Abstract
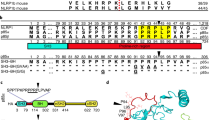
The pathogenesis of Bacillus anthracis, the bacterium that causes anthrax, depends on secretion of three factors that combine to form two bipartite toxins. Edema toxin, consisting of protective antigen (PA) and edema factor (EF), causes the edema associated with cutaneous anthrax infections, whereas lethal toxin (LeTx), consisting of PA and lethal factor (LF), is believed to be responsible for causing death in systemic anthrax infections1. EF and LF can be transported by PA into the cytosol of many cell types2. In mouse macrophages, LF can cause rapid necrosis that may be related to the pathology of systemic infections3,4,5. Inbred mouse strains display variable sensitivity to LeTx-induced macrophage necrosis6,7. This trait difference has been mapped to a locus on chromosome 11 named Ltxs1 (refs. 7,8). Here we show that an extremely polymorphic gene in this locus, Nalp1b, is the primary mediator of mouse macrophage susceptibility to LeTx. We also show that LeTx-induced macrophage death requires caspase-1, which is activated in susceptible, but not resistant, macrophages after intoxication, suggesting that Nalp1b directly or indirectly activates caspase-1 in response to LeTx.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Stephen, J. Anthrax toxin. Pharmacol. Ther. 12, 501–513 (1981).
Collier, R.J. & Young, J.A. Anthrax toxin. Annu. Rev. Cell Dev. Biol. 19, 45–70 (2003).
Friedlander, A.M. Macrophages are sensitive to anthrax lethal toxin through an acid-dependent process. J. Biol. Chem. 261, 7123–7126 (1986).
Hanna, P.C., Kochi, S. & Collier, R.J. Biochemical and physiological changes induced by anthrax lethal toxin in J774 macrophage-like cells. Mol. Biol. Cell 3, 1269–1277 (1992).
Hanna, P.C., Acosta, D. & Collier, R.J. On the role of macrophages in anthrax. Proc. Natl. Acad. Sci. USA 90, 10198–10201 (1993).
Friedlander, A.M., Bhatnagar, R., Leppla, S.H., Johnson, L. & Singh, Y. Characterization of macrophage sensitivity and resistance to anthrax lethal toxin. Infect. Immun. 61, 245–252 (1993).
Roberts, J.E., Watters, J.W., Ballard, J.D. & Dietrich, W.F. Ltx1, a mouse locus that influences the susceptibility of macrophages to cytolysis caused by intoxication with Bacillus anthracis lethal factor, maps to chromosome 11. Mol. Microbiol. 29, 581–591 (1998).
Watters, J.W. & Dietrich, W.F. Genetic, physical, and transcript map of the Ltxs1 region of mouse chromosome 11. Genomics 73, 223–231 (2001).
Watters, J.W., Dewar, K., Lehoczky, J., Boyartchuk, V. & Dietrich, W.F. Kif1C, a kinesin-like motor protein, mediates mouse macrophage resistance to anthrax lethal factor. Curr. Biol. 11, 1503–1511 (2001).
Summerton, J. & Weller, D. Morpholino antisense oligomers: design, preparation, and properties. Antisense Nucleic Acid Drug Dev. 7, 187–195 (1997).
Draper, B.W., Morcos, P.A. & Kimmel, C.B. Inhibition of zebrafish fgf8 pre-mRNA splicing with morpholino oligos: a quantifiable method for gene knockdown. Genesis 30, 154–156 (2001).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol. Cell 10, 417–426 (2002).
Cordoba-Rodriguez, R., Fang, H., Lankford, C.S. & Frucht, D.M. Anthrax lethal toxin rapidly activates caspase-1/ICE and induces extracellular release of interleukin (IL)-1β and IL-18. J. Biol. Chem. 279, 20563–20566 (2004).
Tschopp, J., Martinon, F. & Burns, K. Nalps: a novel protein family involved in inflammation. Nat. Rev. Mol. Cell Biol. 4, 95–104 (2003).
Martinon, F., Hofmann, K. & Tschopp, J. The pyrin domain: a possible member of the death domain-fold family implicated in apoptosis and inflammation. Curr. Biol. 11, R118–R120 (2001).
Geddes, B.J. et al. Human CARD12 is a novel CED4/Apaf-1 family member that induces apoptosis. Biochem. Biophys. Res. Commun. 284, 77–82 (2001).
