Abstract
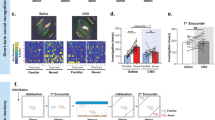
The development of social familiarity in rodents depends predominantly on olfactory cues and can critically influence reproductive success1,2. Researchers have operationally defined this memory by a reliable decrease in olfactory investigation in repeated or prolonged encounters with a conspecific3,4,5,6. Brain oxytocin (OT) and vasopressin (AVP) seem to modulate a range of social behaviour from parental care to mate guarding7. Pharmacological studies indicate that AVP administration may enhance social memory8,9,10, whereas OT administration may either inhibit or facilitate social memory depending on dose, route or paradigm1,11,12,13. We found that male mice mutant for the oxytocin gene (Oxt−/−) failed to develop social memory, whereas wild-type (Oxt+/+) mice showed intact social memory. Measurement of both olfactory foraging and olfactory habituation tasks indicated that olfactory detection of non-social stimuli is intact in Oxt−/− mice. Spatial memory and behavioural inhibition measured in a Morris water-maze, Y-maze, or habituation of an acoustic startle also seemed intact. Treatment with OT but not AVP rescued social memory in Oxt−/− mice, and treatment with an OT antagonist produced a social amnesia-like effect in Oxt+/+ mice. Our data indicate that OT is necessary for the normal development of social memory in mice and support the hypothesis that social memory has a neural basis distinct from other forms of memory.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kendrick, K.M. et al. Neural control of maternal behaviour and olfactory recognition of offspring. Brain Res. Bull. 44, 383–395 (1997).
Keverne, E.B. & Brennan, P.A. Olfactory recognition memory. J. Physiol. Paris 90, 399–401 (1996).
Thor, D.H. & Holloway, W.R. Jr Persistence of social investigatory behavior in the male rat: Evidence for long-term memory of initial copulatory experience. Anim. Learn. Behav. 9, 28–42 (1981).
Gheusi, G., Bluthe, R.M., Goodall, G. & Dantzer, R. Social and individual recognition in rodents: methodological aspects and neurobiological bases. Behav. Proc. 33, 59–87 (1994).
Winslow, J.T. & Camacho, F. Cholinergic modulation of a decrement in social investigation following repeated contacts between mice. Psychopharmacol. (Berl) 121, 164–172 (1995).
Dluzen, D.E. & Kreutzberg, J.D. 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) disrupts social memory/recognition processes in the male mouse. Brain Res. 609, 98–102 (1993).
Insel, T.R., Winslow, J.T., Wang, Z. & Young, L.J. Oxytocin, vasopressin, and the neuroendocrine basis of pair bond formation. Adv. Exp. Med. Biol. 449, 215–224 (1998).
Popik, P. & van Ree, J.M. Neurohypophyseal peptides and social recognition in rats. Prog. Brain Res. 119, 415–436 (1998).
Dantzer, R., Bluthe, R.M., Koob, G.F. & Le Moal, M. Modulation of social memory in male rats by neurohypophyseal peptides. Psychopharmacol. (Berl) 91, 363–368 (1987).
Dantzer, R. Vasopressin, gonadal steroids and social recognition. Prog. Brain Res. 119, 409–414 (1998).
Popik, P. & van Ree, J.M. Oxytocin but not vasopressin facilitates social recognition following injection into the medial preoptic area of the rat brain. Eur. Neuropsychopharmacol. 1, 555–560 (1991).
Engelmann, M., Ebner, K., Wotjak, C.T. & Landgraf, R. Endogenous oxytocin is involved in short-term olfactory memory in female rats. Behav. Brain Res. 90, 89–94 (1998).
Dluzen, D.E., Muraoka, S. & Landgraf, R. Olfactory bulb norepinephrine depletion abolishes vasopressin and oxytocin preservation of social recognition responses in rats. Neurosci. Lett. 254, 161–164 (1998).
Jirikowski, G.F., Caldwell, J.D., Haussler, H.U. & Pedersen, C.A. Mating alters topography and content of oxytocin immunoreactivity in male mouse brain. Cell Tissue Res. 266, 399–403 (1991).
Kubota, Y. et al. Structure and expression of the mouse oxytocin receptor gene. Mol. Cell. Endocrinol. 124, 25–32 (1996).
