Abstract
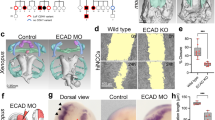
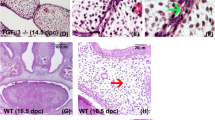
Craniofacial malformations are among the most frequent congenital birth defects in humans; cleft palate, that is inadequate fusion of the palatal shelves, occurs with an annual incidence of 1 in 700 to 1 in 1,000 live births among individuals of European descent1. The secondary palate arises as bilateral outgrowths from the maxillary processes2, and its formation depends on the coordinated development of craniofacial structures including the Meckel's cartilage and the mandible3. Cleft lip and palate syndromes in humans are associated with polymorphisms in the gene (TGFA) encoding transforming growth factor-α (TGF-α), an epidermal growth factor receptor (EGFR) ligand made by most epithelia1,4. Here we have characterized craniofacial development in Egfr -deficient (Egfr-/-) mice. Newborn Egfr-/- mice have facial mediolateral defects including narrow, elongated snouts, underdeveloped lower jaw and a high incidence of cleft palate. Palatal shelf explants from Egfr-/- mice fused, but frequently had residual epithelium in the midline. In addition, morphogenesis of Meckel's cartilage was deficient in cultured mandibular processes from Egfr-/- embryos. The secretion of matrix metalloproteinases (MMPs) was diminished in Egfr-/- explants, consistent with the ability of EGF to increase MMP secretion and with the decreased MMP expression caused by inhibition of Egfr signalling in wild-type explants. Accordingly, inactivation of MMPs in wild-type explants phenocopied the defective morphology of Meckel's cartilage seen in Egfr-/- explants. Our results indicate that EGFR signalling is necessary for normal craniofacial development and that its role is mediated in part by its downstream targets, the MMPs, and may explain the genetic correlation of human cleft palate with polymorphisms in TGFA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chenevix-Trench, G., Jones, K., Green, A.C., Duffy, D.L. & Martin, N.G. Cleft lip with or without cleft palate: associations with transforming growth factor α and retinoic acid receptor loci. Am. J. Hum. Genet. 51, 1377–1385 (1992).
Ferguson, M.W.J. Palate development. Development 103 (suppl.), 41–60 (1988).
Seegmiller, R.E. & Fraser, F.C. Mandibular growth retardation as a cause of cleft palate in mice homozygous for the chondrodysplasia gene. J. Embryol. Exp. Morphol. 38, 227– 238 (1977).
Shiang, R. et al. Association of transforming growth-factor α gene polymorphism with nonsyndromic cleft palate only (CPO). Am. J. Hum. Genet. 53, 836–843 (1993).
Iamaroon, A., Tait, B. & Diewert, V.M. Cell proliferation and expression of EGF, TGF-α, and EGF receptor in the developing primary palate. J. Dent. Res. 75, 1534–1539 ( 1996).
Dixon, M.J. & Ferguson, M.W.J. The effects of epidermal growth factor, transforming growth factor α and β and platelet-derived growth factor on murine palatal shelves in organ culture. Arch. Oral Biol. 37, 395–410 ( 1992).
Shum, L. et al. EGF abrogation-induced fusilli-form dysmorphogenesis of Meckel's cartilage during embryonic mouse mandibular morphogenesis in vitro. Development 118, 903–917 (1993).
Partanen, A.M. & Thesleff, I. Localization and quantitation of 125I-epidermal growth factor binding in mouse embryonic tooth and other embryonic tissues at different developmental stages. Dev. Biol. 120, 186–197 ( 1987).
Miettinen, P.J. et al. Epithelial immaturity and multiorgan failure in mice lacking epidermal growth factor receptor. Nature 376, 337–341 (1995).
Shuler, C.F., Guo, Y., Majumder, A. & Luo, R. Molecular and morphologic changes during the epithelial-mesenchymal transformantion of palatal shelf medial edge epithelium in vitro. Int. J. Dev. Biol. 35, 463–472 (1991).
Shuler, C.F. Programmed cell death and cell transformation in craniofacial development. Crit. Rev. Oral Biol. Med. 6, 202– 217 (1995).
