. (PDF 72 kb)
Supplementary Fig. 2
Eye movement recording in family N1. (PDF 380 kb)
Supplementary Fig. 3
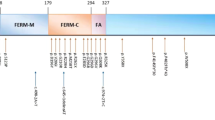
Location of mutations in FRMD7 relative to the B41 and FERM-C domains and the effect of mutations in families N5 and N7. (PDF 169 kb)
Supplementary Table 1
Primer sequences. (PDF 29 kb)
Supplementary Video 1
Eye movement in older brother from family N1. Nystagmus is pronounced in the primary gaze. (WMV 1862 kb)
Supplementary Video 2
Eye movement in younger brother from family N1. Nystagmus is visible only on side gaze. (WMV 2924 kb)
Supplementary Video 3
Eye movement in mother from familiy N1. Nystagmus is absent. (WMV 2002 kb)
Rights and permissions
About this article
Cite this article
Tarpey, P., Thomas, S., Sarvananthan, N. et al. Mutations in FRMD7, a newly identified member of the FERM family, cause X-linked idiopathic congenital nystagmus. Nat Genet 38, 1242–1244 (2006). https://doi.org/10.1038/ng1893
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1893
This article is cited by
-
Truncated FRMD7 proteins in congenital Nystagmus: novel frameshift mutations and proteasomal pathway implications
BMC Medical Genomics (2024)
-
Correlations of FRMD7 gene mutations with ocular oscillations
Scientific Reports (2022)
-
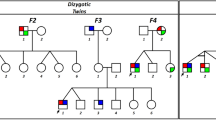
Discordant phenotypes in twins with infantile nystagmus
Scientific Reports (2021)
-
Clinical utility gene card for FRMD7-related infantile nystagmus
European Journal of Human Genetics (2021)
-
Structural variations in a non-coding region at 1q32.1 are responsible for the NYS7 locus in two large families
Human Genetics (2020)