Abstract
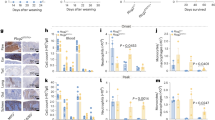
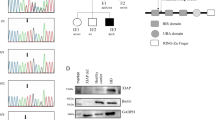
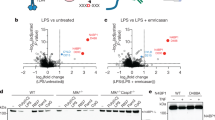
The alymphoplasia (aly) mutation of mouse is autosomal recessive and characterized by the systemic absence of lymph nodes (LN) and Peyer's patches (PP) and disorganized splenic and thymic structures with immunodeficiency1,2,3. Although recent reports have shown that the interaction between lymphotoxin (LT) and the LT β-receptor (Ltβr, encoded by Ltbr ) provides a critical signal for LN genesis in mice4,5,6,7,8,9,10, the aly locus on chromosome 11 (ref. 11) is distinct from those for LT and its receptor. We found that the aly allele carries a point mutation causing an amino acid substitution in the carboxy-terminal interaction domain of Nf-κb-inducing kinase12,13 (Nik, encoded by the gene Nik). Transgenic complementation with wild-type Nik restored the normal structures of LN, PP, spleen and thymus, and the normal immune response in aly/aly mice. In addition, the aly mutation in a kinase domain-truncated Nik abolished its dominant-negative effect on Nf-κb activation induced by an excess of Ltβr. Our observations agree with previous reports that Ltβr-deficient mice showed defects in LN genesis4 and that Nik is a common mediator of Nf-κb activation by the tumour necrosis factor (TNF) receptor family12,13. Nik is able to interact with members of the TRAF family (Traf1, 2, 3, 5 and 6; ref. 13), suggesting it acts downstream of TRAF-associating receptor signalling pathways, including Tnfr ( ref. 12), Cd40 (Refs 14, 15), Cd30 (Refs 16, 17) and Ltβr (refs 18, 19, 20, 21). The phenotypes of aly/aly mice are more severe than those of Ltbr–/– mice, however, indicating involvement of Nik in signal transduction mediated by other receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Miyawaki, S. et al. A new mutation, aly, that induces a generalized lack of lymph nodes accompanied by immunodeficiency in mice. Eur. J. Immunol. 24, 429–434 ( 1994).
Shinkura, R. et al. Defects of somatic hypermutation and class switching in alymphoplasia (aly) mutant. Int. Immunol. 8, 1067 –1075 (1996).
Koike, R. et al. The splenic marginal zone is absent in alymphoplastic aly mutant mice. Eur. J. Immunol. 26, 669 –675 (1996).
Fütterer, A., Mink, K., Luz, A., Kosco-Vilbois, M.H. & Pfeffer, K. The lymphotoxin β receptor controls organogenesis and affinity maturation in peripheral lymphoid tissues. Immunity 9, 59–70 (1998 ).
Rennert, P.D., Browning, J.L., Mebius, R., Mackey, F. & Hochman, P.S. Surface lymphotoxin α/β complex is required for the development of peripheral lymphoid organs. J. Exp. Med. 184, 1999–2006 (1996).
Rennert, P.D., Browning, J.L. & Hochman, P.S. Selective disruption of lymphotoxin ligands reveals a novel set of mucosal lymph nodes and unique effects on lymph node cellular organization. Int. Immunol. 9, 1627– 1639 (1997).
De Togni, P. et al. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin α. Science 264, 703–707 (1994).
Banks, T.A. et al. Lymphotoxin-α-deficient mice: effects on secondary lymphoid organ development and humoral immune responsiveness. J. Immunol. 155, 1685–1693 ( 1995).
Koni, P.A. et al. Distinct roles in lymphoid organogenesis for lymphotoxins α and β revealed in lymphotoxin β-deficient mice. Immunity 6, 491–500 ( 1997).
Eugster, H.-P. et al. Multiple immune abnormalities in tumor necrosis factor and lymphotoxin-α double-deficient mice. Int. Immunol. 8, 23–36 (1996).
Kuramoto, T. et al. The alymphoplasia (aly) mutation co-segregates with the intercellular adhesion molecule-2 (Icam-2) on mouse chromosome 11. Int. Immunol. 6, 991–994 (1994).
Malinin, N.L., Boldin, M.P., Kovalenko, A.V. & Wallach, D. MAP3K-related kinase involved in NF-κB induction by TNF, CD95 and IL-1. Nature 385, 540–544 (1997).
