Abstract
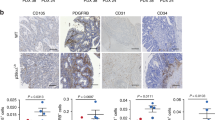
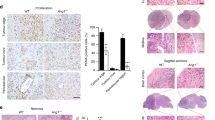
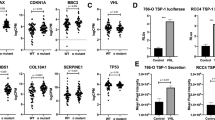
Human adenocarcinomas commonly harbor mutations in the KRAS and MYC proto-oncogenes and the TP53 tumor suppressor gene. All three genetic lesions are potentially pro-angiogenic, as they sustain production of vascular endothelial growth factor (VEGF). Yet Kras-transformed mouse colonocytes lacking p53 formed indolent, poorly vascularized tumors, whereas additional transduction with a Myc-encoding retrovirus promoted vigorous vascularization and growth. In addition, VEGF levels were unaffected by Myc, but enhanced neovascularization correlated with downregulation of anti-angiogenic thrombospondin-1 (Tsp1) and related proteins, such as connective tissue growth factor (CTGF). Both Tsp1 and CTGF are predicted targets for repression by the miR-17-92 microRNA cluster, which was upregulated in colonocytes coexpressing K-Ras and c-Myc. Indeed, miR-17-92 knockdown with antisense 2′-O-methyl oligoribonucleotides partly restored Tsp1 and CTGF expression; in addition, transduction of Ras-only cells with a miR-17-92–encoding retrovirus reduced Tsp1 and CTGF levels. Notably, miR-17-92–transduced cells formed larger, better-perfused tumors. These findings establish a role for microRNAs in non–cell-autonomous Myc-induced tumor phenotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
20 September 2006
In the version of this article initially published, the affiliation of authors Danielle Murphy, William M. Lee, and Greg H. Enders was incorrect. The affiliation for these authors should be 'Department of Medicine', not 'Department of Pathology'. This error has been corrected in the PDF version of this article.
References
Pelengaris, S., Littlewood, T., Khan, M., Elia, G. & Evan, G.I. Reversible activation of c-Myc in skin: induction of a complex neoplastic phenotype by a single oncogenic lesion. Mol. Cell 3, 565–577 (1999).
Brandvold, K.A., Neiman, P. & Ruddell, A. Angiogenesis is an early event in the generation of myc-induced lymphomas. Oncogene 19, 2780–2785 (2000).
Ngo, C. et al. An in vivo function for the transforming myc protein: elicitation of the angiogenic phenotype. Cell Growth Differ. 11, 201–210 (2000).
Baudino, T.A. et al. c-Myc is essential for vasculogenesis and angiogenesis during development and tumor progression. Genes Dev. 16, 2530–2543 (2002).
Ruddell, A., Mezquita, P., Brandvold, K.A., Farr, A. & Iritani, B.M. B lymphocyte-specific c-Myc expression stimulates early and functional expansion of the vasculature and lymphatics during lymphomagenesis. Am. J. Pathol. 163, 2233–2245 (2003).
Knies-Bamforth, U.E., Fox, S.B., Poulsom, R., Evan, G.I. & Harris, A.L. c-Myc interacts with hypoxia to induce angiogenesis in vivo by a vascular endothelial growth factor-dependent mechanism. Cancer Res. 64, 6563–6570 (2004).
Tikhonenko, A.T., Black, D.J. & Linial, M.L. Viral Myc oncoproteins in infected fibroblasts down-modulate thrombospondin-1, a possible tumor suppressor gene. J. Biol. Chem. 271, 30741–30747 (1996).
Janz, A., Sevignani, C., Kenyon, K., Ngo, C. & Thomas-Tikhonenko, A. Activation of the Myc oncoprotein leads to increased turnover of thrombospondin-1 mRNA. Nucleic Acids Res. 28, 2268–2275 (2000).
Watnick, R.S., Cheng, Y.N., Rangarajan, A., Ince, T.A. & Weinberg, R.A. Ras modulates Myc activity to repress thrombospondin-1 expression and increase tumor angiogenesis. Cancer Cell 3, 219–231 (2003).
Sevignani, C. et al. Tumorigenic conversion of p53-deficient colon epithelial cells by an activated Ki-Ras gene. J. Clin. Invest. 101, 1572–1580 (1998).
Thomas-Tikhonenko, A. et al. Myc-transformed epithelial cells down-regulate clusterin which inhibits their growth in vitro and carcinogenesis in vivo. Cancer Res. 64, 3126–3136 (2004).
Tucker, R.P. The thrombospondin type 1 repeat superfamily. Int. J. Biochem. Cell Biol. 36, 969–974 (2004).
Inoki, I. et al. Connective tissue growth factor binds vascular endothelial growth factor (VEGF) and inhibits VEGF-induced angiogenesis. FASEB J. 16, 219–221 (2002).
Perbal, B. CCN proteins: multifunctional signalling regulators. Lancet 363, 62–64 (2004).
Hwang, H.W. & Mendell, J.T. MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br. J. Cancer 94, 776–780 (2006).