Chin, J.R. & Werb, Z. Matrix metalloproteinases regulate morphogenesis, migration and remodeling of epithelium, tongue, skeletal muscle and cartilage in the mandibular arch. Development 124 , 1519–1530 (1997).
van der Zee, E., Jansen, I., Hoeben, K., Beertsen, W. & Everts, V. EGF and IL-1 α modulate the release of collagenase, gelatinase and TIMP-1 as well as the release of calcium by rabbit calvarial bone explants. J. Periodontal Res. 33, 65 –72 (1998).
Sibilia, M. & Wagner, E. Strain-dependent epithelial defects in mice lacking EGF receptor. Science 269, 234–238 (1995).
Threadgill, D.W. et al. Targeted disruption of mouse EGF receptor: effect of genetic background on mutant phenotype. Science 269, 230–234 (1995).
Wilson, J.B. et al. Transgenic mouse model of X-linked cleft palate. Cell Growth Differ. 4, 67–76 (1993).
Diehl, S.R. & Erickson, R.P. Genome scan for teratogen-induced clefting susceptibility loci in the mouse: evidence of both allelic and locus heterogeneity distinguishing cleft lip and palate. Proc. Natl Acad. Sci. USA 94, 5231–5236 ( 1997).
Neel, J.V. A study of major congenital defects in Japanese infants. Am. J. Hum. Genet. 10, 398–445 (1958).
Chung, C.S. & Myrianthopoulos, N.C. Racial and prenatal factors in major congenital malformations. Am. J. Hum. Genet. 20, 44–60 (1968).
Christensen, K., Schmidt, M.M., Væth, M. & Olsen, J. Absence of an environmental effect on the recurrence of facial-cleft defects. N. Engl. J. Med. 333, 161– 164 (1995).
Abbott, B.D. Review of the interaction between TCDD and glucocorticoids in embryonic palate. Toxicology 105, 365–373 (1995).
Abbott, B.D. & Pratt, R.M. Retinoids and EGF alter embryonic mouse palatal epithelial and mesenchymal cell differentiation in organ culture. J. Craniofac. Genet. Dev. Biol. 7, 219– 240 (1987).
Satokata, I. & Maas, R. Msx1-deficient mice exhibit cleft palate and abnormalities of craniofacial and tooth development. Nature Genet. 6, 348–355 (1994).
Kaartinen, V. et al. Abnormal lung development and cleft palate in mice lacking TGF-β3 indicates defects of epthelial-mesenchymal interaction. Nature Genet. 11, 415–421 (1995).
Lidral, A.C. et al. Association of Msx1 and Tgfβ3 with nonsyndromic clefting in humans. Am. J. Hum. Genet. 63, 557–568 (1998).
Mitchell, L.E. Transforming growth factor α locus and non-syndromic cleft lip with or without cleft palate: a reappraisal. Genet. Epidemiol. 14, 231–240 (1997).
Ardinger, H.H. et al. Association of genetic variation of the transforming growth factor-α gene with cleft lip and palate. Am. J. Hum. Genet. 45, 348–353 ( 1989).
Sheehan, D.C. & Hrapchak, B.B. Theory and Practice of Histotechnology (C.V. Mosby, St. Louis, 1980).
Acknowledgements
We thank J. Berger for technical assistance and J. Helms for a critical reading of the manuscript. This work was supported by grants from the National Institutes of Health (DE10306 to Z.W. and CA54826 to R.D.; AR41114 to L.S. and H.S.) and the Academy of Finland (P.J.M.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miettinen, P., Chin, J., Shum, L. et al. Epidermal growth factor receptor function is necessary for normal craniofacial development and palate closure. Nat Genet 22, 69–73 (1999). https://doi.org/10.1038/8773
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/8773
This article is cited by
-
New insights into the genetics of mandibular retrognathism: novel candidate genes
Journal of Orofacial Orthopedics / Fortschritte der Kieferorthopädie (2024)
-
Inactive rhomboid proteins RHBDF1 and RHBDF2 (iRhoms): a decade of research in murine models
Mammalian Genome (2021)
-
An integrative, genomic, transcriptomic and network-assisted study to identify genes associated with human cleft lip with or without cleft palate
BMC Medical Genomics (2020)
-
Transferrin receptor facilitates TGF-β and BMP signaling activation to control craniofacial morphogenesis
Cell Death & Disease (2016)
-
Mesenchymal signaling in dorsoventral differentiation of palatal epithelium
Cell and Tissue Research (2015)