Song, H.Y., Régnier, C.H., Kirschning, C.J., Goeddel, D.V. & Rothe, M. Tumor necrosis factor (TNF)-mediated kinase cascades: bifurcation of nuclear factor-κB and c-jun N-terminal kinase (JNK/SAPK) pathways at TNF receptor-associated factor 2. Proc. Natl Acad. Sci. USA 94, 9792– 9796 (1997).
Cheng, G. et al. Involvement of CRAF1, a relative of TRAF, in CD40 signaling. Science 267, 1494–1498 (1995).
Ishida, T. et al. TRAF5, a novel tumor necrosis factor receptor-associated factor family protein, mediates CD40 signaling. Proc. Natl Acad. Sci. USA 93, 9437–9442 ( 1996).
Duckett, C.S., Gedrich, R.W., Gilfillan, M.C. & Thompson, C.B. Induction of nuclear factor κB by the CD30 receptor is mediated by TRAF1 and TRAF2. Mol. Cell. Biol. 17, 1535– 1542 (1997).
Lee, S.Y. et al. CD30/TNF receptor-associated factor interaction: NF-κB activation and binding specificity. Proc. Natl Acad. Sci. USA 93, 9699–9703 (1996).
Nakano, H. et al. TRAF5, an activator of NF-κB and putative signal transducer for the lymphotoxin-β receptor. J. Biol. Chem. 271, 14661–14664 (1996).
VanArsdale, T.L. et al. Lymphotoxin-β receptor signaling complex: role of tumor necrosis factor receptor-associated factor 3 recruitment in cell death and activation of nuclear factor κB. Proc. Natl Acad. Sci. USA 94, 2460–2465 ( 1997).
Force, W.R., Cheung, T.C. & Ware, C.F. Dominant negative mutants of TRAF3 reveal an important role for the coiled coil domains in cell death signaling by the lympnotoxin-β receptor. J. Biol. Chem. 272, 30835– 30840 (1997).
Krajewska, M. et al. TRAF-4 expression in epithelial progenitor cells. Analysis in normal adult, fetal, and tumor tissues. Am. J. Pathol. 152, 1549–1561 (1998).
Kvist, S., Roberts, L. & Dobberstein, B. Mouse histocompatibility genes: structure and organization of a Kd gene. EMBO J. 2, 245–254 (1983).
Lin, X. et al. Molecular determinants of NF-κB-inducing kinase action. Mol. Cell. Biol. 18, 5899–5907 (1998).
Beutler, B., Milsark, I.W. & Cerami, A.C. Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science 229, 869–871 (1985).
Nishi, M., Ishida, Y. & Honjo, T. Expression of functional interleukin-2 receptors in human light chain/Tac transgenic mice. Nature 331, 267–269 (1988).
Nakamura, T. et al. The murine lymphotoxin-β receptor cDNA: isolation by the signal sequence trap and chromosomal mapping. Genomics 30, 312–319 (1995).
Kosco, M.H., Pflugfelder, E. & Gray, D. Follicular dendritic cell-dependent adhesion and proliferation of B cells in vitro. J. Immunol. 148, 2331 –2339 (1992).
Schreiber, E., Mattias, P., Muller, M.M. & Schaffuer, W. Rapid detection of octamer binding proteins with 'mini-extracts', prepared from a small number of cells. Nucleic Acids Res. 17 , 6419 (1989).
Acknowledgements
We thank A. Shimizu, S. Nishikawa and H. Yoshida for discussions and technical suggestions; S. Miyawaki for animals; M. Kosco-Vilbois for FDC-M1 antibody; S. Miyamoto for Nf-κb-dependent luciferase reporter gene plasmids; M. Yamazaki, K. Watanabe, C. Sugawara and Y. Fukuzumi for large-scale sequencing; M. Tanaka, N. Tomikawa and H. Kurooka for technical assistance; and K. Fukui for help in preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shinkura, R., Kitada, K., Matsuda, F. et al. Alymphoplasia is caused by a point mutation in the mouse gene encoding Nf-κb-inducing kinase. Nat Genet 22, 74–77 (1999). https://doi.org/10.1038/8780
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/8780
This article is cited by
-
Functions of MAP3Ks in antiviral immunity
Immunologic Research (2023)
-
A T cell-intrinsic function for NF-κB RelB in experimental autoimmune encephalomyelitis
Scientific Reports (2021)
-
Fibroblasts as a source of self-antigens for central immune tolerance
Nature Immunology (2020)
-
The roles of TRAF3 mutation in the oncogenic progression and drug response of multiple myeloma
Genome Instability & Disease (2020)
-
A NIK–SIX signalling axis controls inflammation by targeted silencing of non-canonical NF-κB
Nature (2019)