Keverne, E.B. The vomeronasal organ. Science 286, 716–720 (1999).
Yu, G.Z., Kaba, H., Okutani, F., Takahashi, S. & Higuchi, T. The olfactory bulb: a critical site of action for oxytocin in the induction of maternal behaviour in the rat. Neuroscience 72, 1083–1088 (1996).
Levy, F., Kendrick, K.M., Goode, J.A., Guevara-Guzman, R. & Keverne, E.B. Oxytocin and vasopressin release in the olfactory bulb of parturient ewes: changes with maternal experience and effects on acetylcholine, γ-aminobutyric acid, glutamate and noradrenaline release. Brain Res. 669, 197–206 (1995).
Engelmann, M., Wotjak, C.T., Neumann, I., Ludwig, M. & Landgraf, R. Behavioral consequences of intracerebral vasopressin and oxytocin: focus on learning and memory. Neurosci. Biobehav. Rev. 20, 341–358 (1996).
Popik, P., Vetulani, J. & Van Ree, J.M. Social memory and neurohypophyseal hormones. Eur. Neuropsychopharmacol. 3, 200–201 (1993).
Benelli, A. et al. Polymodal dose-response curve for oxytocin in the social recognition test. Neuropeptides 28, 251–255 (1995).
Popik, P., Vetulani, J. & Van Ree, J.M. Low doses of oxytocin facilitate social recognition in rats. Psychopharmacol. (Berl) 106, 71–74 (1992).
Popik, P. & Vetulani, J. Opposite action of oxytocin and its peptide antagonists on social memory in rats. Neuropeptides 18, 23–27 (1991).
Popik, P., Vos, P.E. & Van Ree, J.M. Neurohypophyseal hormone receptors in the septum are implicated in social recognition in the rat. Behav. Pharmacol. 3, 351–358 (1992).
Nishimori, K. et al. Oxytocin is required for nursing but is not essential for parturition or reproductive behavior. Proc. Natl Acad. Sci. USA 93, 11699–11704 (1996).
Mencio-Wszalek, T., Ramirez, V.D. & Dluzen, D.E. Age-dependent changes in olfactory-mediated behavioral investigations in the male rat. Behav. Neural Biol. 57, 205–212 (1992).
Paolini, A.G. & McKenzie, J.S. Effects of lesions in the horizontal diagonal band nucleus on olfactory habituation in the rat. Neuroscience 57, 717–724 (1993).
Paolini, A.G. & McKenzie, J.S. Lesions in the magnocellular preoptic nucleus decrease olfactory investigation in rats. Behav. Brain Res. 81, 223–231 (1996).
Smith, C.P. et al. Pharmacological activity and safety profile of P10358, a novel, orally active acetylcholinesterase inhibitor for Alzheimer's disease. J. Pharmacol. Exp. Ther. 280, 710–720 (1997).
Contarino, A. et al. Reduced anxiety-like and cognitive performance in mice lacking the corticotropin-releasing factor receptor 1. Brain Res. 835, 1–9 (1999).
Acknowledgements
Portions of this research were supported by National Institute of Health Yerkes Regional Primate Research Center Base Grant RR0016, National Institute of Mental Health Grant RO1-M-56538-01 and funds from the National Alliance for Autism Research. The Oxt−/− mice were initially generated with funds from National Institute of Health grant HD33438.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferguson, J., Young, L., Hearn, E. et al. Social amnesia in mice lacking the oxytocin gene. Nat Genet 25, 284–288 (2000). https://doi.org/10.1038/77040
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77040
This article is cited by
-
A prefrontal-thalamic circuit encodes social information for social recognition
Nature Communications (2024)
-
A systematic review and meta-analysis of how social memory is studied
Scientific Reports (2024)
-
Oxytocin activity in the paraventricular and supramammillary nuclei of the hypothalamus is essential for social recognition memory in rats
Molecular Psychiatry (2023)
-
Social circuits and their dysfunction in autism spectrum disorder
Molecular Psychiatry (2023)
-
A dorsal CA2 to ventral CA1 circuit contributes to oxytocinergic modulation of long-term social recognition memory
Journal of Biomedical Science (2022)