Abstract
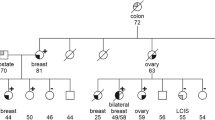
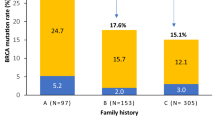
BRCA1 and BRCA2 are the two major identified causes of inherited breast cancer, with mutations in either gene conferring up to 80–90% lifetime risk of breast cancer in carrier females1–4. Mutations in BRCA1 account for approximately 45% of familial breast cancer and 90% of inherited breast/ovarian cancer1,2, whereas mutations in BRCA2 account for a comparable percentage of inherited breast cancer cases3,4. Over 85 distinct BRCA1 mutations and a growing list of BRCA2 mutations have been identified, with the majority resulting in protein truncation5. A specific BRCA1 mutation, 185delAG, has a reported increased carrier frequency of approximately 0.9% in the Ashkenazi Jewish population6, but is also found in rare non-Jewish patients with a different haplotype7. The 6174delT mutation in BRCA2 was recently identified as a frequent mutation in 8 out of 107 Ashkenazi Jewish women diagnosed with breast cancer by age 50 (ref. 8), as well as in three Ashkenazi male breast cancer patients9. We have conducted a large-scale population study to investigate the prevalence of specific BRCA1 and BRCA2 mutations in Ashkenazi Jewish individuals who were unselected for breast cancer. BRCA1 mutation screening on approximately 3,000 Ashkenazi Jewish samples determined a carrier frequency of 1.09% for the 185delAG mutation and 0.13% for the 5382insC mutation. BRCA2 analysis on 3,085 individuals from the same population showed a carrier frequency of 1.52% for the 6174delT mutation. This expanded population-based study confirms that the BRCA1 185delAG mutation and the BRCA2 6174delT mutation constitute the two most frequent mutation alleles predisposing to hereditary breast cancer among the Ashkenazim, and suggests a relatively lower penetrance for the 6174delT mutation in BRCA2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Miki, Y. et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 266, 66–71 (1994).
Szabo, C.I. & King, M.-C. Inherited breast and ovarian cancer. Hum. Mol. Genet. 4, 1811–1817 (1995).
Wooster, R. et al. Identification of the breast cancer susceptibility gene BRCA2. Nature 378, 789–792 (1995).
Tavtigian, S.V. et al. The complete BRCA2 gene and mutations in chromosome 13q-linked kindreds. Nature Genet. 12, 333–337 (1996).
The Breast Cancer Information Core http://www. nchgr. nih. gov/ lntramural_research/Lab_transfer/Bic/index. html
Struewing, J.P. et al. The carrier frequency of the BRCA1 185delAG mutation is approximately 1 percent in Ashkenazi Jewish individuals. Nature Genet. 11, 198–200 (1995).
Berman, D.B. et al. Two distinct origins of a common BRCA1 mutation in breast-ovarian cancer families: a genetic study of 15 185delAG-mutation kindreds. Am. J. Hum. Genet. 58, 1166–1176 (1996).
Neuhausen, S. et al. Recurrent BRCA2 6174delT mutations in Ashkenazi Jewish women affected by breast cancer. Nature Genet. 13, 126–128 (1996).
Couch, F.J. et al. BRCA2 germiine mutations in male breast cancer cases and breast cancer families. Nature Genet. 13, 123–125 (1996).
DeMarchi, J.M., Caskey, C.T. & Richards, C.S. Population screening by mutation analysis for diseases frequent in Ashkenazi Jews. Hum. Mut. (in the press).
Fitzgerald, M.G. et al. Germ-line BRCA1 mutations in Jewish and non-Jewish women with early-onset breast cancer. N. Engl. J. Med. 334, 143–149 (1996).
Easton, D.F. et al. Genetic linkage analysis in familial breast and ovarian cancer: results from 214 families. Am. J. Hum. Genet. 52, 678–701 (1993).
Feuer, E.J. et al. The lifetime risk of developing breast cancer. J. Natn. Canc. Inst. 85, 892–897 (1993).
DeMarchi, J.M., Richards, C.S., Fenwick, R.G., Pace, R. & Beaudet, A.L. A robotics-assisted procedure for large scale cystic fibrosis mutation analysis. Human Mut. 4, 281–290 (1994).
Kogan, S.C., Doherty, M. & Gitschier, J. An improved method for prenatal diagnosis of genetic diseases by analysis of amplified DNA sequences. Application to hemophilia A. N. Engl. J. Med. 317, 985–990 (1987).
Friedman, L.S. et al. Confirmation of BRCA1 by analysis of germiine mutations linked to breast and ovarian cancer in ten families. Nature Genet. 8, 399–404 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Roa, B., Boyd, A., Volcik, K. et al. Ashkenazi Jewish population frequencies for common mutations in BRCA1 and BRCA2. Nat Genet 14, 185–187 (1996). https://doi.org/10.1038/ng1096-185
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1096-185
This article is cited by
-
Variable number tandem repeats (VNTRs) as modifiers of breast cancer risk in carriers of BRCA1 185delAG
European Journal of Human Genetics (2023)
-
Racial and ethnic variation in BRCA1 and BRCA2 genetic test results among individuals referred for genetic counseling at a large urban comprehensive cancer center
Cancer Causes & Control (2023)
-
Analysis of pathogenic variants in BRCA1 and BRCA2 genes using next-generation sequencing in women with triple negative breast cancer from South India
Molecular Biology Reports (2022)
-
Rate of breast biopsy referrals in female BRCA mutation carriers aged 50 years or more: a retrospective comparative study and matched analysis
Breast Cancer Research and Treatment (2022)
-
Newly synthesized metal complexes of sulfonamides: DNA cleavage, BRCA1 gene interaction, expression analysis, antioxidant and antiglycation studies
Chemical Papers (2022)