O'Donnell, K.A., Wentzel, E.A., Zeller, K.I., Dang, C.V. & Mendell, J.T. c-Myc-regulated microRNAs modulate E2F1 expression. Nature 435, 839–843 (2005).
He, L. et al. A microRNA polycistron as a potential human oncogene. Nature 435, 828–833 (2005).
John, B. et al. Human microRNA targets. PLoS Biol. 2, e363 (2004).
Meister, G., Landthaler, M., Dorsett, Y. & Tuschl, T. Sequence-specific inhibition of microRNA- and siRNA-induced RNA silencing. RNA 10, 544–550 (2004).
Kerbel, R.S. & Folkman, J. Clinical translation of angiogenesis inhibitors. Nat. Rev. Cancer 2, 727–739 (2002).
Rak, J., Yu, J.L., Kerbel, R.S. & Coomber, B.L. What do oncogenic mutations have to do with angiogenesis/vascular dependence of tumors? Cancer Res. 62, 1931–1934 (2002).
Chin, L. et al. Essential role for oncogenic Ras in tumour maintenance. Nature 400, 468–472 (1999).
Ravi, R. et al. Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1 alpha. Genes Dev. 14, 34–44 (2000).
Dameron, K.M., Volpert, O.V., Tainsky, M.A. & Bouck, N. Control of angiogenesis in fibroblasts by p53 regulation of thrombospondin-1. Science 265, 1582–1584 (1994).
Himelstein, B.P., Lee, E.J., Sato, H., Seiki, M. & Muschel, R.J. Transcriptional activation of the matrix metalloproteinase-9 gene in an H-ras and v-myc transformed rat embryo cell line. Oncogene 14, 1995–1998 (1997).
Esquela-Kerscher, A. & Slack, F.J. Oncomirs - microRNAs with a role in cancer. Nat. Rev. Cancer 6, 259–269 (2006).
Volinia, S. et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl. Acad. Sci. USA 103, 2257–2261 (2006).
Hayashita, Y. et al. A polycistronic microRNA cluster, miR-17–92, is overexpressed in human lung cancers and enhances cell proliferation. Cancer Res. 65, 9628–9632 (2005).
Krutzfeldt, J. et al. Silencing of microRNAs in vivo with 'antagomirs'. Nature 438, 685–689 (2005).
Hurwitz, H. et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 350, 2335–2342 (2004).
Acknowledgements
We are indebted to C. Dang (Johns Hopkins University) for fostering collaborations between the investigators. We are grateful to C. Simon and C.-J. Hu (University of Pennsylvania) for providing HIF1α-overexpressing cell lysates for protein blot analysis and J. Tobias (University of Pennsylvania) for his help with microarray data analysis. We thank W. El-Deiry, A. Rustgi, and S. Fuchs (University of Pennsylvania) for stimulating discussions and comments on the manuscript. M. Goldschmidt and J. Burns (University of Pennsylvania) are acknowledged for their help with histopathological analyses. This work was supported by US National Institutes of Health grants CA 097932 and DK 050306 and a University of Pennsylvania Research Foundation grant to A.T.-T.
Author information
Authors and Affiliations
Contributions
This study was designed by M.D. and A.T.-T; experiments with transformed colonocytes were performed by M.D., A.H., and D.Y.; colonocytes lacking p53 were generated by C.S.; some microRNA experments were performed by E.W. and J.T.M.; histopathological analyses were carried out by G.H.E. and E.E.F.; the analysis of blood vessels was performed by D.M. and W.M.L. and the manuscript was co-written by M.D. and A.T.-T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
TSR-encoding mRNAs downregulated by Myc. (PDF 10 kb)
Supplementary Table 2
TSR proteins that are predicted targets of the miR17-92 cluster. (PDF 12 kb)
Supplementary Table 3
Sequences of qRT-PCR primers and 2′-O-methyl oligoribonucleotides. (PDF 19 kb)
Rights and permissions
About this article
Cite this article
Dews, M., Homayouni, A., Yu, D. et al. Augmentation of tumor angiogenesis by a Myc-activated microRNA cluster. Nat Genet 38, 1060–1065 (2006). https://doi.org/10.1038/ng1855
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1855
This article is cited by
-
Are Vascular Endothelium and Angiogenesis Effective MicroRNA Biomarkers Associated with the Prediction of Early-Onset Preeclampsia (EOPE) and Adverse Perinatal Outcomes?
Reproductive Sciences (2024)
-
The various role of microRNAs in breast cancer angiogenesis, with a special focus on novel miRNA-based delivery strategies
Cancer Cell International (2023)
-
c-MYC mediates the crosstalk between breast cancer cells and tumor microenvironment
Cell Communication and Signaling (2023)
-
A deep learning model predicts the presence of diverse cancer types using circulating tumor cells
Scientific Reports (2023)
-
A novel retinoic acid drug, EYE-502, inhibits choroidal neovascularization by targeting endothelial cells and pericytes
Scientific Reports (2023)