Poyet, J.-L. et al. Identification of Ipaf, a human caspase-1-activating protein related to Apaf-1. J. Biol. Chem. 276, 28309–28313 (2001).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Brennan, M.A. & Cookson, B.T. Salmonella induces macrophage death by caspase-1-dependent necrosis. Mol. Microbiol. 38, 31–40 (2000).
Jesenberger, V., Procyk, K.J., Yuan, J., Reipert, S. & Baccarini, M. Salmonella-induced caspase-2 activation in macrophages: a novel mechanism in pathogen-mediated apoptosis. J. Exp. Med. 192, 1035–1045 (2000).
Klimpel, K.R., Arora, N. & Leppla, S.H. Anthrax toxin lethal factor contains a zinc metalloprotease consensus sequence which is required for lethal toxin activity. Mol. Microbiol. 13, 1093–1100 (1994).
Vitale, G., Bernardi, L., Napolitani, G., Mock, M. & Montecucco, C. Susceptibility of mitogen-activated protein kinase kinase family members to proteolysis by anthrax lethal factor. Biochem. J. 352, 739–745 (2000).
Pellizzari, R., Guidi-Rontani, C., Vitale, G., Mock, M. & Montecucco, C. Anthrax lethal factor cleaves MKK3 in macrophages and inhibits LPS/IFNγ-induced release of NO and TNFα. FEBS Lett. 462, 199–204 (1999).
Kim, S.O. et al. Sensitizing anthrax lethal toxin-resistant macrophages to lethal toxin-induced killing by tumor necrosis factor-α. J. Biol. Chem. 278, 7413–7421 (2003).
Menard, A., Papini, E., Mock, M. & Montecucco, C. The cytotoxic activity of Bacillus anthracis lethal factor is inhibited by leukotriene A4 hydrolase and metallopeptidase inhibitors. Biochem. J. 320, 687–691 (1996).
Growney, J.D. & Dietrich, W.F. High-resolution genetic and physical map of the Lgn1 interval in C57BL/6J implicates Naip2 or Naip5 in Legionella pneumophila pathogenesis. Genome Res. 10, 1158–1171 (2000).
Yui, M.A. et al. Production of congenic mouse strains carrying NOD-derived diabetogenic genetic intervals: an approach for the genetic dissection of complex traits. Mamm. Genome 7, 331–334 (1996).
Reifsnyder, P.C. et al. Genotypic and phenotypic characterization of six new recombinant congenic strains derived from NOD/Shi and CBA/J genomes. Mamm. Genome 10, 161–167 (1999).
Svenson, K.L. et al. Strain distribution pattern for SSLP markers in the SWXJ recombinant inbred strain set: chromosomes 1 to 6. Mamm. Genome 6, 867–872 (1995).
Shultz, K.L., Svenson, K.L., Cheah, Y.-C., Paigen, B. & Beamer, W.G. Strain distribution pattern for SSLP markers in the SWXJ recombinant inbred strain set: chromosomes 7 to X. Mamm. Genome 7, 526–532 (1996).
Acknowledgements
We thank A. Abney, S. Goodart, E. Kazyanskaya, K. McAuliffe and K. Sadigh for technical assistance; J. Watters and members of the Dietrich laboratory for helpful discussion and S. Boyden, L. Boyden, N. Andrews, F. Ausubel, L. Kunkel and R. Mosher for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Translated sequence alignment of Nalp1b alleles 1–5. (PDF 74 kb)
Supplementary Table 1
Primer and morpholino sequences. (PDF 71 kb)
Rights and permissions
About this article
Cite this article
Boyden, E., Dietrich, W. Nalp1b controls mouse macrophage susceptibility to anthrax lethal toxin. Nat Genet 38, 240–244 (2006). https://doi.org/10.1038/ng1724
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1724
This article is cited by
-
Involvement of inflammasomes in tumor microenvironment and tumor therapies
Journal of Hematology & Oncology (2023)
-
The NLR gene family: from discovery to present day
Nature Reviews Immunology (2023)
-
Integrated NLRP3, AIM2, NLRC4, Pyrin inflammasome activation and assembly drive PANoptosis
Cellular & Molecular Immunology (2023)
-
NLRP1- A CINDERELLA STORY: a perspective of recent advances in NLRP1 and the questions they raise
Communications Biology (2023)
-
Virulence-related gene wx2 of Toxoplasma gondii regulated host immune response via classic pyroptosis pathway
Parasites & Vectors (2022)