Abstract
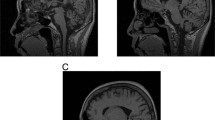
The genetic aetiologies of at least 20% of autosomal dominant spinocerebellar ataxias (SCAs) have yet to be elucidated1. We have recently identified a novel form of autosomal dominant SCA, termed SCA12, in a large pedigree ('R') of German descent. The phenotype is variable, but the prototypical phenotype is that of a classic spinocerebellar ataxia, and the disease resembles the spinocerebellar ataxias more closely than any other form of neurodegenerative disorder. Age of onset ranges from 8 to 55 years. Most individuals present in the fourth decade with upper extremity tremor, progressing over several decades to include head tremor, gait ataxia, dysmetria, dysdiadokinesis, hyperreflexia, paucity of movement, abnormal eye movements and, in the oldest subjects, dementia. MRI or CT scans of five cases indicate both cortical and cerebellar atrophy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Schols, L. et al. Ann. Neurol. 42, 924– 932 (1997).
Schalling, M., Hudson, T.J., Buetow, K.W. & Housman, D.E. Nature Genet. 4, 135–139 (1993).
Margolis, R.L., McInnis, M.G., Rosenblatt, A. & Ross, C.A. Arch. Gen. Psychiatry 56, 1019–1031 (1999).
Koob, M.D. et al. Nature Genet. 18, 72– 75 (1998).
Ott, J. Analysis of Human Genetic Linkage (Johns Hopkins University Press, Baltimore, 1991).
Mayer, R.E. et al. Biochemistry 30, 3589– 3597 (1991).
Strack, S., Zaucha, J.A., Ebner, F.F., Colbran, R.J. & Wadzinski, B.E. J. Comp. Neurol. 392, 515–527 (1998).
Millward, T.A., Zolnierowicz, S. & Hemmings, B.A. Trends Biochem. Sci. 24, 186 –191 (1999).
Ince, T.A. & Scotto, K.W. J. Biol. Chem. 270, 30249–30252 (1995).
Burke, T.W., Willy, P.J., Kutach, A.K., Butler, J.E. & Kadonaga, J.T. Cold Spring Harb. Symp. Quant. Biol. 63, 75–82 ( 1998).
Trottier, Y. et al. Nature 378, 403–406 (1995).
Mayer-Jaekel, R.E. et al. Cell 72, 621–633 (1993).
Sontag, E. et al. J. Biol. Chem. 274, 25490– 25498 (1999).
Deng, X., Ito, T., Carr, B., Mumby, M. & May, W.S. Jr J. Biol. Chem. 273, 34157–34163 (1998).
Santoro, M.F. et al. J. Biol. Chem. 273, 13119– 13128 (1998).
Acknowledgements
We thank family members and their physicians for cooperation; T. Swift-Scanlan, S. Wang, L. Monfredo, V. Willour, F. Guarnieri, B. Hemmings, B. Wadzinski, O.C. Stine, S. Reich and A. Scott for assistance; R. Seeger for LA-N-1 cells for control experiments; L. Schoels, S. Krueger and W. Berger for collection of German control subjects; and P.R. McHugh for support and encouragement. This work was supported by grants from the Huntington's Disease Society of America and NIH NS16375, MH01275 and MH50763.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Holmes, S., O'Hearn, E., McInnis, M. et al. Expansion of a novel CAG trinucleotide repeat in the 5′ region of PPP2R2B is associated with SCA12. Nat Genet 23, 391–392 (1999). https://doi.org/10.1038/70493
Issue Date:
DOI: https://doi.org/10.1038/70493
This article is cited by
-
Fusion of 3D feature extraction techniques to enhance classification of spinocerebellar ataxia type 12
International Journal of Information Technology (2024)
-
Investigation of RFC1 tandem nucleotide repeat locus in diverse neurodegenerative outcomes in an Indian cohort
neurogenetics (2023)
-
The length of uninterrupted CAG repeats in stem regions of repeat disease associated hairpins determines the amount of short CAG oligonucleotides that are toxic to cells through RNA interference
Cell Death & Disease (2022)
-
Rapid and comprehensive diagnostic method for repeat expansion diseases using nanopore sequencing
npj Genomic Medicine (2022)
-
Use of single guided Cas9 nickase to facilitate precise and efficient genome editing in human iPSCs
Scientific Reports (2021)