Abstract
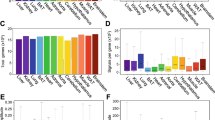
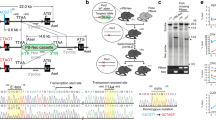
Mammalian circadian clocks consist of complexly integrated regulatory loops1,2,3,4,5, making it difficult to elucidate them without both the accurate measurement of system dynamics and the comprehensive identification of network circuits6. Toward a system-level understanding of this transcriptional circuitry, we identified clock-controlled elements on 16 clock and clock-controlled genes in a comprehensive surveillance of evolutionarily conserved cis elements and measurement of their transcriptional dynamics. Here we report the roles of E/E′ boxes, DBP/E4BP4 binding elements7 and RevErbA/ROR binding elements8 in nine, seven and six genes, respectively. Our results indicate that circadian transcriptional circuits are governed by two design principles: regulation of E/E′ boxes and RevErbA/ROR binding elements follows a repressor-precedes-activator pattern, resulting in delayed transcriptional activity, whereas regulation of DBP/E4BP4 binding elements follows a repressor-antiphasic-to-activator mechanism, which generates high-amplitude transcriptional activity. Our analysis further suggests that regulation of E/E′ boxes is a topological vulnerability in mammalian circadian clocks, a concept that has been functionally verified using in vitro phenotype assay systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gekakis, N. et al. Role of the CLOCK protein in the mammalian circadian mechanism. Science 280, 1564–1569 (1998).
Kume, K. et al. mCRY1 and mCRY2 are essential components of the negative limb of the circadian clock feedback loop. Cell 98, 193–205 (1999).
Shearman, L.P. et al. Interacting molecular loops in the mammalian circadian clock. Science 288, 1013–1019 (2000).
Preitner, N. et al. The orphan nuclear receptor REV-ERBalpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell 110, 251–260 (2002).
Sato, T.K. et al. A functional genomics strategy reveals Rora as a component of the mammalian circadian clock. Neuron 43, 527–537 (2004).
Kitano, H. Systems biology: a brief overview. Science 295, 1662–1664 (2002).
Falvey, E., Marcacci, L. & Schibler, U. DNA-binding specificity of PAR and C/EBP leucine zipper proteins: a single amino acid substitution in the C/EBP DNA-binding domain confers PAR-like specificity to C/EBP. Biol. Chem. 377, 797–809 (1996).
Harding, H.P. & Lazar, M.A. The orphan receptor Rev-ErbA alpha activates transcription via a novel response element. Mol. Cell. Biol. 13, 3113–3121 (1993).
Panda, S., Hogenesch, J.B. & Kay, S.A. Circadian rhythms from flies to human. Nature 417, 329–335 (2002).
Reppert, S.M. & Weaver, D.R. Coordination of circadian timing in mammals. Nature 418, 935–941 (2002).
King, D.P. et al. Positional cloning of the mouse circadian clock gene. Cell 89, 641–653 (1997).
Bunger, M.K. et al. Mop3 is an essential component of the master circadian pacemaker in mammals. Cell 103, 1009–1017 (2000).
Bae, K. et al. Differential functions of mPer1, mPer2, and mPer3 in the SCN circadian clock. Neuron 30, 525–536 (2001).
Zheng, B. et al. Nonredundant roles of the mPer1 and mPer2 genes in the mammalian circadian clock. Cell 105, 683–694 (2001).
van der Horst, G.T. et al. Mammalian Cry1 and Cry2 are essential for maintenance of circadian rhythms. Nature 398, 627–630 (1999).
Lowrey, P.L. et al. Positional syntenic cloning and functional characterization of the mammalian circadian mutation tau. Science 288, 483–492 (2000).
Mitsui, S., Yamaguchi, S., Matsuo, T., Ishida, Y. & Okamura, H. Antagonistic role of E4BP4 and PAR proteins in the circadian oscillatory mechanism. Genes Dev. 15, 995–1006 (2001).
Wuarin, J. & Schibler, U. Expression of the liver-enriched transcriptional activator protein DBP follows a stringent circadian rhythm. Cell 63, 1257–1266 (1990).
Honma, S. et al. Dec1 and Dec2 are regulators of the mammalian molecular clock. Nature 419, 841–844 (2002).
Zylka, M.J., Shearman, L.P., Weaver, D.R. & Reppert, S.M. Three period homologs in mammals: differential light responses in the suprachiasmatic circadian clock and oscillating transcripts outside of brain. Neuron 20, 1103–1110 (1998).
Reick, M., Garcia, J.A., Dudley, C. & McKnight, S.L. NPAS2: an analog of clock operative in the mammalian forebrain. Science 293, 506–509 (2001).
Ueda, H.R. et al. A transcription factor response element for gene expression during circadian night. Nature 418, 534–539 (2002).
Honma, S. et al. Circadian oscillation of BMAL1, a partner of a mammalian clock gene Clock, in rat suprachiasmatic nucleus. Biochem. Biophys. Res. Commun. 250, 83–87 (1998).
Lee, C., Etchegaray, J.P., Cagampang, F.R., Loudon, A.S. & Reppert, S.M. Posttranslational mechanisms regulate the mammalian circadian clock. Cell 107, 855–867 (2001).
Tei, H. et al. Circadian oscillation of a mammalian homologue of the Drosophila period gene. Nature 389, 512–516 (1997).
Shearman, L.P., Zylka, M.J., Weaver, D.R., Kolakowski, L.F. Jr. & Reppert, S.M. Two period homologs: circadian expression and photic regulation in the suprachiasmatic nuclei. Neuron 19, 1261–1269 (1997).
Panda, S. et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell 109, 307–320 (2002).
Milo, R. et al. Network motifs: simple building blocks of complex networks. Science 298, 824–827 (2002).
Albert, R., Jeong, H. & Barabasi, A.L. Error and attack tolerance of complex networks. Nature 406, 378–382 (2000).
Jeong, H., Mason, S.P., Barabasi, A.L. & Oltvai, Z.N. Lethality and centrality in protein networks. Nature 411, 41–42 (2001).
Acknowledgements
We thank H. Tei and Y. Sakaki for Arntl, Clock and Cry1 expression vectors; T. Kojima, T. Katakura and H. Urata for technical assistance; and D. Sipp and Y. Minami for critical reading of the manuscript. This research was done as part of a research and development project of the Industrial Science and Technology Program supported by the New Energy and Industrial Technology Development Organization and, in part, by intramural Grant-in-Aid from the CDB, Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan, Grant-in-Aid for Strategic Programs in R & D from the Institute of Physical and Chemical Research and Grant-in-Aid for Scientific Research from the New Energy and Industrial Technology Development Organization.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Temporal expression profiles of 16 transcription factors in the suprachiasmatic nucleus and liver under constant darkness conditions. (PDF 144 kb)
Supplementary Fig. 2
Feedback and feedforward loops in the transcriptional circuits underlying mammalian circadian rhythms. (PDF 121 kb)
Supplementary Fig. 3
Positive and negative control in an in vitro circadian dynamics monitoring system. (PDF 137 kb)
Supplementary Fig. 4
Confirmation of evolutionary conserved E-box/E'-boxes on clock/clock-controlled genes. (PDF 21 kb)
Supplementary Fig. 5
Confirmation of evolutionary conserved D-boxes on clock/clock-controlled genes. (PDF 10 kb)
Supplementary Fig. 6
Confirmation of evolutionary conserved RREs on clock/clock-controlled genes. (PDF 20 kb)
Supplementary Fig. 7
Confirmation of evolutionary conserved E-box/E'-box and RREs on Cry1 and Rorc genes. (PDF 77 kb)
Rights and permissions
About this article
Cite this article
Ueda, H., Hayashi, S., Chen, W. et al. System-level identification of transcriptional circuits underlying mammalian circadian clocks. Nat Genet 37, 187–192 (2005). https://doi.org/10.1038/ng1504
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1504
This article is cited by
-
Oscillatory dynamics of the mammalian circadian clock induced by the core delayed negative feedback loop
Nonlinear Dynamics (2024)
-
Potential role of Akt in the regulation of fibroblast growth factor 21 by berberine
Journal of Natural Medicines (2023)
-
Midnight/midday-synchronized expression of cryptochrome genes in the eyes of three teleost species, zebrafish, goldfish, and medaka
Zoological Letters (2022)
-
Diurnal RNAPII-tethered chromatin interactions are associated with rhythmic gene expression in rice
Genome Biology (2022)
-
Rhythmic transcription of Bmal1 stabilizes the circadian timekeeping system in mammals
Nature Communications (2022)